
Synopsis
Synopsis
0
CEP/COS
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
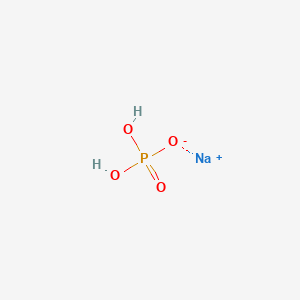

1. Dibasic Sodium Phosphate, Anhydrous
2. Disodium Acid Phosphate
3. Disodium Hydrogen Phosphate
4. Disodium Hydrogen Phosphate Anhydrous
5. Monosodium Dihydrogen Phosphate
6. Neutral Sodium Hydrogen Phosphate
7. Phosphoric Acid, Disodium Salt
8. Phosphoric Acid, Disodium Salt, 32p-labeled
9. Phosphoric Acid, Disodium Salt, Anhydrous
10. Phosphoric Acid, Disodium Salt, Dodecahydrate
11. Phosphoric Acid, Disodium Salt, Heptahydrate
12. Phosphoric Acid, Monosodium Salt
13. Phosphoric Acid, Monosodium Salt, Anhydrous
14. Phosphoric Acid, Sodium (2:3) Salt
15. Phosphoric Acid, Sodium Salt
16. Phosphoric Acid, Trisodium Salt
17. Phosphoric Acid, Trisodium Salt , 32p-labeled
18. Phosphoric Acid, Trisodium Salt , Dodecahydrate
19. Sodium Biphosphate
20. Sodium Dihydrogen Orthophosphate
21. Sodium Dihydrogen Phosphate
22. Sodium Hydrophosphate
23. Sodium Phosphate
24. Sodium Phosphate Monobasic Anhydrous
25. Sodium Phosphate, Dibasic
26. Sodium Phosphate, Dibasic (anhydrous)
27. Sodium Phosphate, Disodium Salt
28. Sodium Phosphate, Monobasic
29. Sodium Phosphate, Monobasic Anhydrous
30. Sodium Phosphate, Tribasic
31. Sodium Phosphate, Tribasic, Dodecahydrate
32. Trisodium Phosphate
33. Trisodium Phosphate Dodecahydrate
1. 7558-80-7
2. Sodium Dihydrogen Phosphate
3. Sodium Phosphate Monobasic
4. Sodium Dihydrogenorthophosphate
5. Sodium Acid Phosphate
6. Sodium Primary Phosphate
7. Monosodium Dihydrogen Orthophosphate
8. Monosodium Monophosphate
9. Sodium Dihydrogenphosphate
10. Monosodium Phosphate, Anhydrous
11. Sodium Phosphate Monobasic Anhydrous
12. Monobasic Sodium Phosphate
13. Acid Sodium Phosphate
14. Monosodium Orthophosphate
15. Sodium Dihydrogen Orthophosphate
16. Monosodium Dihydrogen Phosphate
17. Sodium Dihydrogen Phosphate, Anhydrous
18. Sodium Phosphate (na(h2po4))
19. Sodium;dihydrogen Phosphate
20. Sodium Biphosphate, Anhydrous
21. Sodium Orthophosphate Monobasic
22. Nah2po4
23. Sodium Phosphate,monobasic
24. Kh7i04hpuu
25. Sodium Phosphate, Monobasic Anhydrous
26. Ins No.339(i)
27. Chebi:37585
28. Sodium Phosphate, Monobasic, Anhydrous
29. Monosodium Dihydrogen Monophosphate
30. Ins-339(i)
31. Mfcd00003527
32. Sodium Phosphate Monobasic (anhydrate)
33. Sodium Phosphate Monobasic (anhydrous)
34. 89140-32-9
35. E-339(i)
36. Phosphoric Acid, Monosodium Salt, Anhydrous
37. Monosorb Xp-4
38. Phosphoric Acid, Sodium Salt
39. Primary Sodium Phosphate
40. Mfcd00146206
41. Sodium Biphosphate Anhydrous
42. Monosodium Hydrogen Phosphate
43. Sodium Phosphate (nah2po4)
44. Sodium Orthophosphate, Primary
45. Sodium Dihydrogen Monophosphate
46. Hsdb 738
47. Einecs 231-449-2
48. Unii-kh7i04hpuu
49. Sodium Dihydrogen Phosphate (1:2:1)
50. Sodium Monobasic Phosphate (nah2po4)
51. Sodium Dihydrogen Phosphate (nah2po4)
52. Phosphosoda
53. Buromin
54. Clicolon
55. Armite
56. Instant Calgon
57. Sodiumdihydrogenphosphate-16o4
58. Turrixin St
59. Hy-phos
60. Hemisodium Phosphate
61. Sodium Phosphate Monobasic, Anhydrous
62. Sodium Phosphate Glass
63. Sodiumdihydrogenphosphate
64. Sodium Dihydrogenphoshate
65. Sodium Dihydrogenphospate
66. Sodium Dihydrogenphosphat
67. Sodiumdihydrogen Phosphate
68. Natriumdihydrogen-phosphate
69. Sodium Dihydrogen-phosphate
70. Sodium Phosphate Mono-basic
71. Ec 231-449-2
72. Unii-se337svy37
73. Sodium Salt Of Phosphoric Acid
74. Inkp 100
75. Inkp-100
76. Se337svy37
77. Dtxsid7035222
78. Sodium Phosphate Monobasic Solution
79. Sodium Phosphate, Monobasic (usp)
80. Anhydrous Monobasic Sodium Phosphate
81. Einecs 231-558-5
82. Sodium Dihydrogenphosphate (granular)
83. Phosphoric Acid, Sodium Salt (1:?)
84. Phosphoric Acid, Sodium Salt (1:1)
85. Akos024433265
86. At32901
87. Db09449
88. Sodium Phosphate Monobasic [hsdb]
89. Sodium Phosphate Monobasic, Ar, >=98%
90. Sodium Phosphate Monobasic, Lr, >=97%
91. Sodium Phosphate, Monobasic [mi]
92. E339
93. Sodium Phosphate,monobasic [vandf]
94. Phosphate, 0.2m Buffer Solution, Ph 4.4
95. Phosphate, 0.2m Buffer Solution, Ph 6.8
96. Phosphate, 0.2m Buffer Solution, Ph 7.0
97. Phosphate, 0.2m Buffer Solution, Ph 7.2
98. Phosphate, 0.2m Buffer Solution, Ph 7.4
99. Phosphate, 0.2m Buffer Solution, Ph 7.5
100. Phosphate, 0.2m Buffer Solution, Ph 7.6
101. Phosphate, 0.5m Buffer Solution, Ph 6.5
102. Phosphate, 0.5m Buffer Solution, Ph 7.0
103. Phosphate, 0.5m Buffer Solution, Ph 7.2
104. Phosphate, 0.5m Buffer Solution, Ph 7.4
105. Phosphate, 0.5m Buffer Solution, Ph 8.0
106. Phosphate, 0.5m Buffer Solution, Ph 8.5
107. Phosphate, 0.5m Buffer Solution, Ph 9.0
108. B7779
109. Ft-0698935
110. Sodium Phosphate Monobasic, For Hplc, 99%
111. D04400
112. Sodium Phosphate Monobasic, Bioxtra, >=99.0%
113. Q415877
114. Sodium Phosphate Monobasic, Usp, 98.0-103.0%
115. Sodium Phosphate, 0.2m Buffer Solution, Ph 7.0
116. Sodium Phosphate, 0.2m Buffer Solution, Ph 7.2
117. Sodium Phosphate, 0.2m Buffer Solution, Ph 7.4
118. Sodium Phosphate, 0.2m Buffer Solution, Ph 7.5
119. Sodium Phosphate, 0.2m Buffer Solution, Ph 7.6
120. Sodium Phosphate, 0.2m Buffer Solution, Ph 8.0
121. Sodium Phosphate, 0.2m Buffer Solution, Ph 8.5
122. Sodium Phosphate, 0.2m Buffer Solution, Ph 9.0
123. Sodium Phosphate, 0.2m Buffer Solution, Ph 9.5
124. Sodium Phosphate, 0.5m Buffer Solution, Ph 7.0
125. Sodium Phosphate, 0.5m Buffer Solution, Ph 7.5
126. Sodium Phosphate, 0.5m Buffer Solution, Ph 7.6
127. Sodium Phosphate, 0.5m Buffer Solution, Ph 8.0
128. Sodium Phosphate, 0.5m Buffer Solution, Ph 8.5
129. Sodium Phosphate, 0.5m Buffer Solution, Ph 9.0
130. Sodium Phosphate, 0.5m Buffer Solution, Ph 9.5
131. Sodium Phosphate, Monobasic, Anhydrous [ii]
132. 2-methyl-3-(pyridin-4-ylmethyl)quinazolin-4(3h)-one
133. Sodium Phosphate Monobasic, Reagentplus(r), >=99.0%
134. Sodium Phosphate Monobasic (anhydrous) [who-dd]
135. Sodium Phosphate Monobasic, 99.999% Trace Metals Basis
136. Sodium Phosphate Monobasic Solution, Bioultra, 5 M In H2o
137. Sodium Phosphate Monobasic, Vetec(tm) Reagent Grade, 99%
138. Sodium Phosphate, Monobasic Anhydrous [orange Book]
139. Sodium Phosphate, Monobasic, Anhydrous [orange Book]
140. Sodium Phosphate Monobasic, Saj First Grade, 98.0-101.0%
141. Sodium Phosphate Monobasic, Saj Special Grade, 99.0-101.0%
142. Sodium Phosphate Monobasic, Purum P.a., Anhydrous, >=99.0% (t)
143. Sodium Phosphate Monobasic, Meets Usp Testing Specifications, Anhydrous
144. Sodium Phosphate Monobasic, Anhydrous, Free-flowing, Redi-dri(tm), >=99.0%
145. Sodium Phosphate Monobasic, Bioreagent, For Molecular Biology, Anhydrous, >=98%
146. Sodium Phosphate Monobasic, Biotechnology Performance Certified, Cell Culture Tested
147. Sodium Phosphate Monobasic Molecular Biology Grade, Suitable For Cell Culture, Insect Cell Culture, And Plant Cell Culture
148. Sodium Phosphate Monobasic, Bioperformance Certified, Suitable For Cell Culture, Suitable For Insect Cell Culture, Suitable For Plant Cell Culture, >=99.0% (titration)
| Molecular Weight | 119.977 g/mol |
|---|---|
| Molecular Formula | H2NaO4P |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 119.95883982 g/mol |
| Monoisotopic Mass | 119.95883982 g/mol |
| Topological Polar Surface Area | 80.6 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 61.9 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Visicol Tablets are indicated for cleansing of the colon as a preparation for colonoscopy in adults 18 years of age or older. /Included in US product label; sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous/
NIH; DailyMed. Current Medication Information for VISICOL (sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous) tablet (December, 2010). Available from, as of October 3, 2014: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=57ccd52e-5b13-4b39-8aa0-ebecc49a7db4
To determine whether phosphate supplementation, started soon after birth in adequate quantity, would prevent rickets in very low birth weight infants with prenatal deficiency of phosphate, 40 neonates were given an initial dose of 50 mg/day of phosphate administered as a mixture of 189 g of sodium phosphate dibasic (disodium hydrogen phosphate) and 82 g of sodium phosphate monobasic (sodium dihydrogen phosphate) made up to 2 liters with single strength chloroform water or placebo (single strength chloroform water). Supplementation was increased to 37.5 mg every 12 hr if the plasma phosphate concentration remained less than 1.5 mmol/L after one wk. Results showed that no infant receiving phosphate supplements had radiological evidence of rickets whereas bone changes were apparent in 42% of the control group. It was concluded that prenatal deficiency of phosphate, due to placental insufficiency, can be corrected by phosphate supplementation, thereby preventing rickets of prematurity.
PMID:1969066 Holland PC et al; Lancet 335: 697-701 (1990)
The objective of this study was to determine the safety and efficacy of 0.15 mmol/kg phosphorus (PHOS), administered intravenously as sodium or potassium phosphate over 120 minutes, in the treatment of adults suffering from severe hypophosphatemia. Severe hypophosphatemia was defined as a serum PHOS concentration of /LE/ 1.5 mg/dL. Exclusion criteria were renal impairment and hypercalcemia. Patient assessments included mental status, heart rate, and blood pressure. The timing of post-infusion serum PHOS sampling was at physician discretion. Six men and four women were enrolled in the study. During the study period, the only parenteral PHOS administered was the study dose. There were no patient adverse events associated with PHOS administration. One patient who received potassium phosphates had an elevated post-infusion serum potassium (5.2 mEq). Serum PHOS increased above the study criteria for severe hypophosphatemia in all ten patients, although nine patients received concomitant oral PHOS supplements. The dosing of intravenous sodium or potassium phosphate in the treatment of patients with severe hypophosphatemia is empiric. Historical evidence of toxicity has caused dosing recommendations to be low and slow. These data demonstrate the safety of a moderate PHOS dose when administered over two hours to adults, as measured by patient mental status, vital signs, and blood chemistry analysis.
Rice TL, Alaniz C; ASHP Midyear Clinical Meeting 26: PCR-13 (1991)
Sixty patients were randomly divided into three groups of 20 each. Each group was submitted to a bowel preparation with one of the following solutions: 10% manitol, sodium picosulfate or sodium phosphate. The parameters evaluated were: taste, tolerance, associated side effects and quality of cleansing. Postural blood pressure and pulse rate as well as serum sodium, potassium, calcium and phosphate were compared. ... Sodium phosphate and 10% manitol solutions provided superior results in terms of colon cleansing compared to sodium picosulfate solution...
PMID:18516457 Miki P Jr et al; Acta Cir Bras 23 (Supp 1): 108-11 (2008)
For more Therapeutic Uses (Complete) data for SODIUM DIHYDROGEN PHOSPHATE (9 total), please visit the HSDB record page.
/BOXED WARNING/ There have been rare, but serious reports of acute phosphate nephropathy in patients who received oral sodium phosphate products for colon cleansing prior to colonoscopy. Some cases have resulted in permanent impairment of renal function and some patients required long-term dialysis. While some cases have occurred in patients without identifiable risk factors, patients at increased risk of acute phosphate nephropathy may include those with increased age, hypovolemia, increased bowel transit time (such as bowel obstruction), active colitis, or baseline kidney disease, and those using medicines that affect renal perfusion or function (such as diuretics, angiotensin converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], and possibly nonsteroidal anti-inflammatory drugs [NSAIDs]).
NIH; DailyMed. Current Medication Information for VISICOL (sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous) tablet (December, 2010). Available from, as of October 3, 2014: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=57ccd52e-5b13-4b39-8aa0-ebecc49a7db4
FDA has become aware of reports of acute phosphate nephropathy, a type of acute kidney injury, associated with the use of oral sodium phosphate products (OSP) for bowel cleansing prior to colonoscopy or other procedures. These products include the prescription products, Visicol and OsmoPrep, and OSPs available over-the-counter without a prescription as laxatives (e.g., Fleet Phospho-soda). In some cases when used for bowel cleansing, these serious adverse events have occurred in patients without identifiable factors that would put them at risk for developing acute kidney injury. We cannot rule out, however, that some of these patients were dehydrated prior to ingestion of OSPs or they did not drink sufficient fluids after ingesting OSP. Acute phosphate nephropathy is a form of acute kidney injury that is associated with deposits of calcium-phosphate crystals in the renal tubules that may result in permanent renal function impairment. Acute phosphate nephropathy is a rare, serious adverse event that has been associated with the use of OSPs. The occurrence of these events was previously described in an Information for Healthcare Professionals sheet and an FDA Science Paper issued in May 2006. Additional cases of acute phosphate nephropathy have been reported to FDA and described in the literature since these were issued. Individuals who appear to have an increased risk of acute phosphate nephropathy following the use of OSPs include persons: who are over age 55; who are hypovolemic or have decreased intravascular volume; who have baseline kidney disease, bowel obstruction, or active colitis; and who are using medications that affect renal perfusion or function (such as diuretics, angiotensin converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], and possibly nonsteroidal anti-inflammatory drugs [NSAIDs]). As a result of new safety information received, FDA is requiring the manufacturer of Visicol and OsmoPrep, the two OSPs available by prescription only, to add a Boxed Warning to the labeling for these products. FDA is also requiring that the manufacturer develop and implement a risk evaluation and mitigation strategy (REMS), which will include a Medication Guide, to ensure that the benefits of these products outweigh the risk of acute phosphate nephropathy, and to conduct a postmarketing clinical trial to further assess the risk of acute kidney injury with use of these products. /Sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous/
FDA/CDER; FDA Alert: Oral Sodium Phosphate (OSP) Products for Bowel Cleansing (marketed as Visicol and OsmoPrep, and oral sodium phosphate products available without a prescription) (12/11/2008). Available from, as of January 28, 2014: https://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm126084.htm
Sodium phosphate and 10% mannitol solutions provided superior results in terms of colon cleansing compared to sodium picosulfate solution. All serum electrolytes evaluated were significantly altered in the three groups, without important clinical signs. High levels of serum phosphate were the most striking alteration in patients prepared with sodium phosphate solution, again with no clinical signs. Variations related to blood pressure and pulse rate suggested contraction of intravascular volume, with no clinical effects.
PMID:18516457 Miki P Jr et al; Acta Cir Bras 23 (Supp 1): 108-11 (2008)
Fifteen male subjects received 50 mL of commerical laxative containing 24 g of sodium biphosphate (sodium phosphate monobasic) and 6 g of sodium phosphate (sodium phosphate dibasic; I) (7 g of elemental phosphorus) administered with 500 mL of water and 11 patients received 300 mL of magnesium citrate (II) containing 3.2 g of elemental magnesium. Patients ranged in age from 26 to 86 yr. Serum magnesium, calcium, phosphorus, total protein, and albumin were determined before and at various intervals up to 16 hr after administration of the laxative and prior to radiological study. The administration of I in conventional doses to normal subjects prior to barium enema resulted in a striking increase in serum phosphorus levels followed by a decline in serum calcium levels in all subjects. Changes were highly significant when compared with control subjects who were prepared for the same procedure with II. Levels of serum potassium also decreased significantly but not serum sodium, chloride, bicarbonate, or magnesium. It would seem wise to caution against the use of phosphate containing laxatives in the presence of severe renal insufficiency, hypocalcemia, or convulsive disorders. In patients who are receiving frequent, repetitive dosages because of difficulty in cleansing the bowel for radiological procedures, serum calcium should be closely monitored. /Sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous/
PMID:666471 Wiberg JJ et al; Arch Intern Med 138: 1114-6 (1978)
For more Drug Warnings (Complete) data for SODIUM DIHYDROGEN PHOSPHATE (39 total), please visit the HSDB record page.
The estimated fatal dose of sodium phosphates is 50 g.
Dreisbach, R.H. Handbook of Poisoning. 12th ed. Norwalk, CT: Appleton and Lange, 1987., p. 212
Used to treat constipation or to clean the bowel before a colonoscopy.
FDA Label
Sodium phosphate inceases fecal water content to increase mobility through the large intestine.
Absorption
Tmax for phosphate absorption with orally administered liquid sodium phosphate is 1-3h.
... Phosphates are slowly and incompletely absorbed ... . /Dibasic and monobasic sodium phosphate/
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-120
Intravenously infused phosphorus not taken up by the tissues is excreted almost entirely in the urine. Plasma phosphorus is believed to be filterable by the renal glomeruli, and the major portion of filtered phosphorus (greater than 80%) is actively reabsorbed by the tubules. Many modifying influences tend to alter the amount excreted in the urine.
US Natl Inst Health; DailyMed. Current Medication Information for Sodium Phosphates (Sodium Phosphate) Injection (June 2006). Available from, as of March 20, 2009: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1758
An open-label pharmacokinetic study of Visicol in healthy volunteers was performed to determine the concentration-time profile of serum inorganic phosphorus levels after Visicol administration. All subjects received a total of 60 grams of sodium phosphate with a total liquid volume of 3.6 quarts. Subjects received a 30 gram dose (20 tablets given as 3 tablets every 15 minutes with 8 ounces of clear liquids) beginning at 6 PM and then received a second 30 gram dose (20 tablets given as 3 tablets every 15 minutes with 8 ounces of clear liquids) the following morning beginning at 6 AM. Twenty-three healthy subjects (mean age 57 years old; 57% male and 43% female; and 65% Hispanic, 30% Caucasian, and 4% African-American) participated in this pharmacokinetic study. The serum phosphorus level rose from a mean (+/- standard deviation) baseline of 4.0 (+/- 0.7) mg/dL to 7.7 (+/- 1.6 mg/dL), at a median of 3 hours after the administration of the first 30 gram dose of Visicol tablets The serum phosphorus level rose to a mean of 8.4 (+/- 1.9) mg/dL, at a median of 4 hours after the administration of the second 30 gram dose of Visicol tablets. The serum phosphorus level remained above baseline for a median of 24 hours after the administration of the initial dose of Visicol tablets (range 16 to 48 hours).
US Natl Inst Health; DailyMed. Current Medication Information for VISICOL (sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous) tablet (November 2008). Available from, as of March 20, 2009: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1758
Sodium phosphate is thought to work by increasing the amount of solute present in the intestinal lumen thereby creating an osmotic gradient which draws water into the lumen.
... /Promotes/ defecation by retaining water in the intestinal lumen through osmotic forces. ... May also act by stimulating release of cholecystokinin. /Sodium phosphate & sodium biphosphate (Fleet's enema & Fleet's Phospho-soda)/
Miller, R. R., and D. J. Greenblatt. Handbook of Drug Therapy. New York: Elsevier North Holland, 1979., p. 1055
Phosphorus in the form of organic and inorganic phosphate has a variety of important biochemical functions in the body and is involved in many significant metabolic and enzyme reactions in almost all organs and tissues. It exerts a modifying influence on the steady state of calcium levels, a buffering effect on acid-base equilibrium and a primary role in the renal excretion of hydrogen ion.
US Natl Inst Health; DailyMed. Current Medication Information for Sodium Phosphates (Sodium Phosphate) Injection (June 2006). Available from, as of March 20, 2009: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1758

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Market Place

ABOUT THIS PAGE
65
PharmaCompass offers a list of Monosodium Phosphate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Monosodium Phosphate manufacturer or Monosodium Phosphate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Monosodium Phosphate manufacturer or Monosodium Phosphate supplier.
PharmaCompass also assists you with knowing the Monosodium Phosphate API Price utilized in the formulation of products. Monosodium Phosphate API Price is not always fixed or binding as the Monosodium Phosphate Price is obtained through a variety of data sources. The Monosodium Phosphate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 04269_SIAL manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 04269_SIAL, including repackagers and relabelers. The FDA regulates 04269_SIAL manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 04269_SIAL API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 04269_SIAL manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A 04269_SIAL supplier is an individual or a company that provides 04269_SIAL active pharmaceutical ingredient (API) or 04269_SIAL finished formulations upon request. The 04269_SIAL suppliers may include 04269_SIAL API manufacturers, exporters, distributors and traders.
click here to find a list of 04269_SIAL suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A 04269_SIAL DMF (Drug Master File) is a document detailing the whole manufacturing process of 04269_SIAL active pharmaceutical ingredient (API) in detail. Different forms of 04269_SIAL DMFs exist exist since differing nations have different regulations, such as 04269_SIAL USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 04269_SIAL DMF submitted to regulatory agencies in the US is known as a USDMF. 04269_SIAL USDMF includes data on 04269_SIAL's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 04269_SIAL USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 04269_SIAL suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The 04269_SIAL Drug Master File in Japan (04269_SIAL JDMF) empowers 04269_SIAL API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the 04269_SIAL JDMF during the approval evaluation for pharmaceutical products. At the time of 04269_SIAL JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of 04269_SIAL suppliers with JDMF on PharmaCompass.
A 04269_SIAL written confirmation (04269_SIAL WC) is an official document issued by a regulatory agency to a 04269_SIAL manufacturer, verifying that the manufacturing facility of a 04269_SIAL active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting 04269_SIAL APIs or 04269_SIAL finished pharmaceutical products to another nation, regulatory agencies frequently require a 04269_SIAL WC (written confirmation) as part of the regulatory process.
click here to find a list of 04269_SIAL suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 04269_SIAL as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 04269_SIAL API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 04269_SIAL as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 04269_SIAL and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 04269_SIAL NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 04269_SIAL suppliers with NDC on PharmaCompass.
04269_SIAL Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 04269_SIAL GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 04269_SIAL GMP manufacturer or 04269_SIAL GMP API supplier for your needs.
A 04269_SIAL CoA (Certificate of Analysis) is a formal document that attests to 04269_SIAL's compliance with 04269_SIAL specifications and serves as a tool for batch-level quality control.
04269_SIAL CoA mostly includes findings from lab analyses of a specific batch. For each 04269_SIAL CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
04269_SIAL may be tested according to a variety of international standards, such as European Pharmacopoeia (04269_SIAL EP), 04269_SIAL JP (Japanese Pharmacopeia) and the US Pharmacopoeia (04269_SIAL USP).

