


API Suppliers

US DMFs Filed

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0
0
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
0
USFDA Orange Book Patents
0
USFDA Exclusivities
0
Blog #PharmaFlow
0
News
0
EDQM
0
USP
0
JP
0
Other Listed Suppliers
0
0
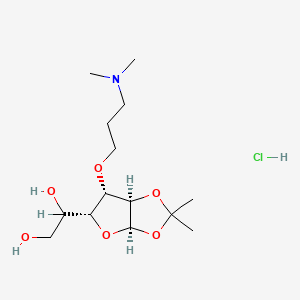

1. 1,2-o-isopropylidene-3-o-3'-(n',n'-dimethylamino-n-propyl)-d-glucofuranose
2. 3-o-(3-(dimethylamino)propyl)-1,2-o-(1-methylethylidene)- Alpha-d-glucofuranose
3. Amiprilose
4. Amprilose
5. Sm 1213
6. Sm-1213
7. Therafectin
1. Dsstox_cid_25883
2. Dsstox_rid_81198
3. Dsstox_gsid_45883
4. 1-[(3ar,5r,6s,6ar)-6-[3-(dimethylamino)propoxy]-2,2-dimethyl-3a,5,6,6a-tetrahydrofuro[2,3-d][1,3]dioxol-5-yl]ethane-1,2-diol;hydrochloride
5. Sr-01000075667
6. Ncgc00095000-01
7. Cas-60414-06-4
8. Schembl41474
9. Chembl3184686
10. Dtxsid5045883
11. Tox21_111382
12. Tox21_111382_1
13. Lp00047
14. Ncgc00162052-05
15. Eu-0100047
16. A 4687
17. Sr-01000075667-1
18. Sr-01000075667-5
19. 1,2-o-isopropylidene-3-o-[3'-(n,n-dimethylamino)propyl]-alpha-d-glucofuranose Hydrochloride
| Molecular Weight | 341.83 g/mol |
|---|---|
| Molecular Formula | C14H28ClNO6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Exact Mass | 341.1605153 g/mol |
| Monoisotopic Mass | 341.1605153 g/mol |
| Topological Polar Surface Area | 80.6 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 337 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
Adjuvants, Immunologic
Substances that augment, stimulate, activate, potentiate, or modulate the immune response at either the cellular or humoral level. The classical agents (Freund's adjuvant, BCG, Corynebacterium parvum, et al.) contain bacterial antigens. Some are endogenous (e.g., histamine, interferon, transfer factor, tuftsin, interleukin-1). Their mode of action is either non-specific, resulting in increased immune responsiveness to a wide variety of antigens, or antigen-specific, i.e., affecting a restricted type of immune response to a narrow group of antigens. The therapeutic efficacy of many biological response modifiers is related to their antigen-specific immunoadjuvanticity. (See all compounds classified as Adjuvants, Immunologic.)
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)


