
Synopsis
Synopsis
0
JDMF
0
KDMF
0
VMF
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
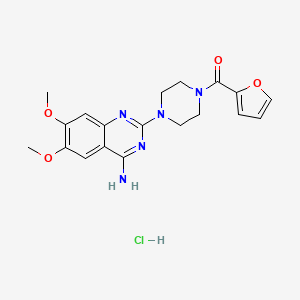

1. Furazosin
2. Hcl, Prazosin
3. Hydrochloride, Prazosin
4. Minipress
5. Pratsiol
6. Prazosin
7. Prazosin Hcl
1. 19237-84-4
2. Prazosin Hcl
3. Minipress
4. Vasoflex
5. Peripress
6. Furazosin Hydrochloride
7. Minipress Xl
8. Hypovase
9. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furanylcarbonyl)piperazine Hydrochloride
10. Cp-12299-1
11. Prazosin (hydrochloride)
12. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl)piperazine Monohydrochloride
13. Cp-12,299-1
14. Prazosinhydrochloride
15. (4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl)(furan-2-yl)methanone Hydrochloride
16. [4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl]-(furan-2-yl)methanone;hydrochloride
17. Mls000028454
18. Piperazine, 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furanylcarbonyl)-, Monohydrochloride
19. Nsc-292810
20. X0z7454b90
21. Deprazolin
22. Hypovasole
23. Pratsiol
24. Sinetens
25. Smr000058384
26. Minipress (tn)
27. 19237-84-4 (hcl)
28. 2-(4-(2-furoyl)piperazin-1-yl)-4-amino-6,7-dimethoxyquinazoline Hydrochloride
29. 2-{4-[(furan-2-yl)carbonyl]piperazin-1-yl}-6,7-dimethoxyquinazolin-4-amine Hydrochloride
30. Chebi:8365
31. Hsdb 3298
32. Methanone, [4-(4-amino-6,7-dimethoxy-2-quinazolinyl)-1-piperazinyl]-2-furanyl-, Hydrochloride (1:1)
33. C19h21n5o4.hcl
34. Nsc 292810
35. Prazosin Clorhidrato [spanish]
36. Prazosine Hydrochloride
37. Sr-01000003052
38. Einecs 242-903-4
39. Mfcd00058177
40. Unii-x0z7454b90
41. Pressin And Hypovase
42. Prazosin Hydrochloride [usan:usp:jan]
43. Prazosin Hydrochloride;
44. Opera_id_370
45. Chembl1558
46. Piperazine, 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl)-, Monohydrochloride
47. Schembl41257
48. Mls001148201
49. Mls001333696
50. Mls002153280
51. Mls002222304
52. Spectrum1500495
53. Prazosin Hcl (furazosin Hcl)
54. Piperazin-1-yl)(furan-2-yl)
55. Hy-b0193a
56. Dtxsid50172822
57. Hms1570n16
58. Hms1920j10
59. Pharmakon1600-01500495
60. Prazosin Hydrochloride [mi]
61. Bcp13643
62. Prazosin Hydrochloride (jp17/usp)
63. Prazosin Hydrochloride [jan]
64. Tox21_501002
65. Ac-699
66. Ccg-39227
67. Nsc292810
68. Nsc757286
69. Prazosin Hydrochloride [hsdb]
70. Prazosin Hydrochloride [usan]
71. S1424
72. Prazosin Hydrochloride [vandf]
73. Akos015895394
74. Prazosin Hydrochloride [mart.]
75. Af-0012
76. H37p844
77. Lp01002
78. Nsc-757286
79. Prazosin Hydrochloride [usp-rs]
80. Prazosin Hydrochloride [who-dd]
81. Sb17352
82. Prazosin Hydrochloride, >=99% (tlc)
83. Quinazoline, 4-amino-6,7-dimethoxy-2-(4-(2-furoyl)piperazin-1-yl)-, Hydrochloride
84. Ncgc00094296-01
85. Ncgc00094296-02
86. Ncgc00094296-03
87. Ncgc00094296-04
88. Ncgc00094296-05
89. Ncgc00261687-01
90. Ba164294
91. Db-044795
92. Prazosin Hydrochloride [ep Impurity]
93. Prazosin Hydrochloride [orange Book]
94. Eu-0101002
95. P0938
96. Prazosin Hydrochloride [ep Monograph]
97. Prazosin Hydrochloride [usp Monograph]
98. D00609
99. H11453
100. Minizide Component Prazosin Hydrochloride
101. Prazosin Hydrochloride 100 Microg/ml In Methanol
102. Prazosin Hydrochloride Component Of Minizide
103. Sr-01000003052-2
104. Sr-01000003052-8
105. W-107719
106. Q27108060
107. Piperazine,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl)-, Monohydrochloride
108. Prazosin Hydrochloride, European Pharmacopoeia (ep) Reference Standard
109. Prazosin Hydrochloride, United States Pharmacopeia (usp) Reference Standard
110. 1-(4-amino-6,7-dimethoxy-2-quinazol Inyl)-4-(2-furanylcarbonyl)piperazine Hydrochloride
111. Piperazine,7-dimethoxy-2-quinazolinyl)-4-(2-furanylcarbonyl)-, Monohydrochloride
| Molecular Weight | 419.9 g/mol |
|---|---|
| Molecular Formula | C19H22ClN5O4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | 419.1360319 g/mol |
| Monoisotopic Mass | 419.1360319 g/mol |
| Topological Polar Surface Area | 107 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 544 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Prazosin hydrochloride |
| Drug Label | Prazosin hydrochloride, a quinazoline derivative, is the first of a new chemical class of antihypertensives. It is the hydrochloride salt of 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl) piperazine and its structural formula is:Prazosin hydro... |
| Active Ingredient | Prazosin hydrochloride |
| Dosage Form | Capsule |
| Route | oral; Oral |
| Strength | eq 5mg base; eq 2mg base; eq 1mg base |
| Market Status | Prescription |
| Company | Ivax Sub Teva Pharms; Teva Pharms; Mylan |
| 2 of 2 | |
|---|---|
| Drug Name | Prazosin hydrochloride |
| Drug Label | Prazosin hydrochloride, a quinazoline derivative, is the first of a new chemical class of antihypertensives. It is the hydrochloride salt of 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl) piperazine and its structural formula is:Prazosin hydro... |
| Active Ingredient | Prazosin hydrochloride |
| Dosage Form | Capsule |
| Route | oral; Oral |
| Strength | eq 5mg base; eq 2mg base; eq 1mg base |
| Market Status | Prescription |
| Company | Ivax Sub Teva Pharms; Teva Pharms; Mylan |
Adrenergic alpha-Antagonists; Antihypertensive Agents; Sympatholytics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Prazosin hydrochloride is used in the management of hypertension. Prazosin's efficacy in hypertensive patients is similar to that of thiazide diuretics, beta-adrenergic blocking agents, hydralazine, and centrally acting adrenergic inhibitors (eg, clonidine, methyldopa). ... Prazosin has been shown to be effective in the management of hypertension in patients undergoing chronic hemodialysis, and the drug may be particularly useful in the acute management of severe hypertension in patients with increased concentrations of circulating catecholamines.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1860
Prazosin has been used with good results alone or in combination with a beta-adrenergic blocking agent for the preoperative management of the signs and symptoms of pheochromocytoma in a limited number of patients; however, these patients may be particularly susceptible to a marked hypotensive response to the initial dose of prazosin. Limited data also suggest that prazosin may be useful for the treatment of Raynaud's disease or phenomenon and ergotamine induced peripheral ischemia.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1861
Prazosin may be used as an adjunct to digoxin and diuretics for the treatment of congestive heart failure. However, prazosin has not been shown to improve survival in these patients. /NOT included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 22nd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2002. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 2430
For more Therapeutic Uses (Complete) data for PRAZOSIN HYDROCHLORIDE (11 total), please visit the HSDB record page.
May cause syncope with sudden loss of consciousness, usually attributable to an excessive postural hypotensive effect; risk may be reduced by limiting the initial dose of the drug to 1 mg, and by subsequently increasing the dosage slowly. Adverse reactions include dizziness, drowsiness, and other CNS effects; patients should be cautioned regarding activities such as driving and operating machinery, as well as interactions with other CNS acting drugs including alcohol. Other adverse reactions include palpitations and nausea. The concurrent use of a beta-adrenergic blocking agent may increase the risk of hypotension. /Prazosin hydrochloride/
Hussar, D.A. (ed.). Modell's Drugs in Current Use and New Drugs. 38th ed. New York, NY: Springer Publishing Co., 1992., p. 135
When prazosin is used as a fixed-combination preparation that includes polythiazide, the cautions, precautions, and contraindications associated with thiazide diuretics must be considered in addition to those associated with prazosin.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1862
Caution should be used when adding prazosin to a preexisting antihypertensive regimen or when adding other hypotensive agents to a prazosin regimen in order to avoid a possible rapid fall in blood pressure. Caution should be used when administering prazosin to patients with chronic renal failure as they may require only small doses of the drug.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1862
Prazosin has been used alone or in combination with other hypotensive agents for the management of severe hypertension in a limited number of pregnant women without apparent adverse effect on the fetus. There are no adequate and well controlled studies to date using prazosin in pregnant women, however, and the drug should be used during pregnancy only when the potential benefits justify the possible risks to the fetus.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1862
For more Drug Warnings (Complete) data for PRAZOSIN HYDROCHLORIDE (18 total), please visit the HSDB record page.
Adrenergic alpha-1 Receptor Antagonists
Drugs that bind to and block the activation of ADRENERGIC ALPHA-1 RECEPTORS. (See all compounds classified as Adrenergic alpha-1 Receptor Antagonists.)
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
There is intraindividual and interindividual variation in the rate of absorption and plasma concentrations of prazosin. The absolute oral bioavailability of prazosin is also variable but is reported to average about 60% (range: 43-82%). Results of one study indicate that the presence of food may delay absorption of the drug in some patients, but does not affect the extent of absorption.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
Following oral administration of prazosin hydrochloride, plasma concentrations of the drug reach a peak in 2-3 hrs in most fasting patients. Plasma concentrations of prazosin generally do not correlate with therapeutic effect. One manufacturer reports that plasma concentrations of the drug after a single 5 mg dose range from 0.01-0.075 ug/ml. Blood pressure begins to decrease within 2 hr after an oral dose; the maximum decrease occurs in 2-4 hr. The hypotensive effect of prazosin lasts less than 24 hr.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
Animal studies indicate that prazosin is widely distributed in body tissues. After iv administration in dogs, highest concentrations of the drug are found in the lungs, coronary arteries, aorta, paw arteries and heart; the lowest concentrations are in the brain. During prazosin therapy, approximately 97% of the drug in plasma is bound to proteins. It is not known whether the drug crosses the placenta. Prazosin is distributed into milk in small amounts.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
Approximately 6-10% of a dose is excreted in urine and the remainder in feces via bile.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
The drug /prazosin hydrochloride/ is tightly bound to plasma proteins (primarily alpha1-acid glycoprotein), and only 5% of the drug is free in the circulation; diseases that modify the concentration of this protein (e.g., inflammatory processes) may change the free fraction. Prazosin is extensively metabolized in the liver, and little unchanged drug is excreted by the kidneys.
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill, 2001., p. 246
Animal studies show that prazosin hydrochloride is metabolized extensively in the liver, principally by demethylation and conjugation, and excreted as unchanged drug (5-11%) and metabolites. Four of the metabolites have been shown to possess 10-25% of the hypotensive activity of prazosin and they may contribute to the antihypertensive effect of the drug.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
The 6-O-demethyl and 7-O-demethyl analogues of the new antihypertensive drug prazosin [2-[4-(2-furoyl)-piperazin-1-yl]-4-amino-6,7-dimethoxyquinazoline hydrochloride] have been unequivocally synthesized via separate 10-step reaction sequences starting from isovanillin and vanillin, respectively. The 6-O-demethyl derivative was found to be identical with the major prazosin metabolite formed in dog and rat, while the 7-O-demethyl derivative was identical with another, less prevalent but significant metabolite. Two minor metabolites of prazosin, 2-(1-piperazinyl)-4-amino-6,7-dimethoxyquinazoline and 2,4-diamino-6,7-dimethoxyquinazoline, are also described. All 4 metabolites are less potent blood pressure lowering agents in dogs than prazosin but may contribute to its antihypertensive effect, since they account for a major portion of the administered dose.
PMID:833813 Althius T, Hess H; J Med Chem 20 (1): 146-9 (1977)
Elimination half-life approx 3 hr.
Haddad, L.M., Clinical Management of Poisoning and Drug Overdose. 2nd ed. Philadelphia, PA: W.B. Saunders Co., 1990., p. 1348
The plasma half-life of prazosin after oral administration has been reported to be 2-4 hr.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1863
Animal studies indicate that prazosin does not have its antihypertensive effect in the CNS. Prazosin does not interfere with nerve impulse transmission across sympathetic ganglia nor does it cause adrenergic neuronal blockade.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 1862
The exact mechanism of the hypotensive action of prazosin is unknown. Prazosin causes a decrease in total peripheral resistance and was originally thought to have a direct relaxant action on vascular smooth muscle. Animal studies have suggested that the vasodilator effect of prazosin is also related to blockade of postsynaptic alpha-adrenoceptors. The results of dog forelimb experiments demonstrate that that the peripheral vasodilator effect of prazosin is confined mainly to the level of the resistance vessels (arterioles). Unlike conventional alpha- blockers, the antihypertensive action of prazosin is usually not accompanied by reflex tachycardia.
Medical Economics Co; Physicians Desk Reference 56th ed p. 2699 (2002)
Prazosin ... is a very potent and selective alpha 1-adrenergic antagonist. ... Interestingly, the drug also is a relatively potent inhibitor of cyclic nucleotide phosphodiesterases...
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill, 2001., p. 246
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
59
PharmaCompass offers a list of Prazosin Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Prazosin Hydrochloride manufacturer or Prazosin Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Prazosin Hydrochloride manufacturer or Prazosin Hydrochloride supplier.
A Alpress LP manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Alpress LP, including repackagers and relabelers. The FDA regulates Alpress LP manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Alpress LP API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Alpress LP manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Alpress LP supplier is an individual or a company that provides Alpress LP active pharmaceutical ingredient (API) or Alpress LP finished formulations upon request. The Alpress LP suppliers may include Alpress LP API manufacturers, exporters, distributors and traders.
click here to find a list of Alpress LP suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Alpress LP DMF (Drug Master File) is a document detailing the whole manufacturing process of Alpress LP active pharmaceutical ingredient (API) in detail. Different forms of Alpress LP DMFs exist exist since differing nations have different regulations, such as Alpress LP USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Alpress LP DMF submitted to regulatory agencies in the US is known as a USDMF. Alpress LP USDMF includes data on Alpress LP's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Alpress LP USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Alpress LP suppliers with USDMF on PharmaCompass.
A Alpress LP CEP of the European Pharmacopoeia monograph is often referred to as a Alpress LP Certificate of Suitability (COS). The purpose of a Alpress LP CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Alpress LP EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Alpress LP to their clients by showing that a Alpress LP CEP has been issued for it. The manufacturer submits a Alpress LP CEP (COS) as part of the market authorization procedure, and it takes on the role of a Alpress LP CEP holder for the record. Additionally, the data presented in the Alpress LP CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Alpress LP DMF.
A Alpress LP CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Alpress LP CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Alpress LP suppliers with CEP (COS) on PharmaCompass.
A Alpress LP written confirmation (Alpress LP WC) is an official document issued by a regulatory agency to a Alpress LP manufacturer, verifying that the manufacturing facility of a Alpress LP active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Alpress LP APIs or Alpress LP finished pharmaceutical products to another nation, regulatory agencies frequently require a Alpress LP WC (written confirmation) as part of the regulatory process.
click here to find a list of Alpress LP suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Alpress LP as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Alpress LP API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Alpress LP as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Alpress LP and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Alpress LP NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Alpress LP suppliers with NDC on PharmaCompass.
Alpress LP Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Alpress LP GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Alpress LP GMP manufacturer or Alpress LP GMP API supplier for your needs.
A Alpress LP CoA (Certificate of Analysis) is a formal document that attests to Alpress LP's compliance with Alpress LP specifications and serves as a tool for batch-level quality control.
Alpress LP CoA mostly includes findings from lab analyses of a specific batch. For each Alpress LP CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Alpress LP may be tested according to a variety of international standards, such as European Pharmacopoeia (Alpress LP EP), Alpress LP JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Alpress LP USP).

