
Synopsis
Synopsis
0
KDMF
0
VMF
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
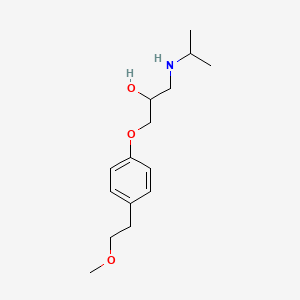

1. Beloc Duriles
2. Beloc-duriles
3. Betaloc
4. Betaloc Astra
5. Betaloc-astra
6. Betalok
7. Cgp 2175
8. Cgp-2175
9. Cgp2175
10. H 93 26
11. H 93-26
12. H 9326
13. Lopressor
14. Metoprolol Cr Xl
15. Metoprolol Cr-xl
16. Metoprolol Succinate
17. Metoprolol Tartrate
18. Seloken
19. Spesicor
20. Spesikor
21. Toprol
22. Toprol Xl
23. Toprol-xl
1. 51384-51-1
2. (rs)-metoprolol
3. Beatrolol
4. Dl-metoprolol
5. Spesicor
6. 37350-58-6
7. Seroken
8. Toprol
9. Metohexal
10. Preblok
11. Meijoprolol
12. Metoprololum
13. Presolol
14. Selo-zok
15. Loresor
16. Toprol-xl
17. 1-(isopropylamino)-3-(4-(2-methoxyethyl)phenoxy)propan-2-ol
18. Cgp 2175
19. Cgp-2175
20. 1-[4-(2-methoxyethyl)phenoxy]-3-(propan-2-ylamino)propan-2-ol
21. 1-(isopropylamino)-3-[4-(2-methoxyethyl)phenoxy]propan-2-ol
22. 1-[4-(2-methoxyethyl)phenoxy]-3-propan-2-ylamino-propan-2-ol
23. (+/-)-metoprolol
24. Metoprolol Slow Release
25. Chembl13
26. Geb06nhm23
27. Chebi:6904
28. Betalok
29. Spesikor
30. 51384-51-1 (free Base)
31. 2-propanol, 1-(4-(2-methoxyethyl)phenoxy)-3-((1-methylethyl)amino)-
32. Lopresoretic
33. 2-propanol, 1-[4-(2-methoxyethyl)phenoxy]-3-[(1-methylethyl)amino]-
34. H-93/26
35. Metoprololum [inn-latin]
36. Metoprolol [usan:inn:ban]
37. Neobloc
38. Metrol
39. {2-hydroxy-3-[4-(2-methoxyethyl)phenoxy]propyl}(propan-2-yl)amine
40. 1-[(1-methylethyl)amino]-3-({4-[2-(methyloxy)ethyl]phenyl}oxy)propan-2-ol
41. 2-propanol, 1-[4-(2-methoxyethyl)phenoxy]-3-[(1-methylethyl)amino]-, (.+/-.)-
42. Lopressidone
43. Ccris 4198
44. Hsdb 6531
45. Cgp2175
46. Metoprolol (usan/inn)
47. Unii-geb06nhm23
48. 1-(isopropylamino)-3-[4-(2-methoxyethyl)phenoxy]-2-propanol
49. 1-(4-(2-methoxyethyl)phenoxy)-3-(propan-2-ylamino)propan-2-ol
50. Metoprolol Tartarate
51. Einecs 257-166-4
52. Lopresol (salt/mix)
53. Metoprolol [mi]
54. Lopresoretic (salt/mix)
55. Metoprolol [inn]
56. (.+/-.)-metoprolol
57. Metoprolol [hsdb]
58. Metoprolol [usan]
59. Dsstox_cid_3309
60. Metoprolol [vandf]
61. Metoprolol [mart.]
62. Schembl4093
63. Metoprolol [who-dd]
64. Dsstox_gsid_23309
65. Gtpl553
66. H 93/26 (salt/mix)
67. Dtxsid2023309
68. Bdbm25756
69. Metoprolol, (+/-)-
70. Hms2090b15
71. Hms3886o04
72. Bca38451
73. Bcp09038
74. Gp-2175e
75. Tox21_303972
76. 1-[4-(2-methoxyethyl)phenoxy]-3-[(propan-2-yl)amino]propan-2-ol
77. Mfcd00599534
78. S5430
79. Stl301858
80. Akos005927923
81. Bg-0006
82. Ccg-204813
83. Cs-3159
84. Db00264
85. Sdccgsbi-0050706.p005
86. Ncgc00021148-03
87. Ncgc00021148-04
88. Ncgc00021148-05
89. Ncgc00021148-21
90. Ncgc00356981-01
91. 54163-88-1
92. Ac-19022
93. Hy-17503
94. Sbi-0050706.p004
95. 3-bromo-4,5-dihydroxybenzylalcohol
96. Cas-51384-51-1
97. Db-049097
98. A2878
99. Ab00053499
100. Ft-0602794
101. Ft-0672382
102. Ft-0672383
103. Ft-0672385
104. C07202
105. D02358
106. Ab00053499-21
107. Ab00053499_22
108. Ab00053499_23
109. A823609
110. A871296
111. L000669
112. Q409468
113. Q-201400
114. Sr-01000003148-2
115. Brd-a03623303-045-02-0
116. Brd-a03623303-045-05-3
117. 1-isopropylamino-3-[p-(2-methoxyethyl)phenoxy]-2-propanol
118. 1-(isopropylamino)-3-[p-(2-methoxyethyl)phenoxy]-2-propanol
119. 1-isopropylamino-3-[4-(2-methoxy-ethyl)-phenoxy]-propan-2-ol
120. (+/-)-1-(isopropylamino)-3-(p-(beta-methoxyethyl)phenoxy)-2-propanol
121. 1-(4-(2-methoxyethyl)-phenoxy)-3-((1-methylethyl)amino)-2-propanol
122. 1-(4-(2-methoxyethyl)phenoxy)-3-((1-methylethyl)amino)-2-propanol
123. (2r,3r)-2,3-dihydroxysuccinic Acid;1-(isopropylamino)-3-[4-(2-methoxyethyl)phenoxy]propan-2-ol
| Molecular Weight | 267.36 g/mol |
|---|---|
| Molecular Formula | C15H25NO3 |
| XLogP3 | 1.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 9 |
| Exact Mass | 267.18344366 g/mol |
| Monoisotopic Mass | 267.18344366 g/mol |
| Topological Polar Surface Area | 50.7 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 215 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 6 | |
|---|---|
| Drug Name | Dutoprol |
| PubMed Health | Metoprolol/Hydrochlorothiazide (By mouth) |
| Drug Classes | Beta-Adrenergic Blocker, Cardioselective/Thiazide Combination, Beta-Adrenergic Blocker/Thiazide Combination |
| Active Ingredient | Hydrochlorothiazide; metoprolol succinate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 25mg tartrate; eq 50mg tartrate; eq 100mg tartrate; 12.5mg |
| Market Status | Prescription |
| Company | Covis Pharma Sarl |
| 2 of 6 | |
|---|---|
| Drug Name | Lopressor |
| PubMed Health | Metoprolol |
| Drug Classes | Antianginal, Antiarrhythmic, Group II, Antihypertensive, Antimigraine, Cardiovascular Agent |
| Drug Label | Lopressor, metoprolol tartrate USP, is a selective beta1-adrenoreceptor blocking agent, available as 50- and 100-mg tablets for oral administration and in 5-mL ampuls for intravenous administration. Each ampul contains a sterile solution of metoprolo... |
| Active Ingredient | Metoprolol tartrate |
| Dosage Form | Tablet; Injectable |
| Route | Injection; Oral |
| Strength | 1mg/ml; 100mg; 50mg |
| Market Status | Prescription |
| Company | Novartis; Us Pharms Holdings I |
| 3 of 6 | |
|---|---|
| Drug Name | Toprol-xl |
| Active Ingredient | Metoprolol succinate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 25mg tartrate; eq 50mg tartrate; eq 200mg tartrate; eq 100mg tartrate |
| Market Status | Prescription |
| Company | Astrazeneca |
| 4 of 6 | |
|---|---|
| Drug Name | Dutoprol |
| PubMed Health | Metoprolol/Hydrochlorothiazide (By mouth) |
| Drug Classes | Beta-Adrenergic Blocker, Cardioselective/Thiazide Combination, Beta-Adrenergic Blocker/Thiazide Combination |
| Active Ingredient | Hydrochlorothiazide; metoprolol succinate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 25mg tartrate; eq 50mg tartrate; eq 100mg tartrate; 12.5mg |
| Market Status | Prescription |
| Company | Covis Pharma Sarl |
| 5 of 6 | |
|---|---|
| Drug Name | Lopressor |
| PubMed Health | Metoprolol |
| Drug Classes | Antianginal, Antiarrhythmic, Group II, Antihypertensive, Antimigraine, Cardiovascular Agent |
| Drug Label | Lopressor, metoprolol tartrate USP, is a selective beta1-adrenoreceptor blocking agent, available as 50- and 100-mg tablets for oral administration and in 5-mL ampuls for intravenous administration. Each ampul contains a sterile solution of metoprolo... |
| Active Ingredient | Metoprolol tartrate |
| Dosage Form | Tablet; Injectable |
| Route | Injection; Oral |
| Strength | 1mg/ml; 100mg; 50mg |
| Market Status | Prescription |
| Company | Novartis; Us Pharms Holdings I |
| 6 of 6 | |
|---|---|
| Drug Name | Toprol-xl |
| Active Ingredient | Metoprolol succinate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 25mg tartrate; eq 50mg tartrate; eq 200mg tartrate; eq 100mg tartrate |
| Market Status | Prescription |
| Company | Astrazeneca |
Adrenergic beta-Antagonists; Anti-Arrhythmia Agents; Antihypertensive Agents; Sympatholytics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Metoprolol /is/ used in the treatment of mitral value prolapse syndrome. /NOT included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 551
Metoprolol ... /is/ used for thyrotoxicosis. /NOT included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 551
/Metoprolol has been used/ to control the physical manifestations of anxiety such as tachycardia and tremor. It is not particularly useful for chronic anxiety or panic attacks but is most useful for reducing anxiety and improving performance in specific stressful situations. /NOT included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 551
For more Therapeutic Uses (Complete) data for METOPROLOL (12 total), please visit the HSDB record page.
Tiredness or dizziness has occurred in about 10% of patients with hypertension or angina receiving metoprolol tartrate in clinical trials; tiredness has been reported in about 1% of patients with myocardial infarction receiving the drug. In addition, vertigo, sleep disturbances/insomnia, hallucinations, nightmares, headache, dizziness, visual disturbances, and confusion have been reported in patients with myocardial infarction receiving the drug, although a causal relationship is unclear. Somnolence or increased dreaming also has been reported with metoprolol therapy; these effects may be alleviated by avoiding late-evening dosing. rarely, impotence, nervousness, and general weakness have occurred. Depression has been reported in about 5% of patients receiving metoprolol tartrate for hypertension or angina. ... /Metoprolol tartrate/
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1766
Diarrhea has occurred in about 5% of patients receiving metoprolol tartrate in clinical trials. Other GI symptoms such as nausea, gastric pain, constipation, flatulence, digestive tract disorders, heartburn, xerostomia, and hiccups also have been reported with oral metoprolol therapy. Nausea and abdominal pain have occurred in less than 1% of patients with myocardial infarction receiving IV or oral metoprolol.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1767
In 10 healthy subjects administration of metoprolol tartrate 50 mg by mouth increased the peripheral platelet count.
Reynolds, J.E.F., Prasad, A.B. (eds.) Martindale-The Extra Pharmacopoeia. 28th ed. London: The Pharmaceutical Press, 1982., p. 1342
Peyronie's disease, tinnitus, restless legs, a polymyalgia-like syndrome, decreased libido, blurred vision, dry eyes, dry mucous membranes, agranulocytosis, and sweating have occurred rarely in patients receiving metoprolol. Pruritus, dry skin, worsening of psoriasis, and psoriasiform, maculopapular, and urticarial rash have occurred in some patients receiving metoprolol.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1767
For more Drug Warnings (Complete) data for METOPROLOL (10 total), please visit the HSDB record page.
Hypotension and nodal rhythm resulted in the survivor of a 50-g overdose. However, cardiac failure and bradycardia occurred in a fatal 7.5 g overdose.
Ellenhorn, M.J. and D.G. Barceloux. Medical Toxicology - Diagnosis and Treatment of Human Poisoning. New York, NY: Elsevier Science Publishing Co., Inc. 1988., p. 188
Metoprolol is indicated for the treatment of angina, heart failure, myocardial infarction, atrial fibrillation, atrial flutter and hypertension. Some off-label uses of metoprolol include supraventricular tachycardia and thyroid storm. All the indications of metoprolol are part of cardiovascular diseases. These conditions correspond to a number of diseases that involve the function of the heart and blood vessels. The underlying causes of these conditions are variable and can be due to genetic disposition, lifestyle decisions such as smoking, obesity, diet, and lack of exercise, and comorbidity with other conditions such as diabetes. The cardiovascular diseases are the leading cause of death on a global scale.
FDA Label
Administration of metoprolol in normal subjects is widely reported to produce a dose-dependent reduction on heart rate and cardiac output. This effect is generated due to a decreased cardiac excitability, cardiac output, and myocardial oxygen demand. In the case of arrhythmias, metoprolol produces its effect by reducing the slope of the pacemaker potential as well as suppressing the rate of atrioventricular conduction. The Metoprolol Atherosclerosis Prevention in Hypertensives (MAPHY) trial showed a significant improvement in sudden cardiac death and myocardial infarction when patients were given with metoprolol as compared with diuretics. As well, in clinical trials performed in 1990, metoprolol reduces mortality and re-infarction in 17% of the individuals when administered chronically after an episode of myocardial infarction.
Anti-Arrhythmia Agents
Agents used for the treatment or prevention of cardiac arrhythmias. They may affect the polarization-repolarization phase of the action potential, its excitability or refractoriness, or impulse conduction or membrane responsiveness within cardiac fibers. Anti-arrhythmia agents are often classed into four main groups according to their mechanism of action: sodium channel blockade, beta-adrenergic blockade, repolarization prolongation, or calcium channel blockade. (See all compounds classified as Anti-Arrhythmia Agents.)
Sympatholytics
Drugs that inhibit the actions of the sympathetic nervous system by any mechanism. The most common of these are the ADRENERGIC ANTAGONISTS and drugs that deplete norepinephrine or reduce the release of transmitters from adrenergic postganglionic terminals (see ADRENERGIC AGENTS). Drugs that act in the central nervous system to reduce sympathetic activity (e.g., centrally acting alpha-2 adrenergic agonists, see ADRENERGIC ALPHA-AGONISTS) are included here. (See all compounds classified as Sympatholytics.)
Adrenergic beta-1 Receptor Antagonists
Drugs that bind to and block the activation of ADRENERGIC BETA-1 RECEPTORS. (See all compounds classified as Adrenergic beta-1 Receptor Antagonists.)
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
C07AB02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
C07AB02
S66 | EAWAGTPS | Parent-Transformation Product Pairs from Eawag | DOI:10.5281/zenodo.3754448
C - Cardiovascular system
C07 - Beta blocking agents
C07A - Beta blocking agents
C07AB - Beta blocking agents, selective
C07AB02 - Metoprolol
Absorption
When metoprolol is administered orally, it is almost completely absorbed in the gastrointestinal tract. The maximum serum concentration is achieved 20 min after intravenous administration and 1-2 hours after oral administration. The bioavailability of metoprolol is of 100% when administered intravenously and when administered orally it presents about 50% for the tartrate derivative and 40% for the succinate derivative. The absorption of metoprolol in the form of the tartrate derivative is increased by the concomitant administration of food.
Route of Elimination
Metoprolol is mainly excreted via the kidneys. From the eliminated dose, less than 5% is recovered unchanged.
Volume of Distribution
The reported volume of distribution of metoprolol is 4.2 L/kg. Due to the characteristics of metoprolol, this molecule is able to cross the blood-brain barrier and even 78% of the administered drug can be found in cerebrospinal fluid.
Clearance
The reported clearance rate on patients with normal kidney function is 0.8 L/min. In cirrhotic patients, the clearance rate changes to 0.61 L/min.
Plasma levels following oral administration of conventional metoprolol tablets, however, approximate 50% of levels following intravenous adminsitration, indicating about 50% first-pass metabolism... Elimination is mainly by biotransformation in the liver.
Medical Economics Co; Physicians Desk Reference 56th ed p. 651 (2002)
Metoprolol tartrate is rapidly and almost completely absorbed from the GI tract; absorption of a single oral dose of 20-100 mg is complete in 2.5-3 hours. After an oral dose, about 50% of the drug administered as conventional tablets appears to undergo first-pass metabolism in the liver. Bioavailability of orally administered metoprolol tartrate increases with increased doses, indicating a possible saturable disposition process of low capacity such as tissue binding in the liver. Steady-state oral bioavailability of extended-release tablets of metoprolol succinate given once daily at dosages equivalent to 50-400 mg of metoprolol tartrate is about 77% of that of conventional tablets at corresponding dosages given once daily or in divided doses. Food does not appear to affect bioavailability of metoprolol succinate extended-release tablets. Following a single oral dose as conventional tablets, metoprolol appears in the plasma within 10 minutes and peak plasma concentrations are reached in about 90 minutes. When metoprolol tartrate conventional tablets are administered with food rather than on an empty stomach, peak plasma concentrations are higher and the extent of absorption of the drug is increased. Following oral administration of metoprolol succinate as extended-release tablets, peak plasma metoprolol concentrations are aobut 25-50% of those attained after administration of metoprolol tartrate conventional tablets given once daily or in divided doses. Time to peak concentration is longer with extended-release tablets, with peak plasma coentrations being reached in about 7 hours following administration of such tablets. Plasma concentrations attained 1 hour after an oral dose are linearly related to metoprolol tartrate doses ranging from 50-400 mg as conventional tablets.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1769
Plasma metoprolol concentrations attained after iv administration of the drug are approximately 2 times those attained following oral administration. Following iv infusion of metoprolol over 10 minutes in healthy individuals, maximum beta-adrenergic blocking activity occurred at 20 minutes. In healthy individuals, a maximum reduction in exercise-induced heart rate of approximately 10 and 15% occurs following iv administration of a single 5 mg and 15 mg metoprolol dose, respectively; the effect on exercise-induced heart rate decreased linearly with time at the same rate for both doses and persisted for approximately 5 and 8 hours for the 5 mg and 15 mg doses, respectively.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1769
Elimination of metoprolol appears to follow first-order kinetics and occurs mainly in the liver; the time required for the process apparently is independent of dose and duration of therapy. In healthy individuals and hypertensive patients, the elimination half-life of both unchanged drug and metabolites is about 3-4 hours. In poor hydroxylators of the drug, the elimination half-life is prolonged to about 7.6 hours. There is more interindividual variation in elimination half-lives in geriatric patients than in young healthy individuals. The half-life of metoprolol does not increase appreciably with impaired renal function.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1770
For more Absorption, Distribution and Excretion (Complete) data for METOPROLOL (7 total), please visit the HSDB record page.
Metoprolol goes through significant first-pass hepatic metabolism which covers around 50% of the administered dose. The metabolism of metoprolol is mainly driven by the activity of CYP2D6 and to a lesser extent due to the activity of CYP3A4. The metabolism of metoprolol is mainly represented by reactions of hydroxylation and O-demethylation.
Metoprolol does not inhibit or enhance its own metabolism. Three main metabolites of the drug are formed by oxidative deamination, O-dealkylation with subsequent oxidation, and aliphatic hydroxylation; these metabolites account for 85% of the total urinary excretion of metabolites. The metabolites apparently do not have appreciable pharmacologic activity. The rate of hydroxylation, resulting in alpha-hydroxymetoprolol, is genetically determined and is subject to considerable interindividual variation. Poor hydroxylators of metoprolol have increased areas under the plasma concentration-time curves, prolonged elimination half-lives (about 7.6 hours), higher urinary concentrations of unchanged drug, and negligible urinary concentrations of alpha-hydroxymetoprolol compared with extensive hydroxylators. Beta-adrenergic blockade of exercise-induced tachycardia persists for at least 24 hours after administration of a single 200-mg oral dose of metoprolol tartrate in poor hydroxylators.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1770
Controlled studies have shown that debrisoquine oxidation phenotype is a major determinant of the metabolism, pharmacokinetics and some of the pharmacological actions of metoprolol. The poor metabolizer phenotype is associated with increased plasma drug concentrations, a prolongation of elimination half-life and more intense and sustained beta blockade. Phenotypic differences have also been observed in the pharmacokinetics of the enantiomers of metoprolol. In vivo and in vitro studies have identified some of the metabolic pathways which are subject to the defect, that is alpha-hydroxylation and O-demethylation.
PMID:2868819 Lennard MS et al; Clin Pharmacokinet 11 (1): 1-17 (1986)
Metropolol is a racemic mixture of R-and S-enantiomers, and is primarily metabolized by CYP2D6.
Medical Economics Co; Physicians Desk Reference 56th ed p. 651 (2002)
The immediate release formulations of metoprolol present a half-life of about 3-7 hours.
The plasma half-life ranges from approximately 3 to 7 hours.
Medical Economics Co; Physicians Desk Reference 56th ed p. 651 (2002)
Metoprolol is a beta-1-adrenergic receptor inhibitor specific to cardiac cells with negligible effect on beta-2 receptors. This inhibition decreases cardiac output by producing negative chronotropic and inotropic effects without presenting activity towards membrane stabilization nor intrinsic sympathomimetics.
Beta-adenoreceptor blocking property the amount of beta1 and beta2 effect depends on the cardioselectivity of the drug. Decreased automaticity. Reduced conduction velocity and increased refractoriness in accessory bundles (Wolff- Parkinson-White syndrome). /Class II- beta-Blocking Agents/
Ellenhorn, M.J. and D.G. Barceloux. Medical Toxicology - Diagnosis and Treatment of Human Poisoning. New York, NY: Elsevier Science Publishing Co., Inc. 1988., p. 172
At low doses, metoprolol is a selective inhibitor of beta 1-adrenergic receptors. Like propranolol, metoprolol inhibits response to adrenergic stimuli by competitively blocking b1-adrenergic receptors within the myocardium. Unlike propranolol, however, metoprolol blocks b2-adrenergic receptors within bronchial and vascular smooth muscle only in high doses.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1769
The precise mechanism of metoprolol's hypotensive action has not been determined. It has been postulated that beta-adrenergic blocking agents reduce blood pressure by blocking peripheral (especially cardiac) adrenergic receptors (decreasing cardiac output), by decreasing sympathetic outflow from the CNS, and/or by suppressing renin release.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1769
In the management of angina pectoris, the mechanism of action of metoprolol is thought to be blockage of catecholamine-induced increases in heart rate, velocity and extent of myocardial contraction, and blood pressure, which results in a net decrease in myocardial oxygen consumption.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 1769
For more Mechanism of Action (Complete) data for METOPROLOL (7 total), please visit the HSDB record page.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
89
PharmaCompass offers a list of Metoprolol Tartrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Metoprolol Tartrate manufacturer or Metoprolol Tartrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Metoprolol Tartrate manufacturer or Metoprolol Tartrate supplier.
PharmaCompass also assists you with knowing the Metoprolol Tartrate API Price utilized in the formulation of products. Metoprolol Tartrate API Price is not always fixed or binding as the Metoprolol Tartrate Price is obtained through a variety of data sources. The Metoprolol Tartrate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Metohexal manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Metohexal, including repackagers and relabelers. The FDA regulates Metohexal manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Metohexal API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Metohexal manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Metohexal supplier is an individual or a company that provides Metohexal active pharmaceutical ingredient (API) or Metohexal finished formulations upon request. The Metohexal suppliers may include Metohexal API manufacturers, exporters, distributors and traders.
click here to find a list of Metohexal suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Metohexal DMF (Drug Master File) is a document detailing the whole manufacturing process of Metohexal active pharmaceutical ingredient (API) in detail. Different forms of Metohexal DMFs exist exist since differing nations have different regulations, such as Metohexal USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Metohexal DMF submitted to regulatory agencies in the US is known as a USDMF. Metohexal USDMF includes data on Metohexal's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Metohexal USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Metohexal suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Metohexal Drug Master File in Japan (Metohexal JDMF) empowers Metohexal API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Metohexal JDMF during the approval evaluation for pharmaceutical products. At the time of Metohexal JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Metohexal suppliers with JDMF on PharmaCompass.
A Metohexal CEP of the European Pharmacopoeia monograph is often referred to as a Metohexal Certificate of Suitability (COS). The purpose of a Metohexal CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Metohexal EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Metohexal to their clients by showing that a Metohexal CEP has been issued for it. The manufacturer submits a Metohexal CEP (COS) as part of the market authorization procedure, and it takes on the role of a Metohexal CEP holder for the record. Additionally, the data presented in the Metohexal CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Metohexal DMF.
A Metohexal CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Metohexal CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Metohexal suppliers with CEP (COS) on PharmaCompass.
A Metohexal written confirmation (Metohexal WC) is an official document issued by a regulatory agency to a Metohexal manufacturer, verifying that the manufacturing facility of a Metohexal active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Metohexal APIs or Metohexal finished pharmaceutical products to another nation, regulatory agencies frequently require a Metohexal WC (written confirmation) as part of the regulatory process.
click here to find a list of Metohexal suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Metohexal as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Metohexal API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Metohexal as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Metohexal and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Metohexal NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Metohexal suppliers with NDC on PharmaCompass.
Metohexal Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Metohexal GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Metohexal GMP manufacturer or Metohexal GMP API supplier for your needs.
A Metohexal CoA (Certificate of Analysis) is a formal document that attests to Metohexal's compliance with Metohexal specifications and serves as a tool for batch-level quality control.
Metohexal CoA mostly includes findings from lab analyses of a specific batch. For each Metohexal CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Metohexal may be tested according to a variety of international standards, such as European Pharmacopoeia (Metohexal EP), Metohexal JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Metohexal USP).

