
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
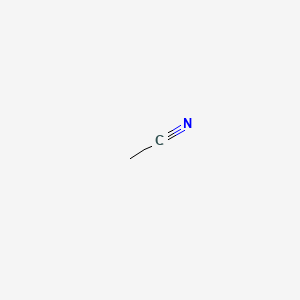

1. Acetonitrile, 1-(14)c-labeled
2. Acetonitrile, 3h-labeled
1. Cyanomethane
2. Methyl Cyanide
3. 75-05-8
4. Ethanenitrile
5. Ethyl Nitrile
6. Methanecarbonitrile
7. Methane, Cyano-
8. Acetonitril
9. Cyanure De Methyl
10. Methylkyanid
11. Mecn
12. Methylkyanid [czech]
13. Usaf Ek-488
14. Rcra Waste Number U003
15. Nci-c60822
16. Cyanure De Methyl [french]
17. Acetonitril [german, Dutch]
18. Acetonitrile, Anhydrous
19. Ch3cn
20. Ncme
21. Acetonitrile, Dimer
22. 148642-19-7
23. Acetonitrile With 0.1% Ammonium Acetate
24. Ch3-c#n
25. Chebi:38472
26. Z072sb282n
27. Nsc-7593
28. Acetonitrile With 0.1per Cent Ammonium Acetate
29. Mfcd00001878
30. Acetonitrile [un1648] [flammable Liquid]
31. Acetonitril (german, Dutch)
32. Acetonitrile, For Dna Synthesis
33. Acetnitrile
34. Ethanonitrile
35. 66016-35-1
36. Ccn
37. Hsdb 42
38. Ccris 1628
39. Nsc 7593
40. Acetonitrile, For Hplc, Gradient Grade, >=99.9%
41. Einecs 200-835-2
42. Un1648
43. Rcra Waste No. U003
44. Acetonitile
45. Acetonitnle
46. Acetonitriie
47. Acteonitril
48. Acteonitrile
49. Actonitrile
50. Methylcyanide
51. Methylnitrile
52. Ace-tonitrile
53. Aceto-nitrile
54. Acetonitrile-
55. Unii-z072sb282n
56. Ai3-00327
57. Acetonitrile Acs
58. Cc.equiv.n
59. Acetonitrile Lc-ms
60. Acetonitrile,homopolymer
61. Cyanomethylidyne Radical
62. Dsstox_cid_9
63. Acetonitrile Hplc Grade
64. H3ccn
65. Acetonitrile [ii]
66. Acetonitrile [mi]
67. Acetonitrile, Lcms Grade
68. Bmse000826
69. Bmse000896
70. Acetonitrile [hsdb]
71. Ec 200-835-2
72. Acetonitrile, Hplc Reagent
73. Wln: Nc1
74. Acetonitrile, >=99.5%
75. Dsstox_rid_75320
76. Dsstox_gsid_20009
77. Acetonitrile [mart.]
78. Acetonitrile With Formic Acid
79. Acetonitrile [usp-rs]
80. Acetonitrile, Puriss., 95%
81. Chembl45211
82. Acetonitrile, For Chromatography
83. Dtxsid7020009
84. Acetonitrile Uv/hplc Acs Grade
85. Acetonitrile, Analytical Standard
86. Acetonitrile For Preparative Hplc
87. Acetonitrile, Ar, >=99.5%
88. Acetonitrile, Environmental Grade
89. Nsc7593
90. Acetonitrile, Anhydrous, 99.8%
91. Acetonitrile, >=99.5% (gc)
92. Acetonitrile, Hplc Gradient Grade
93. Amidite Diluent, For Dna Synthesis
94. Str02933
95. Acetonitrile, Far Uv/gradient Grade
96. Tox21_202481
97. Acetonitrile, Hplc Grade (far Uv)
98. Acetonitrile, P.a., Dry, 99.9%
99. Acetonitrile, Reagentplus(r), 99%
100. C1151
101. Stl283937
102. Acetonitrile, Spectrophotometric Grade
103. Acetonitrile, >=99.8%, For Hplc
104. Acetonitrile, For Hplc, >=99.9%
105. Akos000269067
106. Acetonitrile, Hplc Plus, >=99.9%
107. Na 1648
108. Un 1648
109. Acetonitrile, >=99.5%, Acs Reagent
110. Acetonitrile, Acs Reagent, >=99.5%
111. Acetonitrile, Aldrasorb(tm), 99.8%
112. Acetonitrile, Purum, >=99.0% (gc)
113. Cas-75-05-8
114. Acetonitrile (for Hplc) Isocratic Grade
115. Acetonitrile, Hplc Grade, >=99.93%
116. Ncgc00091552-01
117. Ncgc00260030-01
118. Acetonitrile 1000 Microg/ml In Methanol
119. Acetonitrile, Purification Grade, 99.8%
120. Ultrapure Acetonitrile, For Dna Synthesis
121. Acetonitrile With 0.1% Formic Acid (v/v)
122. Acetonitrile, Biotech. Grade, >=99.93%
123. Acetonitrile, P.a., Acs Reagent, 99.8%
124. Acetonitrile, Saj First Grade, >=99.0%
125. A0060
126. A0293
127. A0793
128. Acetonitrile, Jis Special Grade, >=99.5%
129. Ft-0621807
130. Ft-0621808
131. Acetonitrile, Anhydrous, Zero2(tm), 99.8%
132. Acetonitrile, For Hplc-gc, >=99.8% (gc)
133. Acetonitrile, For Uhplc, For Mass Spectrometry
134. Acetonitrile, Supergradient Hplc Grade (far Uv)
135. Acetonitrile, Spectrophotometric Grade, >=99.5%
136. Q408047
137. Acetonitrile, For Hplc, For Uv, >=99.9% (gc)
138. Acetonitrile, Puriss. P.a., Acs Reagent, 99.8%
139. J-008497
140. Acetonitrile, For Preparative Hplc, >=99.8% (gc)
141. Acetonitrile, For Synthesis Of Dna, >=99.9% (gc)
142. Acetonitrile, Hplc Plus, >=99.9%, Poly-coated Bottles
143. Acetonitrile Solution, Contains 0.035 % (v/v) Acetic Acid
144. Acetonitrile Solution, Contains 0.05 % (v/v) Acetic Acid
145. Acetonitrile Solution, Contains 0.1 % (v/v) Acetic Acid
146. Acetonitrile, Electronic Grade, 99.999% Trace Metals Basis
147. Acetonitrile, For Hplc, Gradient Grade, >=99.9% (gc)
148. Acetonitrile, For Hplc, Gradient Grade, >=99.90% (gc)
149. Acetonitrile, Puriss. P.a., Acs Reagent, >=99.5% (gc)
150. Acetonitrile Solution, Contains 0.1 % (v/v) Ammonium Hydroxide
151. Acetonitrile With 0.1% Ammonium Acetate, Tested For Uhplc-ms
152. Acetonitrile, For Protein Sequence Analysis, >=99.8% (gc)
153. Acetonitrile, For Residue Analysis, Suitable For 5000 Per Jis
154. Acetonitrile, Vetec(tm) Reagent Grade, Anhydrous, >=99.8%
155. Acetonitrile Solution, ~20% In H2o, For Protein Sequence Analysis
156. Acetonitrile Solution, Contains 0.05 % (v/v) Trifluoroacetic Acid
157. Acetonitrile Solution, Contains 0.1 % (v/v) Formic Acid, For Hplc
158. Acetonitrile, Preparateur, >=99.9% (gc), Customized Plastic Drum
159. Acetonitrile Solution, Contains 0.1 % (v/v) Trifluoroacetic Acid, For Hplc
160. Acetonitrile Solution, For Hplc, Acetonitrile:water 56 : 44% (w/w)
161. Acetonitrile, >=99.8%, For Residue Analysis, Suitable For 1000 Per Jis
162. Acetonitrile, >=99.8%, For Residue Analysis, Suitable For 300 Per Jis
163. Acetonitrile, Puriss. P.a., Acs Reagent, Reag. Ph. Eur., >=99.5% (gc)
164. Acetonitrile Solution, Contains 0.035 % (v/v) Trifluoroacetic Acid, For Hplc
165. Acetonitrile Solution, Contains 0.1 % (v/v) Formic Acid, For Uhplc, For Mass Spectrometry
166. Acetonitrile Solution, Contains 0.10 % (v/v) Trifluoroacetic Acid, 10.0 % (v/v) Water
167. Acetonitrile Solution, Contains 10.0% Acetone, 40.0% 2-propanol, 0.05% Formic Acid
168. Acetonitrile, Configured For Perkinelmer 8900, Configured For Polygen, For Dna Synthesis
169. Acetonitrile, Pharmaceutical Secondary Standard; Certified Reference Material
170. Acetonitrile, Preparateur, >=99.9% (gc), One-time Steel-plastic (sp) Drum
171. Alcohol Determination - Acetonitrile, United States Pharmacopeia (usp) Reference Standard
172. Acetonitrile Solution, Contains 5 % (v/v) Water, 0.05 % (w/v) Ammonium Formate, 0.1 % (v/v) Formic Acid, For Hplc
173. Acetonitrile Solution, For Hplc, Acetonitrile:water 5:95% (v/v), 10 Mm Ammoniumbicarbonate, Ph 10,0
174. Acetonitrile Solution, Nmr Reference Standard, 0.23 Wt. % In D2o (99.9 Atom % D), Water 0.05 Wt. %, Nmr Tube Size 6.5 Mm X 8 In.
175. Residual Solvent - Acetonitrile, Pharmaceutical Secondary Standard; Certified Reference Material
176. Residual Solvent Class 2 - Acetonitrile, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 41.05 g/mol |
|---|---|
| Molecular Formula | C2H3N |
| XLogP3 | 0 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Exact Mass | 41.026549100 g/mol |
| Monoisotopic Mass | 41.026549100 g/mol |
| Topological Polar Surface Area | 23.8 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 29.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Like hydrogen cyanide (HCN), acetonitrile (ACN) is readily absorbed from the lungs and gastrointestinal tract, and is distributed throughout the body in both humans and laboratory animals. In a group of male and female test subjects, 74% of inhaled ACN was absorbed when cigarette smoke was held in the mouth for 2 seconds (and not inhaled), and 91% was absorbed when smoke was inhaled. Autopsy of an individual who died 2 days following inhalation of ACN vapors showed that cyanide reaches the spleen, lungs, and kidneys, but was not detected in the liver
U.S. Environmental Protection Agency's Integrated Risk Information System (IRIS).Toxicological Review of Acetonitrile (75-05-8). Available from, as of July 28, 2008: https://cfpub.epa.gov/ncea/iris/index.cfm
The kinetics of distribution were studied in mice following intravenous dosing. After 5 minutes, the highest levels of radioactivity were found in the liver and kidney with levels declining with time. At 24 and 48 hours, radioactivity was found highest in the gastrointestine, thymus, liver, and testes. Covalent binding studies showed approximately one-half of the radioactivity in the liver bound to macromolecular fractions. The radioactivity in other organs was primarily in the lipid fractions. Acetonitrile was shown to be converted to cyanide by rat nasal and liver tissues with the maximum rate being ten times higher per gram of protein in the nasal tissue than in any other tissue monitored.
American Conference of Governmental Industrial Hygienists. Documentation of the TLV's and BEI's with Other World Wide Occupational Exposure Values. CD-ROM Cincinnati, OH 45240-1634 2007.
Whole body autoradiography in male mice injected intravenously with acetonitrile (ACN) radiolabeled with (14)C in the methyl group indicated that radioactivity was widely distributed throughout the body (e.g., liver, thymus, and reproductive organs). Interestingly, nonvolatile radioactivity was also observed in nasal secretions, mouth cavity, esophagus, and stomach contents. One could infer from these observations that ACN could also distribute to the stomach upon inhalation exposure.
U.S. Environmental Protection Agency's Integrated Risk Information System (IRIS).Toxicological Review of Acetonitrile (75-05-8). Available from, as of July 28, 2008: https://cfpub.epa.gov/ncea/iris/index.cfm
Absorption of acetonitrile (ACN) is rapid in beagle dogs exposed to 16,000 ppm ACN (26,880 mg/cu m) vapors for 4 hours, based on blood cyanide concentrations peaking and reaching steady-state concentrations of 305-433 ug/100 mL after approximately 3 hours.
U.S. Environmental Protection Agency's Integrated Risk Information System (IRIS).Toxicological Review of Acetonitrile (75-05-8). Available from, as of July 28, 2008: https://cfpub.epa.gov/ncea/iris/index.cfm
For more Absorption, Distribution and Excretion (Complete) data for ACETONITRILE (6 total), please visit the HSDB record page.
Inorganic cyanide has long been known to react with trivalent iron of cytochrome oxidase in mitochondria and block the reduction of oxygen needed for cellular respiration, thus leading to cytotoxic anoxia. The toxicity of acetonitrile (ACN) is believed to be mediated, in part, through this mechanism. ACN is metabolized to inorganic cyanide, but the conversion occurs slowly compared to other nitriles (which may explain the delay in onset of acute symptoms). /Previous studies/ suggest that the conversion to cyanide is oxygen- and NADPH-dependent, possibly mediated by P450 isozyme (2E1 or P-450j). Some /studies/ suggest that ACN produces cyanohydrin by a P450 reaction, which is then decomposed by catalase to release cyanide. Formaldehyde and formic acid are also postulated to be by-products of ACN metabolism. Cyanide can be further oxidized to thiocyanate, a less toxic compound that is excreted in urine, but one that may interfere with thyroid function. Conversion is mediated by rhodanese, a sulfurtransferase found in liver and human nasal respiratory mucosa. A minor urinary metabolite that has been detected after administration of ACN in drinking water to rats is 2-aminothiolazine-4-carboxylic acid. Cyanide also can be oxidized to cyanate ion with further oxidation to formic acid).
U.S. Environmental Protection Agency's Integrated Risk Information System (IRIS).Toxicological Review of Acetonitrile (75-05-8). Available from, as of July 28, 2008: https://cfpub.epa.gov/ncea/iris/index.cfm
/Male Wistar/ rats given 2340 or 1500 mg/kg died within 3 to 28 hr after the intraperitoneal injection, but rats given 600 mg/kg survived with no apparent symptoms. After administration of 2340 mg/kg, concentrations of acetonitrile and free and combined cyanide in various organs ranged from 900 to 1700 mg/kg, 200 to 3500 ug/kg, and 3.5 to 17 mg/kg tissue, respectively. Mean total urinary acetonitrile and free and combined cyanide (essentially all thiocyanate) excreted during the 11 days following an intraperitoneal injection of 600 mg/kg were 28, 0.2 and 12 mg, respectively. These values were equivalent to 3, 0.035 and 2.3% of the acetonitrile dose, respectively. Urinary acetonitrile was detectable for 4 days after dosing, whereas free and combined cyanide were detectable until 11 days, at which time the animals were sacrificed.
IPCS/WHO; Environmental Health Criteria 154: Acetonitrile (1993). Available from, as of August 1, 2008: https://www.inchem.org/documents/ehc/ehc/ehc154.htm
Thiocyanate was measured as index of cyanide ion release in urine of rats given equimolar doses of nitriles. More thiocyanate was excreted after oral administration than after ip administration. Oral administration of acetonitrile yielded 37% of dose as thiocyanate (SCN-), whereas after ip injection, 4.5% of dose was excreted as thiocyanate. /nitriles/
PMID:6128199 SILVER EH ET AL; DRUG METAB DISPOS 10 (5): 495-8 (1982)
When rat liver microsomes were incubated with glycolonitrile or acetonitrile, cyanide was liberated without the formation of formaldehyde. Based on the amount of cytochrome P450 in the microsomal preparation and the rates of cyanide formed, the action of an enzyme system was postulated in the metabolism of both compounds.
PMID:3801055 Freeman JJ, Hayes EP; Biochem Pharmacol 36 (1): 184-7 (1987)
For more Metabolism/Metabolites (Complete) data for ACETONITRILE (7 total), please visit the HSDB record page.
Whole body: 15-32 hours; [TDR, p. 23]
TDR - Ryan RP, Terry CE, Leffingwell SS (eds). Toxicology Desk Reference: The Toxic Exposure and Medical Monitoring Index, 5th Ed. Washington DC: Taylor & Francis, 1999., p. 23
A case of suicidal oral acetonitrile ingestion in a previously healthy 30-year-old man /was reported/. He ingested about 5 mL (64 mg/kg) of acetonitrile (98%) and, 30 min later, about 1 mL of ammonia and vomited once. ...Half-lives were calculated for acetonitrile and cyanide and found to be 32 and 15 hr, respectively.
IPCS/WHO; Environmental Health Criteria 154: Acetonitrile (1993). Available from, as of August 1, 2008: https://www.inchem.org/documents/ehc/ehc/ehc154.htm
Aliphatic nitriles (including acetonitrile) possess little if any acute toxicity in absence of normal hepatic function & are activated by hepatic mechanisms to release cyanide which accounts for major acute toxic effects.
PMID:6267734 WILLHITE CC, SMITH RP; TOXICOL APPL PHARMACOL 59 (3): 589-602 (1981)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

ABOUT THIS PAGE
17
PharmaCompass offers a list of Acetonitrile API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Acetonitrile manufacturer or Acetonitrile supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Acetonitrile manufacturer or Acetonitrile supplier.
PharmaCompass also assists you with knowing the Acetonitrile API Price utilized in the formulation of products. Acetonitrile API Price is not always fixed or binding as the Acetonitrile Price is obtained through a variety of data sources. The Acetonitrile Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 00683_FLUKA manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 00683_FLUKA, including repackagers and relabelers. The FDA regulates 00683_FLUKA manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 00683_FLUKA API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 00683_FLUKA manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A 00683_FLUKA supplier is an individual or a company that provides 00683_FLUKA active pharmaceutical ingredient (API) or 00683_FLUKA finished formulations upon request. The 00683_FLUKA suppliers may include 00683_FLUKA API manufacturers, exporters, distributors and traders.
click here to find a list of 00683_FLUKA suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A 00683_FLUKA DMF (Drug Master File) is a document detailing the whole manufacturing process of 00683_FLUKA active pharmaceutical ingredient (API) in detail. Different forms of 00683_FLUKA DMFs exist exist since differing nations have different regulations, such as 00683_FLUKA USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 00683_FLUKA DMF submitted to regulatory agencies in the US is known as a USDMF. 00683_FLUKA USDMF includes data on 00683_FLUKA's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 00683_FLUKA USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 00683_FLUKA suppliers with USDMF on PharmaCompass.
00683_FLUKA Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 00683_FLUKA GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 00683_FLUKA GMP manufacturer or 00683_FLUKA GMP API supplier for your needs.
A 00683_FLUKA CoA (Certificate of Analysis) is a formal document that attests to 00683_FLUKA's compliance with 00683_FLUKA specifications and serves as a tool for batch-level quality control.
00683_FLUKA CoA mostly includes findings from lab analyses of a specific batch. For each 00683_FLUKA CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
00683_FLUKA may be tested according to a variety of international standards, such as European Pharmacopoeia (00683_FLUKA EP), 00683_FLUKA JP (Japanese Pharmacopeia) and the US Pharmacopoeia (00683_FLUKA USP).

