
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
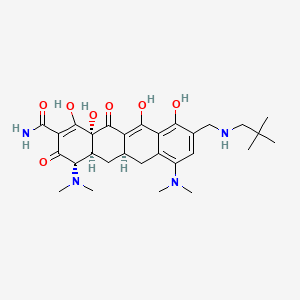

1. Nuzyra
1. Amadacycline
2. 389139-89-3
3. Nuzyra
4. Ptk 0796
5. Omadacycline [usan]
6. Ptk-0796
7. Bay 73-6944
8. 090ip5rv8f
9. 389139-89-3 (free Base)
10. (4s,4as,5ar,12ar)-4,7-bis(dimethylamino)-9-[(2,2-dimethylpropylamino)methyl]-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide
11. Omadacycline (usan)
12. (4s,4as,5ar,12as)-4,7-bis(dimethylamino)-9-(((2,2-dimethylpropyl)amino)methyl)- 3,10,12,12a- Tetrahydroxy-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2- Carboxamide
13. Amadacycline Methanesulfonate
14. Omadacycline [usan:inn]
15. Unii-090ip5rv8f
16. (4s,4as,5ar,12as)-4,7-bis(dimethylamino)-9-[(2,2-dimethylpropylamino)methyl]-3,10,12,12a-tetrahydroxy-1,11-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide
17. 2-naphthacenecarboxamide, 4,7-bis(dimethylamino)-9-(((2,2-dimethylpropyl)amino)methyl)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-, (4s,4as,5ar,12as)-
18. 2-naphthacenecarboxamide, 4,7-bis(dimethylamino)-9-[[(2,2-dimethylpropyl)amino]methyl]-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-, (4s,4as,5ar,12as)-
19. Mk-2764
20. Omadacycline [mi]
21. Omadacycline [inn]
22. Omadacycline [who-dd]
23. Schembl1525961
24. 9-neopentylaminomethylminocycline
25. Chembl1689772
26. Schembl17150976
27. Schembl20952297
28. Gtpl10839
29. Ptk-796
30. Ptk0796
31. Chebi:177758
32. Dtxsid201027687
33. Bcp12946
34. Ex-a4252
35. Zinc4836283
36. Compound 6 [pmid: 21302930]
37. Cs-1338
38. Db12455
39. Bay-73-6944
40. Bay-73-7388
41. Ncgc00378946-03
42. Ac-33245
43. Hy-14865
44. Ptk 0796, Bay 73-6944
45. D09647
46. E80520
47. Q15426992
48. (4s,4as,5ar,12as)-4,7-bis(dimethylamino)-9-(((2,2-dimethylpropyl)amino)methyl)-3,10,12,12a- Tetrahydroxy-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide
49. 2-naphthacenecarboxamide, 4,7-bis(dimethylamino)-9-(((2,2- Dimethylpropyl)amino)methyl)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a- Tetrahydroxy-1,11-dioxo-, (4s,4as,5ar,12as)-
| Molecular Weight | 556.6 g/mol |
|---|---|
| Molecular Formula | C29H40N4O7 |
| XLogP3 | 3 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 7 |
| Exact Mass | 556.28969963 g/mol |
| Monoisotopic Mass | 556.28969963 g/mol |
| Topological Polar Surface Area | 177 Ų |
| Heavy Atom Count | 40 |
| Formal Charge | 0 |
| Complexity | 1140 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Omadacycline is indicated for the treatment of community acquired bacterial pneumonia and acute bacterial skin and skin structure infections caused by omadacycline-susceptible organisms in adults.
FDA Label
Treatment of bacterial pneumonia
Treatment of acute bacterial skin and skin structure infections
Omadacycline can be either bacteriostatic or bacteriocidal depending on the organism involved. It disrupts bacterial protein synthesis without affecting DNA, RNA, or peptidoglycan synthesis. Omadacycline represents an improvement over existing tetracycline agents as it has not been found to be subject to tetracycline resistance mediated by tetracycline efflux pumps encoded by the tet(K), tet(L), and tet(B) or to ribosomal protection proteins encoded by tet(O) and tet(M). Omadacycline is susceptible to RNA mutations which confer resistance to tetracyclines.
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01A - Tetracyclines
J01AA - Tetracyclines
J01AA15 - Omadacycline
Absorption
Omadacycline has an mean absolute oral bioavailability of 34.5% and a mean Tmax of2.5 h with oral dosing. With multiple dosing, Omadacycline displays an accumulation factor of 1.5. Official labeling states that food does not significantly impact rate or extent of absorption, however, conflicting data exists suggesting food may lower the bioavailability of omadacycline taken after eating. The exposure in alveolar cells and epithelial lining fluid is 25.8 and 1.5 fold higher than plasma exposure after IV administration, suggesting Omadacycline penetrates the lungs to a significant degree.
Route of Elimination
After IV dosing 27% of Omadacycline was eliminated by the kidneys. In oral dosing 14.4% was found to be eliminated by the kidneys and 81.1% in the feces. Neither renal nor hepatic impairment appears to produce a clinically relevant effect elimination.
Volume of Distribution
Omadacycline has a mean Vd of 256 L after a single dose and a Vss of 190 L.
Clearance
Omadacycline has a mean systemic clearance of 11.24 L/h and a renal clearance of 2.4-3.3 L/h.
Omadacycline is not known to be metabolized in humans.
Omadacycline has a mean half life of elimination of 16.2 h.
Omadacycline binds to the primary tetracycline binding site on the bacterial 30s ribosomal subunit with high specificity. There it acts to block protein synthesis, disrupting many facets of cellular function and resulting in either cell death or stasis.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
99
PharmaCompass offers a list of Omadacycline API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Omadacycline manufacturer or Omadacycline supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Omadacycline manufacturer or Omadacycline supplier.
A 090IP5RV8F manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 090IP5RV8F, including repackagers and relabelers. The FDA regulates 090IP5RV8F manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 090IP5RV8F API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 090IP5RV8F manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 090IP5RV8F supplier is an individual or a company that provides 090IP5RV8F active pharmaceutical ingredient (API) or 090IP5RV8F finished formulations upon request. The 090IP5RV8F suppliers may include 090IP5RV8F API manufacturers, exporters, distributors and traders.
click here to find a list of 090IP5RV8F suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 090IP5RV8F DMF (Drug Master File) is a document detailing the whole manufacturing process of 090IP5RV8F active pharmaceutical ingredient (API) in detail. Different forms of 090IP5RV8F DMFs exist exist since differing nations have different regulations, such as 090IP5RV8F USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 090IP5RV8F DMF submitted to regulatory agencies in the US is known as a USDMF. 090IP5RV8F USDMF includes data on 090IP5RV8F's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 090IP5RV8F USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 090IP5RV8F suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 090IP5RV8F as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 090IP5RV8F API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 090IP5RV8F as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 090IP5RV8F and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 090IP5RV8F NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 090IP5RV8F suppliers with NDC on PharmaCompass.
090IP5RV8F Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 090IP5RV8F GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 090IP5RV8F GMP manufacturer or 090IP5RV8F GMP API supplier for your needs.
A 090IP5RV8F CoA (Certificate of Analysis) is a formal document that attests to 090IP5RV8F's compliance with 090IP5RV8F specifications and serves as a tool for batch-level quality control.
090IP5RV8F CoA mostly includes findings from lab analyses of a specific batch. For each 090IP5RV8F CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
090IP5RV8F may be tested according to a variety of international standards, such as European Pharmacopoeia (090IP5RV8F EP), 090IP5RV8F JP (Japanese Pharmacopeia) and the US Pharmacopoeia (090IP5RV8F USP).

