
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
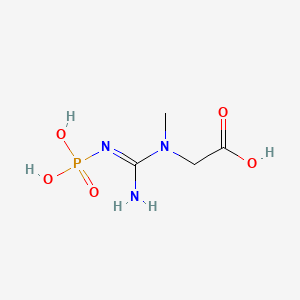

1. Creatine Phosphate
2. Disodium Salt Phosphocreatine
3. Neoton
4. Phosphate, Creatine
5. Phosphocreatine, Disodium Salt
6. Phosphorylcreatine
1. Creatine Phosphate
2. Phosphorylcreatine
3. N-phosphocreatine
4. Fosfocreatine
5. Creatine-p
6. 67-07-2
7. N-(phosphonoamidino)sarcosine
8. N-phosphorylcreatine
9. Phosphagen
10. Creatinephosphoric Acid
11. N-(phosphonoamidino)-sarcosine
12. Creatine Phosphic Acid
13. N-(n-phosphonoamido)sarcosine
14. Chebi:17287
15. 020iuv4n33
16. 2-(1-methyl-3-phosphonoguanidino)acetic Acid
17. Glycine, N-(imino(phosphonoamino)methyl)-n-methyl-
18. 2-(3-methyl-1-phosphonocarbamimidamido)acetic Acid
19. Glycine, N-[imino(phosphonoamino)methyl]-n-methyl-
20. N-phosphorocreatine
21. Einecs 200-643-9
22. [[imino(phosphonoamino)methyl](methyl)amino]acetic Acid
23. {[imino(phosphonoamino)methyl](methyl)amino}acetic Acid
24. Unii-020iuv4n33
25. N-methyl-n-(n-phosphonocarbamimidoyl)glycine
26. N(omega)-phosphonocreatine
27. Bmse000079
28. Phosphocreatine [mi]
29. Chembl1204644
30. Dtxsid0058776
31. Creatine Phosphate [mart.]
32. Creatine Phosphate [who-dd]
33. Hy-d0885
34. Mfcd00152044
35. Stl452993
36. Akos027340220
37. Db13191
38. As-12479
39. 2-(1-methyl-3-phosphonoguanidino)aceticacid
40. Cs-0013742
41. C02305
42. N-[imino(phosphonoamino)methyl]-n-methyl-glycine
43. 067c072
44. A917893
45. 2-(1-methyl-3-phosphonocarbamimidamido)acetic Acid
46. W-104734
47. 2-[methyl-(n'-phosphonocarbamimidoyl)amino]acetic Acid
| Molecular Weight | 211.11 g/mol |
|---|---|
| Molecular Formula | C4H10N3O5P |
| XLogP3 | -2.6 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Exact Mass | 211.03580743 g/mol |
| Monoisotopic Mass | 211.03580743 g/mol |
| Topological Polar Surface Area | 136 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 271 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Phosphocreatine is a naturally occuring substance that is found predominantly in the skeletal muscles of vertebrates. Its primary utility within the body is to serve in the maintanence and recycling of adenosine triphosphate (ATP) for muscular activity like contractions. Given this utility of phosphocreatine to recycle ATP, the most plausible therapeutic potentials for its use involve conditions caused by energy shortage or by increased energy requirements - such as in ischemic stroke and other cerebrovascular diseases. It is important to note however that relatively little clinical research has been done to significantly further the evidence for any such indications, although it is administered intravenously for cardiovascular conditions in some countries. Additionally, because phosphocreatine is not regulated as a controlled substance it is taken as a supplement by some professional athletes as a means to perhaps increase short bursts of muscle strength or energy for professional athletics.
Creatine is a naturally occurring chemical within the body and is primarily stored in skeletal muscle in both free and phosphorylated forms. Phosphocreatine is the name given to the phosphorylated form of creatine. Additionally, phosphocreatine can also be found in other areas of the body like the kidneys, liver, and brain. In fact, most *in vivo* synthesis of creatine occurs in the liver where amidine groups from arginine are transfered to glycine with the help of the glycine transaminidase enzyme to form guanidinoacetic acid. This acid is then methylated with the methyl group of S-adenosylmethionine via guanidinoacetate methyltransferase to generate creatine. The synthesized creatine is transported to storage sites in skeletal muscle via the bloodstream. The phosphorylation of creatine is reversible in both a forwards and backwards reaction. That is, while phosphocreatine is capable of anaerobically donating a phosphate group to adenosine diphosphate (ADP) to regenerate ATP, at the same time excess ATP can be dephosphorylated during periods of low muscle activity to convert creatine to phosphocreatine. This dual activity in synthesizing phosphocreatine from excess levels of ATP during rest and use of phosphocreatine to regenerate ATP during high activity demonstrates the crucial utility of phosphocreatine in acting as an energy buffer in body mucle cells. Phosphocreatine's fast regeneration of ATP is considered a coupled reaction - in essence, the energy released from transferring a donating a phosphate group from phosphocreatine is used to regenerate ATP. Phosphocreatine consequently plays an essential role in body tissues that have high, fluctuating energy requirments like muscle and brain tissues.
Cardiotonic Agents
Agents that have a strengthening effect on the heart or that can increase cardiac output. They may be CARDIAC GLYCOSIDES; SYMPATHOMIMETICS; or other drugs. They are used after MYOCARDIAL INFARCT; CARDIAC SURGICAL PROCEDURES; in SHOCK; or in congestive heart failure (HEART FAILURE). (See all compounds classified as Cardiotonic Agents.)
C - Cardiovascular system
C01 - Cardiac therapy
C01E - Other cardiac preparations
C01EB - Other cardiac preparations
C01EB06 - Fosfocreatine
Route of Elimination
Phosphocreatine is eliminated renally. The end result of creatine degredation is the product creatinine, which enters the bloodstream from its storage sites in body muscle. When creatinine enters the renal parenchyma it is filtered in the renal glomerulus to be excreted in the urine.
Adenosine triphosphate (ATP) is the primary source of chemical energy that body muscles use to perform contractions. During such contraction processes, ATP molecules are depleted as they undergo hydrolysis reactions and become adenosine diphosphate (ADP). To maintain homeostasis in muscle activity, the ATP supply of muscles must be regenerated regularly. Phosphocreatine occurs naturally within the body and is capable of regenerating ATP by transferring a high-energy phosphate from itself to ADP, resulting in the formation of ATP and creatine. This kind of regeneration of ATP with phosphocreatine typically occurs within seconds of intense muscular or neuronal effort, acting as a quickly accessible reserve of high-energy phosphates for the recycling of ATP in body muscle tissues. ATP recycling from phosphocreatine is in fact known as the quickest form of ATP regeneration.
Market Place

ABOUT THIS PAGE
90
PharmaCompass offers a list of Phosphocreatine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Phosphocreatine manufacturer or Phosphocreatine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Phosphocreatine manufacturer or Phosphocreatine supplier.
A 020IUV4N33 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 020IUV4N33, including repackagers and relabelers. The FDA regulates 020IUV4N33 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 020IUV4N33 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 020IUV4N33 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 020IUV4N33 supplier is an individual or a company that provides 020IUV4N33 active pharmaceutical ingredient (API) or 020IUV4N33 finished formulations upon request. The 020IUV4N33 suppliers may include 020IUV4N33 API manufacturers, exporters, distributors and traders.
click here to find a list of 020IUV4N33 suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
020IUV4N33 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 020IUV4N33 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 020IUV4N33 GMP manufacturer or 020IUV4N33 GMP API supplier for your needs.
A 020IUV4N33 CoA (Certificate of Analysis) is a formal document that attests to 020IUV4N33's compliance with 020IUV4N33 specifications and serves as a tool for batch-level quality control.
020IUV4N33 CoA mostly includes findings from lab analyses of a specific batch. For each 020IUV4N33 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
020IUV4N33 may be tested according to a variety of international standards, such as European Pharmacopoeia (020IUV4N33 EP), 020IUV4N33 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (020IUV4N33 USP).

