Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
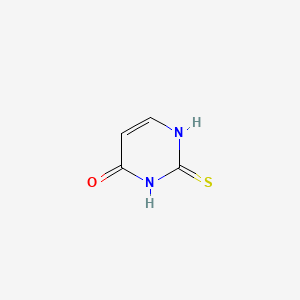

1. Thiouracil
1. Thiouracil
2. 141-90-2
3. 2-mercaptopyrimidin-4-ol
4. 4-hydroxy-2-mercaptopyrimidine
5. Deracil
6. Antagothyroid
7. 2-thioxo-2,3-dihydropyrimidin-4(1h)-one
8. Antagothyroil
9. Nobilen
10. 2-mercapto-4-hydroxypyrimidine
11. 2-mercapto-4-pyrimidone
12. 80275-68-9
13. 2-mercapto-4-pyrimidinol
14. 6-thiouracil
15. 4-hydroxy-2-pyrimidinethiol
16. 4(1h)-pyrimidinone, 2,3-dihydro-2-thioxo-
17. Uracil, 2-thio-
18. 4-pyrimidinol, 2-mercapto-
19. 6-hydroxy-2-mercaptopyrimidine
20. 2-thio-1,3-pyrimidin-4-one
21. 2,3-dihydro-2-thioxo-4(1h)-pyrimidinone
22. 2-sulfanylidene-1h-pyrimidin-4-one
23. 2-tu
24. 2-sulfanylpyrimidin-4-ol
25. Mfcd00006039
26. 2-mercapto-4(1h)-pyrimidinone
27. 2-thio-2,4-(1h,3h)-pyrimidinedione
28. 4-hydroxy-2(1h)-pyrimidinethione
29. 156-82-1
30. Nsc 19473
31. 125220-28-2
32. 126160-72-3
33. 2-sulfanylpyrimidin-4(3h)-one
34. 4(1h)-pyrimidinone, 2-mercapto- (9ci)
35. 4-hydroxypyrimidine-2(1h)-thione
36. 2(1h)-pyrimidinethione, 4-hydroxy- (9ci)
37. 2-mercaptopyrimid-4-one
38. 124700-71-6
39. Tu
40. 2-mercapto-pyrimidin-4-ol
41. 4-hydroxypyrimidine-2-thiol
42. 2-thioxo-2,3-dihydro-1h-pyrimidin-4-one
43. 4(3h)-pyrimidinone, 2-mercapto- (9ci)
44. 59x161scyl
45. 2(1h)-pyrimidinethione, 6-hydroxy- (9ci)
46. Chebi:348530
47. Nsc-19473
48. 124700-72-7
49. 2-sulfanyl-3,4-dihydropyrimidin-4-one
50. Ncgc00095088-01
51. 2,3-dihydro-2-thioxopyrimidin-4(1h)-one
52. Dsstox_cid_1347
53. Dsstox_rid_76100
54. Dsstox_gsid_21347
55. Tiouracyl [polish]
56. 2-thiouracil (van)
57. Tiouracyl
58. 2-thio-4-oxypyrimidine
59. 2-thio-6-oxypyrimidine
60. Cas-141-90-2
61. Ccris 587
62. 2-thio-4-oxo-1,3-pyrimidine
63. Hsdb 2954
64. Nci-c01490
65. Einecs 205-508-8
66. Uracil, 2-thio- (van)
67. Unii-59x161scyl
68. 2-thioxo-1h-pyrimidin-4-one
69. Ai3-25474
70. 2-thio-4-pyrimidone
71. Spectrum_001652
72. Specplus_000746
73. 4-hydroxypyrimidinethione
74. Spectrum3_000979
75. Spectrum4_001104
76. Spectrum5_001378
77. Thiouracil [hsdb]
78. Thiouracil [iarc]
79. Wln: T6mymvj Bus
80. 2-mercaptopyrimidine-4-one
81. 2-thiouracil, >=99%
82. Schembl9172
83. 2-thiouracil [mi]
84. Thiouracil [who-dd]
85. 2-sulfanyl-4-pyrimidinol #
86. 6-hydroxypyrimidine-2-thione
87. Bspbio_002617
88. Kbiogr_001548
89. Kbioss_002132
90. Divk1c_000104
91. Divk1c_006842
92. Spectrum1503973
93. 2-sulfanylidene-1,2,3,4-tetrahydropyrimidin-4-one
94. Chembl345768
95. Dtxsid4021347
96. Hms500f06
97. Kbio1_000104
98. Kbio1_001786
99. Kbio2_002132
100. Kbio2_004700
101. Kbio2_007268
102. Kbio3_001837
103. Ninds_000104
104. Hms1922o08
105. Hms3651f22
106. Hms3885a09
107. Pharmakon1600-01503973
108. Hy-b0503
109. Nsc19473
110. Zinc5127810
111. Tox21_111418
112. Tox21_202418
113. Tox21_302955
114. Ab7033
115. Ccg-39926
116. Mfcd00044203
117. Mfcd09276345
118. Nsc758660
119. S3100
120. Stl011919
121. Stl134904
122. Stl168055
123. Stl185506
124. Akos000119226
125. Akos002303247
126. Akos005366847
127. Akos022061520
128. Tox21_111418_1
129. Am84374
130. Ccg-266107
131. Gs-6727
132. Nsc-758660
133. Sb57767
134. Idi1_000104
135. Smp1_000293
136. Ncgc00095088-02
137. Ncgc00095088-03
138. Ncgc00095088-04
139. Ncgc00095088-05
140. Ncgc00249223-01
141. Ncgc00256423-01
142. Ncgc00259967-01
143. Sy014748
144. 2-thioxo-2,3-dihydro-4(1h)-pyrimidinone
145. Sbi-0051878.p002
146. Db-004877
147. Db-028206
148. Cs-0369245
149. Ft-0613452
150. Ft-0725874
151. Sw220234-1
152. T0224
153. 2-thiouracil, Vetec(tm) Reagent Grade, 99%
154. C19304
155. T-3800
156. T-3808
157. Ab00052397_02
158. Ab00171996_02
159. 2-thiouracil, Vetranal(tm), Analytical Standard
160. Sr-01000872748
161. Sr-01000872748-1
162. Q20892027
163. Z56771015
164. F0001-0490
165. Tul
| Molecular Weight | 128.15 g/mol |
|---|---|
| Molecular Formula | C4H4N2OS |
| XLogP3 | -0.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 128.00443393 g/mol |
| Monoisotopic Mass | 128.00443393 g/mol |
| Topological Polar Surface Area | 73.2 Ų |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 163 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antimetabolites; Antithyroid Agents; Vasodilator Agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
THIOURACIL HAS REPORTEDLY BEEN USED IN HUMAN MEDICINE AS ANTI-THYROID AGENT & IN TREATMENT OF ANGINA PECTORIS & CONGESTIVE HEART FAILURE... HOWEVER, NO EVIDENCE WAS FOUND THAT THIOURACIL PRESENTLY FINDS USE IN US IN THESE APPLICATIONS.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V7 86
TREATMENT OF HYPERTHYROIDISM; ANGINA PECTORIS; CONGESTIVE HEART FAILURE /PRC: FORMER USES IN US/
The Merck Index. 9th ed. Rahway, New Jersey: Merck & Co., Inc., 1976., p. 1210
MEDICATION (VET): THYROID DEPRESSANT; IN HYPERTHYROIDISM & TO PROMOTE FATTENING
The Merck Index. 9th ed. Rahway, New Jersey: Merck & Co., Inc., 1976., p. 1210
MAIN DRAWBACK TO THERAPY WITH ANTITHYROID DRUGS IS HIGH INCIDENCE OF RELAPSE WHEN TREATMENT IS STOPPED. /ANTITHYROID DRUGS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1414
PT SHOULD IMMEDIATELY REPORT DEVELOPMENT OF SORE THROAT OR FEVER, WHICH USUALLY HERALDS ONSET OF.../AGRANULOCYTOSIS/. MILD GRANULOCYTOPENIA, IF NOTED, MAY BE SIGN OF THYROTOXICOSIS OR MAY BE FIRST SIGN OF THIS DANGEROUS DRUG REACTION. CAUTION & FREQUENT LEUKOCYTE COUNTS ARE THEN REQUIRED. /THIOAMIDE DERIV/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1413
...WOMEN TAKING THESE AGENTS SHOULD NOT BREAST-FEED THEIR INFANTS. /ANTITHYROID DRUGS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1413
AT WHAT STAGE IN PREGNANCY...TREATMENT MUST BE INITIATED FOR SUCH EFFECT /FETAL GOITER & HYPOTHYROIDISM/ ON FETUS TO DEVELOP IS NOT KNOWN PRECISELY, BUT IN SOME...INSTANCES MEDICATION WAS NOT BEGUN UNTIL TWO-THIRDS THROUGH PREGNANCY. IT IS FEASIBLE...MIGHT RESULT FROM EVEN LATER MATERNAL ADMIN. /ANTI-THYROID DRUGS/
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 669
For more Drug Warnings (Complete) data for 2-THIOURACIL (6 total), please visit the HSDB record page.
Antithyroid Agents
Agents that are used to treat hyperthyroidism by reducing the excessive production of thyroid hormones. (See all compounds classified as Antithyroid Agents.)
Vasodilator Agents
Drugs used to cause dilation of the blood vessels. (See all compounds classified as Vasodilator Agents.)
DRUG TRANSMISSION TO FETUS: TIME TO APPEAR IN FETUS 10 MIN; TIME TO FETAL/MATERNAL CONCN EQUILIBRIUM 40 MIN; FETAL/MATERNAL CONCN RATIO 1.0 /FROM TABLE/
LaDu, B.N., H.G. Mandel, and E.L. Way. Fundamentals of Drug Metabolism and Disposition. Baltimore: Williams and Wilkins, 1971., p. 102
/IN RATS/ ACCUMULATION OF (35)S LABEL WAS FASTER & OCCURRED TO GREATER EXTENT FROM SMALLER DOSE, SUGGESTING PRESENCE OF SATURABLE TRANSPORT MECHANISM. SIMILAR STUDIES WITH (14)C-THIOURACIL HAVE SHOWN ACCUM OF RADIOACTIVITY IN BOTH MATERNAL & FETAL THYROID TISSUES OF RABBITS...
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 162
...WITH BOTH LARGE (39 UMOL) & SMALL (1.2 UMOL) DOSES OF (35)S-THIOURACIL, CONSIDERABLE ACCUM OF BOTH TOTAL RADIOACTIVITY & UNMETABOLIZED (35)S-THIOURACIL OCCURS IN THYROID TISSUE RELATIVE TO PLASMA /IN RATS/.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 162
THIOURACIL IS ABSORBED FROM GI TRACT IN RATS... IN RATS ADMIN 5 MG BY IV INJECTION, 30% OF THIOURACIL...RECOVERED FROM CARCASSES AFTER 3 HR & ONLY TRACES AFTER 24 HR.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V7 90
For more Absorption, Distribution and Excretion (Complete) data for 2-THIOURACIL (7 total), please visit the HSDB record page.
IN HOMOGENIZED RAT LIVER PREPN FROM FEMALE HOLTZMAN RATS, 28-35% OF THIOURACIL WAS METABOLIZED WITHIN 3 HR. PATHWAY FOR BREAKDOWN OF THIOURACIL WAS SUGGESTED TO BE AS FOLLOWS: URACIL; BETA-UREIDOPROPIONIC ACID, WHICH WAS FURTHER METABOLIZED TO BETA-ALANINE; AMMONIA & CARBON DIOXIDE...
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V7 90
ANTITHYROID DRUGS INHIBIT FORMATION OF THYROID HORMONE LARGELY BY INTERFERING WITH INCORPORATION OF IODINE INTO ORGANIC FORM. THIS IMPLIES THAT THEY INTERFERE WITH OXIDN OF IODIDE ION, BUT ELUCIDATION OF DETAILED MECHANISM... HAMPERED BY INCOMPLETE UNDERSTANDING OF IODIDE-OXIDIZING SYSTEM OF...GLAND. /ANTITHYROID DRUGS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1411
THIOAMIDE DERIV DO NOT HAVE PERMANENT EFFECT UPON THYROID GLAND BUT INHIBIT HORMONE SYNTHESIS & SECRETION UNTIL SPONTANEOUS REMISSION OCCURS DURING COURSE OF DISEASE. /THIOAMIDE DERIV/
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 607
ABOUT THIS PAGE
58
PharmaCompass offers a list of 2-Thiouracil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 2-Thiouracil manufacturer or 2-Thiouracil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred 2-Thiouracil manufacturer or 2-Thiouracil supplier.
A 2-Thiouracil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 2-Thiouracil, including repackagers and relabelers. The FDA regulates 2-Thiouracil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 2-Thiouracil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A 2-Thiouracil supplier is an individual or a company that provides 2-Thiouracil active pharmaceutical ingredient (API) or 2-Thiouracil finished formulations upon request. The 2-Thiouracil suppliers may include 2-Thiouracil API manufacturers, exporters, distributors and traders.
click here to find a list of 2-Thiouracil suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 2-Thiouracil DMF (Drug Master File) is a document detailing the whole manufacturing process of 2-Thiouracil active pharmaceutical ingredient (API) in detail. Different forms of 2-Thiouracil DMFs exist exist since differing nations have different regulations, such as 2-Thiouracil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 2-Thiouracil DMF submitted to regulatory agencies in the US is known as a USDMF. 2-Thiouracil USDMF includes data on 2-Thiouracil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 2-Thiouracil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 2-Thiouracil suppliers with USDMF on PharmaCompass.
2-Thiouracil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 2-Thiouracil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 2-Thiouracil GMP manufacturer or 2-Thiouracil GMP API supplier for your needs.
A 2-Thiouracil CoA (Certificate of Analysis) is a formal document that attests to 2-Thiouracil's compliance with 2-Thiouracil specifications and serves as a tool for batch-level quality control.
2-Thiouracil CoA mostly includes findings from lab analyses of a specific batch. For each 2-Thiouracil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
2-Thiouracil may be tested according to a variety of international standards, such as European Pharmacopoeia (2-Thiouracil EP), 2-Thiouracil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (2-Thiouracil USP).

