



API Suppliers

US DMFs Filed

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
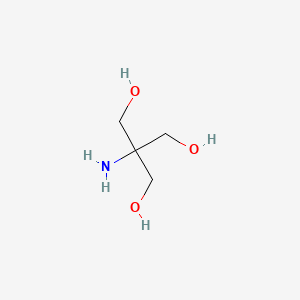

1. Tri(hydroxymethyl)aminomethane
2. Tris Buffer
3. Tris(hydroxymethyl)aminomethane
4. Tris-magnesium(ii)-potassium Chloride Buffer
5. Tris-mg(ii)-kcl Buffer
6. Trisamine
7. Trizma
8. Trometamol
1. Trometamol
2. 77-86-1
3. Tris(hydroxymethyl)aminomethane
4. Tris
5. 2-amino-2-(hydroxymethyl)propane-1,3-diol
6. Tham
7. Trizma
8. Trisamine
9. 2-amino-2-(hydroxymethyl)-1,3-propanediol
10. Tris Base
11. Tris Buffer
12. Tromethane
13. Tris-base
14. Trisaminol
15. 1,3-propanediol, 2-amino-2-(hydroxymethyl)-
16. Pehanorm
17. Talatrol
18. Trisamin
19. Trispuffer
20. Tutofusin Tris
21. Apiroserum Tham
22. Tris-steril
23. Addex-tham
24. Tris, Free Base
25. Trimethylolaminomethane
26. Tris Amino
27. Aminotrimethylolmethane
28. Aminotris(hydroxymethyl)methane
29. Tham-e
30. Tris (buffering Agent)
31. Tromethanmin
32. Tris(hydroxymethyl)methanamine
33. Tris(hydroxymethyl)methylamine
34. Trometamole
35. Trizma Base
36. 2-amino-2-methylol-1,3-propanediol
37. Tris-hydroxymethylaminomethane
38. Tri(hydroxymethyl)aminomethane
39. Tris-amino
40. Methylamine, 1,1,1-tris(hydroxymethyl)-
41. Nsc 6365
42. 2-(hydroxymethyl)-2-amino-1,3-propanediol
43. Trometamol [inn]
44. Methanamine, 1,1,1-tris(hydroxymethyl)-
45. Nsc-6365
46. Mfcd00004679
47. Bakerbond(tm) Cyano (cn)
48. 2-amino-2-hydroxymethyl-1,3-propanediol
49. 023c2whx2v
50. Chebi:9754
51. Tris(hydroxymethyl) Aminomethane
52. Abx (antibody Exchanger)
53. Tris-(hydroxymethyl)-aminomethane
54. Nsc6365
55. 1,1,1-tris(hydroxymethyl)methanamine
56. Tris (tromethamine)
57. 126850-03-1
58. 126850-06-4
59. 126850-08-6
60. Ncgc00159412-02
61. Ncgc00159412-04
62. Tris Buffertris-hydroxymethyl-aminomethan
63. Wp Quat (strong Anion Exchanger)
64. Tromethamolum
65. Dsstox_cid_3723
66. Trishydroxymethylaminomethane
67. Wln: Q1xz1q1q
68. Dsstox_rid_77165
69. Dsstox_gsid_23723
70. 126850-05-3
71. 136760-04-8
72. Amino (nh2) Narrow-pore Media-normal Phase
73. 4-anilino-1-(2-hydroxyethylamino)anthracene-9,11-dione
74. Tromethamine [usan]
75. (tris(hydroxymethyl)aminomethane)
76. [tris(hydroxymethyl)aminomethane]
77. Caswell No. 036
78. Trometamolum
79. 1, 2-amino-2-(hydroxymethyl)-
80. Triladyl
81. Trigmo Base
82. Mfcd00132476
83. Methanamine,1,1-tris(hydroxymethyl)-
84. Methylamine,1,1-tris(hydroxymethyl)-
85. Trometamolum [inn-latin]
86. Tris(hydroxymethyl)amino Methane
87. .beta.-d-ribo-hexopyranose, 1,6-anhydro-3-deoxy-2-o-(1-methylethyl)-4-o-(phenylmethyl)-
88. Cas-77-86-1
89. Tris(hydroxymethyl)aminomethane, >=99.8%
90. 2-amino-2-(hydroxymethyl)-1, 3-propanediol
91. Hsdb 3408
92. Einecs 201-064-4
93. Tris(hydroxymethyl)aminomethane, Electrophoresis Grade
94. Tris-mg(ii)-kcl Buffer
95. Tromethamine [usan:usp]
96. Epa Pesticide Chemical Code 083901
97. Tris-hydroxymethyl-aminomethan [german]
98. Unii-023c2whx2v
99. Trometamina
100. Tromethamin
101. Aminotri(hydroxymethyl)methane
102. Tribase
103. Tris-hydroxymethyl-aminomethan
104. Tris-buffer
105. Tris-amine
106. Ai3-03948
107. Tro.meta.mole
108. Tris(hydroxymethyl)-aminomethane
109. Tro.meta.mol
110. Tris(hydroxymethyly)amino Methane
111. 1gng
112. Tris Base Solution
113. Trs
114. Tromethamine (usp)
115. 1h4n
116. Trometamol (jan/inn)
117. Trometamol [jan]
118. Tromethamine [ii]
119. Tromethamine [mi]
120. Tris-magnesium(ii)-potassium Chloride Buffer
121. Schembl975
122. Trishydroxymethylmethylamine
123. Tham (tn)
124. Buffer Salt, Ph 10.5
125. Ec 201-064-4
126. Tromethamine [hsdb]
127. Tromethamine [inci]
128. Trometamol [mart.]
129. Nciopen2_000263
130. Nciopen2_001720
131. Trometamol [who-dd]
132. Tromethamine [vandf]
133. 2-amino-2-hydroxymethyl-1,3-propanediol Solution
134. Oprea1_677781
135. Trishydroxymethyl Aminomethane
136. Tris-hydroxymethyl-methylamine
137. Mls000028643
138. Tris Hydroxymethyl Aminomethane
139. Tromethamine [usp-rs]
140. Tris-(hydroxymethyl)methylamine
141. Tris (hydroxymethyl)aminoethane
142. Gtpl7328
143. Tris (hydroxymethyl)aminomethane
144. Chembl1200391
145. Dtxsid2023723
146. Tris (hydroxymethyl) Methylamine
147. Tris (hydroxymethyl) Aminomethane
148. Trometamol [ep Monograph]
149. Trometamol(tris),proteomics Grade
150. 2-amino-2-hydroxymethylpropanediol
151. Hms3652l05
152. Hms3885h09
153. Tris-(hydroxymethyl)-amino-methane
154. Tromethamine [orange Book]
155. Zinc896695
156. 2-amino-2-hydroxymethyl-propane-1
157. Tris-base, Molecular Biology Grade
158. Bcp05578
159. Hy-d0227
160. Nsc65434
161. Str03166
162. Tham-e Component Tromethamine
163. Tox21_111645
164. Tox21_201646
165. Tox21_303167
166. Tromethamine [usp Monograph]
167. Bbl000011
168. Nsc-65434
169. S4176
170. Stk379529
171. 2-amino-2-methylol-propane-1,3-diol
172. Akos000121321
173. Tox21_111645_1
174. Trometamol(tris),for Molecular Biology
175. Am90366
176. Ccg-214012
177. Cs-w018524
178. Db03754
179. Pb47623
180. Trizma(r) Base, >=99.0% (t)
181. Atx Tris Buffer, Ready-to-use Solution
182. Tris(hydroxymethyl)aminomethane, >=99%
183. Tromethamine Component Of Tham-e
184. Ncgc00159412-03
185. Ncgc00159412-05
186. Ncgc00257164-01
187. Ncgc00259195-01
188. Tris, 0.5m Buffer Solution, Ph 6.8
189. Tris, 0.5m Buffer Solution, Ph 7.2
190. Tris, 0.5m Buffer Solution, Ph 7.4
191. Tris, 0.5m Buffer Solution, Ph 7.5
192. Tris, 0.5m Buffer Solution, Ph 8.0
193. Tris, 0.5m Buffer Solution, Ph 8.5
194. Tris, 0.5m Buffer Solution, Ph 8.6
195. Tris, 0.5m Buffer Solution, Ph 8.8
196. Tris, 0.5m Buffer Solution, Ph 9.0
197. Tris, 0.5m Buffer Solution, Ph 9.5
198. Tris, 1.0m Buffer Solution, Ph 6.5
199. Tris, 1.0m Buffer Solution, Ph 7.4
200. Tris, 1.0m Buffer Solution, Ph 7.6
201. Tris, 1.0m Buffer Solution, Ph 7.8
202. Tris, 1.0m Buffer Solution, Ph 8.0
203. Tris, 1.0m Buffer Solution, Ph 8.2
204. Tris, 1.0m Buffer Solution, Ph 8.4
205. Tris, 1.0m Buffer Solution, Ph 8.6
206. Tris, 1.0m Buffer Solution, Ph 8.8
207. Tris, 1.0m Buffer Solution, Ph 9.0
208. Bp-13394
209. Smr000059179
210. Tris(hydroxymethyl)aminomethane Acs Grade
211. Tris(hydroxymethyl)aminomethane, Ultrapure
212. 2-amino-2-[hydroxymethyl]-1,3-propandiol
213. Db-091324
214. Ds-014869
215. Methanamine, 1, 1,1-tris(hydroxymethyl)-
216. Tris Acidimetric, Nist(r) Srm(r) 723e
217. A0321
218. Cs-0201542
219. Cs-0201543
220. Cs-0201544
221. Ft-0611014
222. Sw219208-1
223. T2516
224. Tris-buffered Saline (tbs, 10x) Ph 7.4
225. Tris-buffered Saline (tbs, 10x) Ph 7.6
226. Tris-buffered Saline (tbs, 10x) Ph 8.0
227. Tris-buffered Saline (tbs, 20x) Ph 7.4
228. 2-(hydroxymethyl)-2-amino-1, 3-propanediol
229. 2-amino-2-(hydroxyl-methyl)propane-1,3-diol
230. 2-amino-2-(hydroxymethyl) Propane-1,3-diol
231. 2-amino-2-(hydroxymethyl)-1,3-propanediol;
232. Tris-amino, Tromethane, Trometamol, Trisamine
233. Trizma(r) Base, Bioultra, >=99.8% (t)
234. Trizma(r) Base, Tested According To Ph.eur.
235. Tromethamine, Meets Usp Testing Specifications
236. C07182
237. D00396
238. M02623
239. P17498
240. Ab00443859_03
241. Ab00443859_04
242. Q413961
243. Sigma 7-9(r), >=99% (titration), Crystalline
244. Sr-01000944234
245. Trizma(r) Base, Puriss. P.a., >=99.7% (t)
246. J-610076
247. Sr-01000944234-1
248. Trizma(r) Base, >=99.9% (titration), Crystalline
249. Trizma(r) Base, Vetec(tm) Reagent Grade, >=99%
250. Trometamol(tris), Inverted Exclamation Marky99.5%
251. W-104296
252. Tris(hydroxymethyl)aminomethane, Acs Reagent, 99.9%
253. Tris-buffered Saline (tbs, 10x, Low Salt) Ph 8.0
254. F0001-1979
255. Tris(hydroxymethyl)aminomethane, Acs Reagent, >=99.8%
256. Tris(hydroxymethyl)aminomethane, Molecular Biology Grade
257. Tris-buffered Saline (tbs, 10x, High Salt) Ph 7.4
258. Z1317839150
259. Tris(hydroxymethyl)aminomethane, P.a., Acs Reagent, 99.8%
260. Tris(hydroxymethyl)aminomethane, Ultrapure Grade, >=99.9%
261. Tris-buffered Saline (tbs, 10x), With 1% Triton X-100
262. Trizma(r) Base, Puriss. P.a., Buffer Substance, >=99.5%
263. Trometamol, European Pharmacopoeia (ep) Reference Standard
264. Tris(hydroxymethyl)aminomethane, Jis Special Grade, >=99.0%
265. Tris, 1.0m Buffer Solution, Ph 7.0, 0.2 Micron Filtered
266. Tris, 1.0m Buffer Solution, Ph 8.5, 0.2 Micron Filtered
267. Tris, 1.0m Buffer Solution, Ph 9.0, 0.2 Micron Filtered
268. Tris-buffered Saline (tbs, 10x) Ph 7.4, For Western Blot
269. Trizma(r) Base, Anhydrous, Free-flowing, Redi-dri(tm), >=99.9%
270. Trizma(r) Base, Bioultra, For Molecular Biology, >=99.8% (t)
271. Tromethamine, United States Pharmacopeia (usp) Reference Standard
272. Trizma(r) Base, Cell Culture Tested, >=99.9% (titration), Crystalline
273. Trizma(r) Base, Bioxtra, Ph 10.5-12.0 (1 M In H2o), >=99.9% (titration)
274. Trizma(r) Base, Primary Standard And Buffer, >=99.9% (titration), Crystalline
275. Tromethamine, Pharmaceutical Secondary Standard; Certified Reference Material
276. 79261-03-3
277. Trizma(r) Base, Bioperformance Certified, Meets Ep, Usp Testing Specifications, Cell Culture Tested, >=99.9% (titration)
278. Trizma(r) Base, Certified Reference Material For Titrimetry, Certified By Bam, According To Iso 17025, >=99.5%
279. Tromethamine, Pharmagrade, Manufactured Under Appropriate Controls For Use As A Raw Material In Pharma Or Biopharmaceutical Production, Suitable For Cell Culture, Meets Usp, Ep, Jpc, Bp Testing Specifications.
280. Tromethamine, Pharmagrade, Manufactured Under Appropriate Controls For Use As A Raw Material In Pharma Or Biopharmaceutical Production., Suitable For Cell Culture, Meets Usp Testing Specifications
| Molecular Weight | 121.14 g/mol |
|---|---|
| Molecular Formula | C4H11NO3 |
| XLogP3 | -2.9 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 121.07389321 g/mol |
| Monoisotopic Mass | 121.07389321 g/mol |
| Topological Polar Surface Area | 86.7 Ų |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 54 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Tham |
| PubMed Health | Tromethamine (Injection) |
| Drug Classes | Acid-Base Disorder Agent |
| Drug Label | Tham Solution (tromethamine injection) is a sterile, non-pyrogenic 0.3 M solution of tromethamine, adjusted to a pH of approximately 8.6 with glacial acetic acid. It is administered by intravenous injection, by addition to ACD blood for priming cardi... |
| Active Ingredient | Tromethamine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 3.6gm/100ml |
| Market Status | Prescription |
| Company | Hospira |
| 2 of 2 | |
|---|---|
| Drug Name | Tham |
| PubMed Health | Tromethamine (Injection) |
| Drug Classes | Acid-Base Disorder Agent |
| Drug Label | Tham Solution (tromethamine injection) is a sterile, non-pyrogenic 0.3 M solution of tromethamine, adjusted to a pH of approximately 8.6 with glacial acetic acid. It is administered by intravenous injection, by addition to ACD blood for priming cardi... |
| Active Ingredient | Tromethamine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 3.6gm/100ml |
| Market Status | Prescription |
| Company | Hospira |
Buffers; Excipients
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
/Tromethamine is indicated/ for the prevention and correction of metabolic acidosis. /Included in US product label/
Novak, K.M. (ed.). Drug Facts and Comparisons 59th Edition 2005. Wolters Kluwer Health. St. Louis, Missouri 2005., p. 130
Metabolic Acidosis Associated with Cardiac Bypass Surgery. Tromethamine solution has been found to be primarily beneficial in correcting metabolic acidosis which may occur during or immediately following cardiac bypass surgical procedures. /Included in US product label/
Medical Economics Co; Physicians Desk Reference: Generics 2nd ed p.3033 (1996)
Correction of Acidity of ACD Blood in Cardiac Bypass Surgery. It is well known that ACD blood is acidic and becomes more acidic on storage. Tromethamine effectively corrects this acidity. Tromethamine solution may be added directly to the blood used to prime the pump-oxygenator. When ACD blood is brought to a normal pH range the patient is spared an initial acid load. Additional tromethamine may be indicated during cardiac bypass surgery should metabolic acidosis appear. /Included in US product label/
Medical Economics Co; Physicians Desk Reference: Generics 2nd ed p.3033 (1996)
For more Therapeutic Uses (Complete) data for TROMETHAMINE (6 total), please visit the HSDB record page.
Local reactions associated with administration of tromethamine may include local irritation and tissue inflammation or infection at the site of injection, a febrile response, chemical phlebitis, venospasm, hypervolemia, and iv thrombosis. The drug should be administered through a large needle or indwelling catheter to minimize venous irritation by the highly alkaline tromethamine solution. Extravasation may result in inflammation, necrosis, and sloughing of overlying skin. If perivascular infiltration occurs, tromethamine administration should be discontinued immediately. Infiltration of the affected area with 1% procaine hydrochloride, to which hyaluronidase has been added, will often reduce venospasm and also will dilute any tromethamine remaining in the tissues locally. Local infiltration of an alpha-adrenergic blocking agent, such as phentolamine mesylate, into the vasospastic area has been recommended. If necessary, nerve block of autonomic fibers to the affected area may be performed.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Transient decreases in blood glucose concentration may occur during administration of tromethamine. When larger than recommended doses are used or when administration is too rapid, hypoglycemia may persist for several hours after the drug is discontinued.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Tromethamine should be slowly administered and in amounts sufficient only to correct the existing acidosis, in order to avoid overdosage and alkalosis. Determinations of blood glucose concentrations should be frequently performed during and following therapy.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Respiratory depression may occur in patients receiving large doses of tromethamine, as a result of increased blood pH and reduced carbon dioxide concentrations, and in those with chronic hypoventilation or those receiving other drugs that depress respiration. Dosage must be carefully adjusted so that blood pH does not increase above normal, and facilities for providing mechanical ventilation should be readily available during administration of tromethamine. Tromethamine may be used in conjunction with mechanical ventilatory support if respiratory acidosis is present concomitantly with metabolic acidosis.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
For more Drug Warnings (Complete) data for TROMETHAMINE (18 total), please visit the HSDB record page.
3. 3= MODERATELY TOXIC: PROBABLY ORAL LETHAL DOSE (HUMAN) 0.5-5 G/KG, BETWEEN 1 OZ & 1 PINT FOR 70 KG PERSON (150 LB).
Gosselin, R.E., H.C. Hodge, R.P. Smith, and M.N. Gleason. Clinical Toxicology of Commercial Products. 4th ed. Baltimore: Williams and Wilkins, 1976., p. II-74
For the prevention and correction of metabolic acidosis.
FDA Label
Buffers
A chemical system that functions to control the levels of specific ions in solution. When the level of hydrogen ion in solution is controlled the system is called a pH buffer. (See all compounds classified as Buffers.)
Excipients
Usually inert substances added to a prescription in order to provide suitable consistency to the dosage form. These include binders, matrix, base or diluent in pills, tablets, creams, salves, etc. (See all compounds classified as Excipients.)
B - Blood and blood forming organs
B05 - Blood substitutes and perfusion solutions
B05B - I.v. solutions
B05BB - Solutions affecting the electrolyte balance
B05BB03 - Trometamol
B - Blood and blood forming organs
B05 - Blood substitutes and perfusion solutions
B05X - I.v. solution additives
B05XX - Other i.v. solution additives
B05XX02 - Trometamol
Tromethamine is substantially eliminated by the kidneys. ... Ionized tromethamine (chiefly as the bicarbonate salt) is rapidly and preferentially excreted in urine at a rate that depends on the infusion rate. The manufacturer states that urinary excretion continues over a period of 3 days; 75% or more appears in the urine after 8 hours. In some studies, 50-75% of an iv dose was recovered in urine within 24 hours, but another study reported recovery in healthy adults to be 64% and 77% after 2 and 3 days, respectively.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
It is not known whether tromethamine is distributed in human milk.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Ionized tromethamine is excreted by kidney, so the effect is that of excretion of hydrogen ions. Elimination of drug from body is entirely by renal excretion. Excretion of tromethamine is accompanied by osmotic diuresis, since clinical doses of drug considerably add to osmolarity of glomerular filtrate.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 773
In rats of different age (5 to 240 days old) the renal excretion of Trishydroxymethylaminomethane (THAM) was studied. In 5 and in 240 days old rats the renal excretion of THAM was slower than in rats of other age groups. Stimulation of diuresis by i.p. injection of mannitol, thiazide or by oral water load resulted in an increase in THAM excretion in 5 and in 240 days old rats. The renal excretion of THAM was also increased by repeated administration of THAM in all age groups, except in new born rats. Possible mechanisms of action are discussed.
PMID:240333 Braunlich H; Arch Int Pharmacodyn Ther 216 (1): 144-59 (1975)
The distribution of 14C labelled THAM (tris-hydroxymethylaminomethane) was determined between intra- and extracellular space of nephrectomized Sprague-Dawley rats as a function of time at constant plasma pH of 7.4. The following results were obtained: An equilibrium in the distribution of THAM between ECS and ICS will not occur before 6-12 hours after administration. This indicates that THAM permeates very slowly into the intracellular compartment, which is in contrast to the general assumption that it quickly diffuses into the intracellular space to restore the intracellular acidosis. THAM disappears from the extracellular space in a multiexponential fashion, indicating that it equilibrates with the different body tissues at largely variable rates. The equilibrium which occurs between both body compartments 6-12 hours after THAM application does not agree with the values which are expected for transfer of only the nonionised substance. At plasma pH 7.4 and a "mean whole body pHi" of 6.88, THAM is distributed with a distribution ratio of 4 (ICS/ECS), a value quite different from the value of 11 which would be expected for exclusive nonionic diffusion. Thus THAM is also transferred across the cell membrane in ionized form. These results indicate that the influx of THAM into the intracellular space is too slow (when compared to the renal elimination kinetics) to influence intracellular pH significantly by direct buffer action. Moreover, only a fraction of THAM enters the intracellular space in the nonionized form, thus reducing (to an even greater extent) the direct effect of THAM on the intracellular acid-base equilibrium.
PMID:6711774 Rothe KF, Heisler N; Anasth Intensivther Notfallmed 19 (1): 24-6 (1984)
Tromethamine is not metabolized appreciably.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Tromethamine is an alkalinizing agent which acts as a proton (hydrogen ion) acceptor. Tromethamine is a weak base; following IV injection, it attracts and combines with hydrogen ions and their associated acid anions and the resulting salts are excreted in urine. Tromethamine can combine with lactic, pyruvic, and other metabolic acids and with carbonic acid. ... At pH 7.4, approximately 70% of the tromethamine present in plasma is in the ionized (protonated) form; if pH is decreased from pH 7.4, the ionized fraction of the drug is increased. In contrast to the ionized fraction of tromethamine, which upon administration reacts only with acid in the extracellular fluids, the fraction of the dose which remains un-ionized at physiologic pH is thought to be capable of penetrating the cell membrane to combine with intracellular acid. Since administration of tromethamine reduces hydrogen ion concentration, there is a decrease in proton donor and an increase in proton acceptor concentrations in body buffers. In the bicarbonate:carbonic acid buffer, the concentration of dissolved carbon dioxide is decreased (at least until regulatory mechanisms compensate) and the concentration of bicarbonate is increased. The reduction of carbon dioxide tension removes a potent stimulus to breathing and may result in hypoventilation and hypoxia.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
Tromethamine ... acts as a weak, osmotic diuretic, increasing the flow of alkaline urine containing increased amounts of electrolytes.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2647
By removing protons from hydronium ions, ionization of carbonic acid is shifted so as to decrease pCO2 and to increase bicarbonate. Excess bicarbonate is then gradually excreted in kidney. /Tromethamine is an/ especially useful way to manage excessively high pCO2 in respiratory acidosis...
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 773




