
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
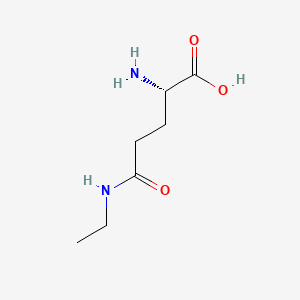

1. Delta-glutamylethylamide
2. Gamma-glutamylethylamide
3. L-glutamic Acid-gamma-ethylamide
4. Theanine
5. Theanine, (d)-isomer
6. Theanine, (dl)-isomer
7. Theanine, (l)-isomer
1. 3081-61-6
2. Theanine
3. Theanin
4. N-ethyl-l-glutamine
5. Suntheanine
6. (s)-2-amino-5-(ethylamino)-5-oxopentanoic Acid
7. N5-ethyl-l-glutamine
8. L-gamma-glutamylethylamide
9. N(5)-ethyl-l-glutamine
10. (2s)-2-amino-5-(ethylamino)-5-oxopentanoic Acid
11. N-gamma-ethyl-l-glutamine
12. Chebi:17394
13. 8021pr16qo
14. Nsc-21308
15. N-ethylglutamine
16. Ccris 7326
17. Einecs 221-379-0
18. Nsc 21308
19. Unii-8021pr16qo
20. L-theamine
21. Mfcd00059653
22. Hsdb 8166
23. (+)-theanine
24. N-ethyl L-glutamine
25. N?-ethyl-l-glutamine
26. H-glu(nhet)-oh
27. Theanine [inci]
28. Theanine [mi]
29. Theanine [vandf]
30. Theanine, L-
31. Spectrum2_001693
32. Spectrum3_001137
33. Spectrum4_001984
34. Spectrum5_000897
35. Glutamine, N-gamma-ethyl-
36. Theanine [who-dd]
37. Glutamine, N-ethyl-, L-
38. Bspbio_002633
39. Kbiogr_002514
40. L-theanine [usp-rs]
41. Schembl190716
42. Spectrum1505254
43. Spbio_001646
44. L-glutamic Acid G-(ethylamide)
45. Chembl3039113
46. Kbio3_002133
47. L-glutamic Acid ?-(ethylamide)
48. Dtxsid80184817
49. L-glutamic Acid -r-monoethylamide
50. L-theanine, >=98% (hplc)
51. Bcp28252
52. Zinc1532828
53. Ccg-38778
54. S3852
55. Akos016842508
56. Am82649
57. Db12444
58. L-theanine (ngamma-ethyl-l-glutamine)
59. Sdccgmls-0066811.p001
60. Ncgc00178565-01
61. Ac-23939
62. As-12265
63. Hy-15121
64. Cs-0003777
65. N2085
66. T0954
67. C01047
68. 081t616
69. A820634
70. Q909931
71. Sr-05000002409
72. Q-201330
73. Sr-05000002409-1
74. (2s)-2-azanyl-5-(ethylamino)-5-oxidanylidene-pentanoic Acid
75. C6b5a580-d410-4464-adb8-543860137d4c
76. L-theanine, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 174.20 g/mol |
|---|---|
| Molecular Formula | C7H14N2O3 |
| XLogP3 | -3.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Exact Mass | 174.10044231 g/mol |
| Monoisotopic Mass | 174.10044231 g/mol |
| Topological Polar Surface Area | 92.4 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 170 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
EXPL THER Recent neuropharmacological research has suggested that certain constituents of tea may have modulatory effects on brain state. The bulk of this research has focused on either L-theanine or caffeine ingested alone (mostly the latter) and has been limited to behavioral testing, subjective rating, or neurophysiological assessments during resting. Here, we investigated the effects of both L-theanine and caffeine, ingested separately or together, on behavioral and electrophysiological indices of tonic (background) and phasic (event-related) visuospatial attentional deployment. Subjects underwent 4 d of testing, ingesting either placebo, 100 mg of L-theanine, 50 mg of caffeine, or these treatments combined. The task involved cued shifts of attention to the left or right visual hemifield in anticipation of an imperative stimulus requiring discrimination. In addition to behavioral measures, we examined overall, tonic attentional focus as well as phasic, cue-dependent anticipatory attentional biasing, as indexed by scalp-recorded alpha-band (8-14 Hz) activity. We found an increase in hit rate and target discriminability (d') for the combined treatment relative to placebo, and an increase in d' but not hit rate for caffeine alone, whereas no effects were detected for L-theanine alone. Electrophysiological results did not show increased differential biasing in phasic alpha across hemifields but showed lower overall tonic alpha power in the combined treatment, similar to previous findings at a larger dosage of L-theanine alone. This may signify a more generalized tonic deployment of attentional resources to the visual modality and may underlie the facilitated behavioral performance on the combined ingestion of these 2 major constituents of tea.
PMID:18641209 Kelly SP et al; J Nutr 138 (8): 1572S-1577S (2008)
EXPL THER The non-proteinic amino acid L-theanine and caffeine, a methylxanthine derivative, are naturally occurring ingredients in tea. The present study investigated the effect of a combination of 97 mg L-theanine and 40 mg caffeine as compared to placebo treatment on cognitive performance, alertness, blood pressure, and heart rate in a sample of young adults (n = 44). Cognitive performance, self-reported mood, blood pressure, and heart rate were measured before L-theanine and caffeine administration (i.e. at baseline) and 20 min and 70 min thereafter. The combination of moderate levels of L-theanine and caffeine significantly improved accuracy during task switching and self-reported alertness (both P < 0.01) and reduced self-reported tiredness (P < 0.05). There were no significant effects on other cognitive tasks, such as visual search, choice reaction times, or mental rotation. The present results suggest that 97 mg of L-theanine in combination with 40 mg of caffeine helps to focus attention during a demanding cognitive task.
PMID:21040626 Giesbrecht T et al; Nutr Neurosci 13 (6): 283-90 (2010)
EXPL THER The aim of this study was to compare 50 mg caffeine, with and without 100 mg L-theanine, on cognition and mood in healthy volunteers. The effects of these treatments on word recognition, rapid visual information processing, critical flicker fusion threshold, attention switching and mood were compared to placebo in 27 participants. Performance was measured at baseline and again 60 min and 90 min after each treatment (separated by a 7-day washout). Caffeine improved subjective alertness at 60 min and accuracy on the attention-switching task at 90 min. The L-theanine and caffeine combination improved both speed and accuracy of performance of the attention-switching task at 60 min, and reduced susceptibility to distracting information in the memory task at both 60 min and 90 min. These results replicate previous evidence which suggests that L-theanine and caffeine in combination are beneficial for improving performance on cognitively demanding tasks.
PMID:18681988 Owen GN et al; Nutr Neurosci 11 (4): 193-8 (2008)
EXPL THER L-Theanine, an ethylamide derivate of glutamate found in abundance in green tea, has been shown to exert beneficial actions in animal models for several neurological disorders. /Authors/ here investigated for the first time the effect of L-theanine intake on seizure susceptibility using acute pilocarpine and pentylenetetrazol (PTZ) mouse models for studying, respectively, limbic seizures or primarily generalized seizures. Moreover, /authors/ studied the effect of l-theanine intake on extracellular hippocampal and cortical glutamate and gamma-aminobutyric acid (GABA) levels, using in vivo microdialysis. Feeding mice with a 4% L-theanine solution significantly decreased their susceptibility to pilocarpine-induced seizures whereas susceptibility to PTZ-induced seizures was increased. The latter effect was linked to decreased extracellular GABA concentrations in frontal cortex.
PMID:23324588 Schallier A et al; Nutr Neurosci 16 (2): 78-82 (2013)
For more Therapeutic Uses (Complete) data for Theanine (16 total), please visit the HSDB record page.
Pregnant women and nursing mothers should avoid L-theanine supplements. Use of L-theanine supplements cocomiitantly with cancer chemotherapeutic agents must be done under medical supervision.
PDR for Nutritional Supplements 2nd ed. Thomson Reuters, Montvale, NJ 2008, p. 367
L-theanine is contraindicated in those who are hypersensitive to any component of an L-theanine-containing product.
PDR for Nutritional Supplements 2nd ed. Thomson Reuters, Montvale, NJ 2008, p. 367
From animal studies, it appears that L-theanine is absorbed from the small intestine via a sodium-coupled active transport process and appears to cross the blood-brain barrier. It has been found in the rat studies that the D-enantiomer of theanine may decrease the absorption of L-theanine.
PDR for Nutritional Supplements 2nd ed. Thomson Reuters, Montvale, NJ 2008, p. 366
In medium containing theanine with glutaminase in vitro, glutamate gradually generated, showing that glutaminase reacted with theanine. Furthermore, the generation of glutamate increased by reaction of theanine and gamma-glutamyltranspeptidase (gamma-GTP), showed that gamma-GTP converted theanine to glutamate. It is expected that theanine metabolism occurred by hydrolysis and rearrangement reaction by gamma-GTP in the liver. Namely, it is suggested that the metabolism of theanine mediated by glutaminase and gamma-GTP and the increase of glutamate mediated GSH is important for theanine-induced action.
PMID:16140449 Sadzuka Y et al; Food Chem Toxicol 44 (2): 286-92 (2006)
L-theanine (N-ethyl-L-glutamine) or theanine is a major amino acid uniquely found in green tea. L-theanine has been historically reported as a relaxing agent, prompting scientific research on its pharmacology. Animal neurochemistry studies suggest that L-theanine increases brain serotonin, dopamine, GABA levels and has micromolar affinities for AMPA, Kainate and NMDA receptors. In addition has been shown to exert neuroprotective effects in animal models possibly through its antagonistic effects on group 1 metabotrophic glutamate receptors. Behavioural studies in animals suggest improvement in learning and memory. Overall, L-theanine displays a neuropharmacology suggestive of a possible neuroprotective and cognitive enhancing agent and warrants further investigation in animals and humans.
PMID:17182482 Nathan PJ et al; J Herb Pharmacother 6 (2): 21-30 (2006)
In an investigation of the mechanisms of the neuroprotective effects of theanine (gamma-glutamylethylamide) in brain ischemia, inhibition by theanine of the binding of [(3)H](RS)-alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA), [(3)H]kainate, and [(3)H](E)-3-(2-phenyl-2-carboxyethenyl)-4,6-dichloro-1-H-indole-2-carboxylic acid (MDL 105,519) to glutamate receptors was studied in terms of its possible inhibiting effects on the three receptor subtypes (AMPA, kainate, and NMDA glycine), with rat cortical neurons. Theanine bound the three receptors, but its IC50 of theanine was 80- to 30,000-fold less than that of L-glutamic acid.
PMID:12596867 Kakuda T et al; Biosci Biotechnol Biochem. 66 (12): 2683-6 (2002)
In this study, the inhibitory effect of L-theanine, an amino acid derivative of tea, on the rewarding effects of nicotine and its underlying mechanisms of action were studied. We found that L-theanine inhibited the rewarding effects of nicotine in a conditioned place preference (CPP) model of the mouse and reduced the excitatory status induced by nicotine in SH-SY5Y cells to the same extent as the nicotine receptor inhibitor dihydro-beta-erythroidine (DHbetaE). Further studies using high performance liquid chromatography, western blotting and immunofluorescence staining analyses showed that L-theanine significantly inhibited nicotine-induced tyrosine hydroxylase (TH) expression and dopamine production in the midbrain of mice. L-theanine treatment also reduced the upregulation of the a(4), beta(2) and a(7) nicotine acetylcholine receptor (nAChR) subunits induced by nicotine in mouse brain regions that related to the dopamine reward pathway, thus decreasing the number of cells that could react to nicotine. In addition, L-theanine treatment inhibited nicotine-induced c-Fos expression in the reward circuit related areas of the mouse brain. Knockdown of c-Fos by siRNA inhibited the excitatory status of cells but not the upregulation of TH induced by nicotine in SH-SY5Y cells. Overall, the present study showed that L-theanine reduced the nicotine-induced reward effects via inhibition of the nAChR-dopamine reward pathway. These results may offer new therapeutic strategies for treatment of tobacco addiction.
PMID:23233221 Di X et al; Sci China Life Sci 55 (12): 1064-74 (2012)
L-theanine, previously shown to penetrate the blood-brain barrier through the leucine-preferring transport system, has been demonstrated to produce significant increases in serotonin and/or dopamine concentrations in the brain principally in the striatum, hypothalamus and hippocampus.
PDR for Nutritional Supplements 2nd ed. Thomson Reuters, Montvale, NJ 2008, p. 366
For more Mechanism of Action (Complete) data for Theanine (9 total), please visit the HSDB record page.
ABOUT THIS PAGE
17
PharmaCompass offers a list of L-Theanine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right L-Theanine manufacturer or L-Theanine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred L-Theanine manufacturer or L-Theanine supplier.
PharmaCompass also assists you with knowing the L-Theanine API Price utilized in the formulation of products. L-Theanine API Price is not always fixed or binding as the L-Theanine Price is obtained through a variety of data sources. The L-Theanine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 081T616 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 081T616, including repackagers and relabelers. The FDA regulates 081T616 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 081T616 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 081T616 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A 081T616 supplier is an individual or a company that provides 081T616 active pharmaceutical ingredient (API) or 081T616 finished formulations upon request. The 081T616 suppliers may include 081T616 API manufacturers, exporters, distributors and traders.
click here to find a list of 081T616 suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
081T616 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 081T616 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 081T616 GMP manufacturer or 081T616 GMP API supplier for your needs.
A 081T616 CoA (Certificate of Analysis) is a formal document that attests to 081T616's compliance with 081T616 specifications and serves as a tool for batch-level quality control.
081T616 CoA mostly includes findings from lab analyses of a specific batch. For each 081T616 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
081T616 may be tested according to a variety of international standards, such as European Pharmacopoeia (081T616 EP), 081T616 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (081T616 USP).

