
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
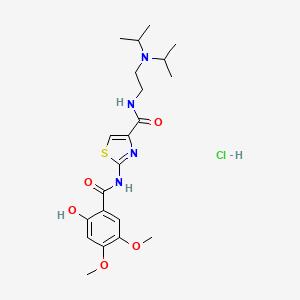

1. Acotiamide
2. N-(2-(diisopropylamino)ethyl)-2-((2-hydroxy-4,5-dimethoxybenzoyl)amino)-1,3-thiazole-4-carboxamide
3. N-(n',n'-diisopropylaminoethyl)-(2-(2-hydroxy-4,5-dimethoxybenzoylamino)-1,3-thiazole-4-yl)carboxyamide
4. Ym 443
5. Ym-443
6. Ym443 Cpd
7. Z 338
8. Z-338
1. 185104-11-4
2. Acotiamide Hcl
3. Acotiamide (hydrochloride)
4. Z338
5. N-[2-[di(propan-2-yl)amino]ethyl]-2-[(2-hydroxy-4,5-dimethoxybenzoyl)amino]-1,3-thiazole-4-carboxamide;hydrochloride
6. Mfcd23103502
7. 510791nn30
8. Ym 443
9. 4-thiazolecarboxamide, N-(2-(bis(1-methylethyl)amino)ethyl)-2-((2-hydroxy-4,5-dimethoxybenzoyl)amino)-, Monohydrochloride
10. N-[2-(diisopropylamino)ethyl]-2-(2-hydroxy-4,5-dimethoxybenzamido)thiazole-4-carboxamide Hydrochloride
11. Unii-510791nn30
12. N-(2-(diisopropylamino)ethyl)-2-(2-hydroxy-4,5-dimethoxybenzamido)thiazole-4-carboxamide Hydrochloride
13. Acotiamidehydrochloride
14. Z-338 Dihydrochloride
15. Acotiamide Dihydrochloride
16. Z 338
17. Schembl6968566
18. Dtxsid30171717
19. Acotiamide Hydrochloride [mi]
20. Akos030529148
21. Hy-121467a
22. Sb19647
23. Acotiamide Hydrochloride [who-dd]
24. Acotiamide Hydrochloride Anhydrous
25. Ds-19577
26. N-(n',n'-diisopropylaminoethyl)-(2-(2-hydroxy-4,5-dimethoxybenzoylamino)-1,3-thiazole-4-yl)carboxyamide
27. N-[2-[bis(1-methylethyl)amino]ethyl]-2-[(2-hydroxy-4,5-dimethoxybenzoyl)amino]-4-thiazolecarboxamide Hydrochloride
28. Sy226332
29. Acotiamide Dihydrochloride, >=98% (hplc)
30. Cs-0103547
31. F14757
32. J-011862
33. Q27260838
34. 4-thiazolecarboxamide, N-(2-(bis(1-methylethyl)amino)ethyl)-2-((2-hydroxy-4,5-dimethoxybenzoyl)amino)-, Hydrochloride (1:1)
35. N-[2-[bis(1-methylethyl)amino]ethyl]-2-[(2-hydroxy-4,5-dimethoxybenzoyl)amino]-4-thiazolecarboxamide
1. Acofide
2. Acotiamide Hydrochloride Hydrate
| Molecular Weight | 487.0 g/mol |
|---|---|
| Molecular Formula | C21H31ClN4O5S |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 10 |
| Exact Mass | 486.1703690 g/mol |
| Monoisotopic Mass | 486.1703690 g/mol |
| Topological Polar Surface Area | 141 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 586 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Cholinesterase Inhibitors
Drugs that inhibit cholinesterases. The neurotransmitter ACETYLCHOLINE is rapidly hydrolyzed, and thereby inactivated, by cholinesterases. When cholinesterases are inhibited, the action of endogenously released acetylcholine at cholinergic synapses is potentiated. Cholinesterase inhibitors are widely used clinically for their potentiation of cholinergic inputs to the gastrointestinal tract and urinary bladder, the eye, and skeletal muscles; they are also used for their effects on the heart and the central nervous system. (See all compounds classified as Cholinesterase Inhibitors.)
Gastrointestinal Agents
Drugs used for their effects on the gastrointestinal system, as to control gastric acidity, regulate gastrointestinal motility and water flow, and improve digestion. (See all compounds classified as Gastrointestinal Agents.)
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 32458
Submission : 2018-01-22
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20144
Submission : 2007-01-12
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Acotiamide Hydrochloride Hydrate IH
Date of Issue : 2026-02-16
Valid Till : 2029-02-01
Written Confirmation Number : WC-0392
Address of the Firm : Unit-ll, Plot no-15, Jawaharlal Nehru Pharma City, Tadi(V), Parwada(M), Anakapal...

Acotiamide Hydrochloride Hydrate IH
Date of Issue : 2024-03-01
Valid Till : 2027-02-28
Written Confirmation Number : WC-0474
Address of the Firm : Village Bhondsi, Tehsil Sohna, District Gurgaon -122102, Haryana, India

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
About the Company : Established in 2004, Metrochem API is one of the fastest-growing APIs, pellets & intermediates manufacturers. It has 6 dedicated manufacturing facilities for its 3 core product gro...
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
About the Company : Cohance Lifesciences is a leading CDMO and API platform delivering products and services across the full molecule lifecycle, from development to commercialization. With strong expe...
 Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
About the Company : Honour is a leading global CDMO and trusted manufacturer of specialty chemicals and ingredients, with seven world-class facilities meeting global safety and quality standards. Thro...
 Lewens Labs transforms healthcare with innovative, sustainable, and affordable pharmaceutical solutions worldwide.
Lewens Labs transforms healthcare with innovative, sustainable, and affordable pharmaceutical solutions worldwide.
About the Company : Lewens Labs was established with a vision to transform the pharmaceutical industry, inspired by an award-winning engineering project that reduced the cost of Aceclofenac and receiv...
 Indian Drugs & Chemicals – delivering trusted APIs, intermediates & fine chemicals worldwide with 40+ years of quality expertise.
Indian Drugs & Chemicals – delivering trusted APIs, intermediates & fine chemicals worldwide with 40+ years of quality expertise.
About the Company : Indian Drugs And Chemicals supplies quality products using modern technologies, supported by strong industry expertise. The company serves global markets with a focus on reliabilit...
About the Company : Emnar Pharma was established in 2005, with a drive to become the premier integrated pharmaceuticals company in India. Its business focuses on Contract Manufacturing, Custom Synthes...

About the Company : The Company was incorporated on 19.02.1993 to carry on the business of manufacturing of bulk drugs viz. Omeprazole,Ciprofloxacin, Dichloroflurobenzene, Naproxen, Benzimadizole, etc...

About the Company : Kimia Biosciences, established in 1993, is focused on manufacturing bulk drugs for various therapeutic segments. With a strong emphasis on excellence and advanced technologies, Kim...

About the Company : Medilux Laboratories has been providing quality back-end support to the pharmaceutical industry since our inception in 1988. Our resources are dedicated to promoting better health ...

About the Company : Virchow Group comprises of six active pharmaceutical ingredient (API) and drug intermediate manufacturing units and two biotech units. Incorporated in 1981, the flagship company of...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
28
PharmaCompass offers a list of Acotiamide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Acotiamide manufacturer or Acotiamide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Acotiamide manufacturer or Acotiamide supplier.
A Acotiamide Hydrochloride Hydrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Acotiamide Hydrochloride Hydrate, including repackagers and relabelers. The FDA regulates Acotiamide Hydrochloride Hydrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Acotiamide Hydrochloride Hydrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Acotiamide Hydrochloride Hydrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Acotiamide Hydrochloride Hydrate supplier is an individual or a company that provides Acotiamide Hydrochloride Hydrate active pharmaceutical ingredient (API) or Acotiamide Hydrochloride Hydrate finished formulations upon request. The Acotiamide Hydrochloride Hydrate suppliers may include Acotiamide Hydrochloride Hydrate API manufacturers, exporters, distributors and traders.
click here to find a list of Acotiamide Hydrochloride Hydrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Acotiamide Hydrochloride Hydrate DMF (Drug Master File) is a document detailing the whole manufacturing process of Acotiamide Hydrochloride Hydrate active pharmaceutical ingredient (API) in detail. Different forms of Acotiamide Hydrochloride Hydrate DMFs exist exist since differing nations have different regulations, such as Acotiamide Hydrochloride Hydrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Acotiamide Hydrochloride Hydrate DMF submitted to regulatory agencies in the US is known as a USDMF. Acotiamide Hydrochloride Hydrate USDMF includes data on Acotiamide Hydrochloride Hydrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Acotiamide Hydrochloride Hydrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Acotiamide Hydrochloride Hydrate suppliers with USDMF on PharmaCompass.
A Acotiamide Hydrochloride Hydrate written confirmation (Acotiamide Hydrochloride Hydrate WC) is an official document issued by a regulatory agency to a Acotiamide Hydrochloride Hydrate manufacturer, verifying that the manufacturing facility of a Acotiamide Hydrochloride Hydrate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Acotiamide Hydrochloride Hydrate APIs or Acotiamide Hydrochloride Hydrate finished pharmaceutical products to another nation, regulatory agencies frequently require a Acotiamide Hydrochloride Hydrate WC (written confirmation) as part of the regulatory process.
click here to find a list of Acotiamide Hydrochloride Hydrate suppliers with Written Confirmation (WC) on PharmaCompass.
Acotiamide Hydrochloride Hydrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Acotiamide Hydrochloride Hydrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Acotiamide Hydrochloride Hydrate GMP manufacturer or Acotiamide Hydrochloride Hydrate GMP API supplier for your needs.
A Acotiamide Hydrochloride Hydrate CoA (Certificate of Analysis) is a formal document that attests to Acotiamide Hydrochloride Hydrate's compliance with Acotiamide Hydrochloride Hydrate specifications and serves as a tool for batch-level quality control.
Acotiamide Hydrochloride Hydrate CoA mostly includes findings from lab analyses of a specific batch. For each Acotiamide Hydrochloride Hydrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Acotiamide Hydrochloride Hydrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Acotiamide Hydrochloride Hydrate EP), Acotiamide Hydrochloride Hydrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Acotiamide Hydrochloride Hydrate USP).

