
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
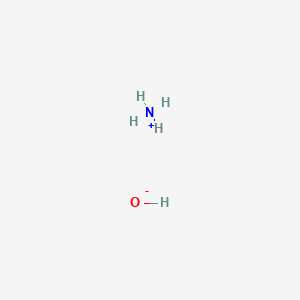

1. Hydroxide, Ammonium
1. 1336-21-6
2. Ammonia Aqueous
3. Ammonium Hydroxide ((nh4)(oh))
4. Ammonium Hydroxide Solution
5. Ammoniumhydroxid
6. Nh4oh
7. Aquammonia
8. Ammonia, Aqua
9. Household Ammonia
10. Ammonia, Aqueous Solution
11. Ammonia, Monohydrate
12. Ammonia Water 29%
13. Caswell No. 044
14. Sx 1 (ammonia Water)
15. Ammoniumhydroxide
16. Mfcd00066650
17. Ammonia, Aqueous
18. Sx 1
19. Ammonia Water [jan]
20. Hsdb 5125
21. Einecs 215-647-6
22. Un2672
23. Un3318
24. Epa Pesticide Chemical Code 005301
25. Ammoniaque
26. Ammonia Hydrate
27. Water Ammonia
28. Ammonium Hydoxide
29. Ammonium Hydroxid
30. Ammonium Hyroxide
31. Amonium Hydroxide
32. Agua De Amoniaco
33. Ammonium Hydorxide
34. Amrnonium Hydroxide
35. Hidroxido De Amonio
36. Nh3 Water
37. Water Nh3
38. Hydroxyde D'ammonium
39. Un2073
40. Ammonia Water (tn)
41. Ammonium Aqueous (28% Or Less Nh3)
42. Amine Hydrate
43. Ammonium Hydroxide (28% Or Less Ammonia)
44. Ammonia Water (jp17)
45. Ec 215-647-6
46. Nh3 H2o
47. Nh3-h2o
48. Nh3.h2o
49. Ammonium Hydroxide Reagent Acs
50. Ammonium Hydroxide, 25% Nh3
51. Ammonia (ammonium Hydroxide) 28% By Weight Or More Nh3
52. Dtxsid4020080
53. Chebi:18219
54. Ammonium Hydroxide, 28% Solution
55. Akos015903971
56. Akos030228272
57. E527
58. Ft-0622304
59. Ammonium Hydroxide, 10% V/v Aqueous Solution
60. Ammonium Hydroxide, 50% V/v Aqueous Solution
61. Ammonium Hydroxide, 0.1n Standardized Solution
62. Ammonium Hydroxide, 1.0n Standardized Solution
63. Ammonium Hydroxide, 5.0n Standardized Solution
64. C01358
65. D04594
66. Ammonium Hydroxide, Solution 28-30% Acs Reagent
67. J-006420
68. Ammonium Hydroxide, Environmental Grade, 20-22% Nh3
69. Ammonium Hydroxide, Solution 14-16% W/w Trace Metals Grade
70. Ammonia Solution, Relative Density <0.880 At 15 Degree C In Water, With >50% Ammonia
71. Ammonium Hydroxide, Acculute Standard Volumetric Solution, Final Concentration 0.1n
72. Ammonia Solution, Relative Density <0.880 At 15 Degree C In Water, With >50% Ammonia [un3318] [nonflammable Gas, Poison Gas]
73. Ammonia Solutions, Relative Density <0.880 At 15 C In Water, With >35% But Not >50% Ammonia
74. Ammonia Solutions, Relative Density <0.880 At 15 C In Water, With >35% But Not >50% Ammonia [un2073] [nonflammable Gas]
75. Ammonia Solutions, Relative Density Between 0.880 And 0.957 At 15 C In Water, With >10% But Not >35% Ammonia
76. Ammonia Solutions, Relative Density Between 0.880 And 0.957 At 15 C In Water, With >10% But Not >35% Ammonia [un2672] [corrosive]
| Molecular Weight | 35.046 g/mol |
|---|---|
| Molecular Formula | H5NO |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Exact Mass | 35.037113783 g/mol |
| Monoisotopic Mass | 35.037113783 g/mol |
| Topological Polar Surface Area | 2 Ų |
| Heavy Atom Count | 2 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
10% ammonia water as a reflex respiratory stimulant; MEDICATION (VET): externally on bites & stings; As rubefacient on bruises, sprains; Inhalant; Internally as an antacid & carminative /Ammonia water-10%/
The Merck Index. 10th ed. Rahway, New Jersey: Merck Co., Inc., 1983., p. 74
... EXCRETION IS PRIMARILY BY WAY OF KIDNEYS, BUT A NOT INSIGNIFICANT AMT IS PASSED THROUGH SWEAT GLANDS.
Clayton, G.D., F.E. Clayton (eds.) Patty's Industrial Hygiene and Toxicology. Volumes 2A, 2B, 2C, 2D, 2E, 2F: Toxicology. 4th ed. New York, NY: John Wiley & Sons Inc., 1993-1994., p. 760
... Ammonium toxicity induced by ammonium acetate administration in frogs led to variable excretion of ammonia and urea into the medium. When 11.45 mM/kg body wt of ammonium acetate was administered, ammonia excretion increased while urea excretion decreased. When 4.17 mM/kg body wt of ammonium acetate was administered, the urea excretion increased while NH3 excretion decreased.
Neeraja P, Swami KS; Indian J Physiol Pharmac 27 (2): 123-8 (1983)
Ammonium hydroxide penetrates fastest, followed by sodium hydroxide, potassium hydroxide, and finally calcium hydroxide.
Sullivan, J.B. Jr., G.R. Krieger (eds.). Hazardous Materials Toxicology-Clinical Principles of Environmental Health. Baltimore, MD: Williams and Wilkins, 1992., p. 433
Male rats gavaged with 1000 umol (15)N-ammonium chloride each day for 5 days excreted low, but significant amounts of excess (15)N-NO3- in urine on the 5 days of treatment & on the 5 subsequent days. An in vitro chemical model system was used to demonstrate that oxidation of ammonia to NO3- by the hydroxyl radical at physiological pH is chemically feasible. These results are consistent with the hypothesis that ammonia is oxidized to NO3- in vivo by a nonenzymic process which involves active O species such as the hydroxyl radical.
PMID:6690089 Saul RL, Archer MC; Carcinogenesis 5 (1): 77-81 (1984)
Market Place

ABOUT THIS PAGE
19
PharmaCompass offers a list of Ammonium Hydroxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Ammonium Hydroxide manufacturer or Ammonium Hydroxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ammonium Hydroxide manufacturer or Ammonium Hydroxide supplier.
PharmaCompass also assists you with knowing the Ammonium Hydroxide API Price utilized in the formulation of products. Ammonium Hydroxide API Price is not always fixed or binding as the Ammonium Hydroxide Price is obtained through a variety of data sources. The Ammonium Hydroxide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 05003_RIEDEL manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 05003_RIEDEL, including repackagers and relabelers. The FDA regulates 05003_RIEDEL manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 05003_RIEDEL API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A 05003_RIEDEL supplier is an individual or a company that provides 05003_RIEDEL active pharmaceutical ingredient (API) or 05003_RIEDEL finished formulations upon request. The 05003_RIEDEL suppliers may include 05003_RIEDEL API manufacturers, exporters, distributors and traders.
05003_RIEDEL Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 05003_RIEDEL GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 05003_RIEDEL GMP manufacturer or 05003_RIEDEL GMP API supplier for your needs.
A 05003_RIEDEL CoA (Certificate of Analysis) is a formal document that attests to 05003_RIEDEL's compliance with 05003_RIEDEL specifications and serves as a tool for batch-level quality control.
05003_RIEDEL CoA mostly includes findings from lab analyses of a specific batch. For each 05003_RIEDEL CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
05003_RIEDEL may be tested according to a variety of international standards, such as European Pharmacopoeia (05003_RIEDEL EP), 05003_RIEDEL JP (Japanese Pharmacopeia) and the US Pharmacopoeia (05003_RIEDEL USP).

