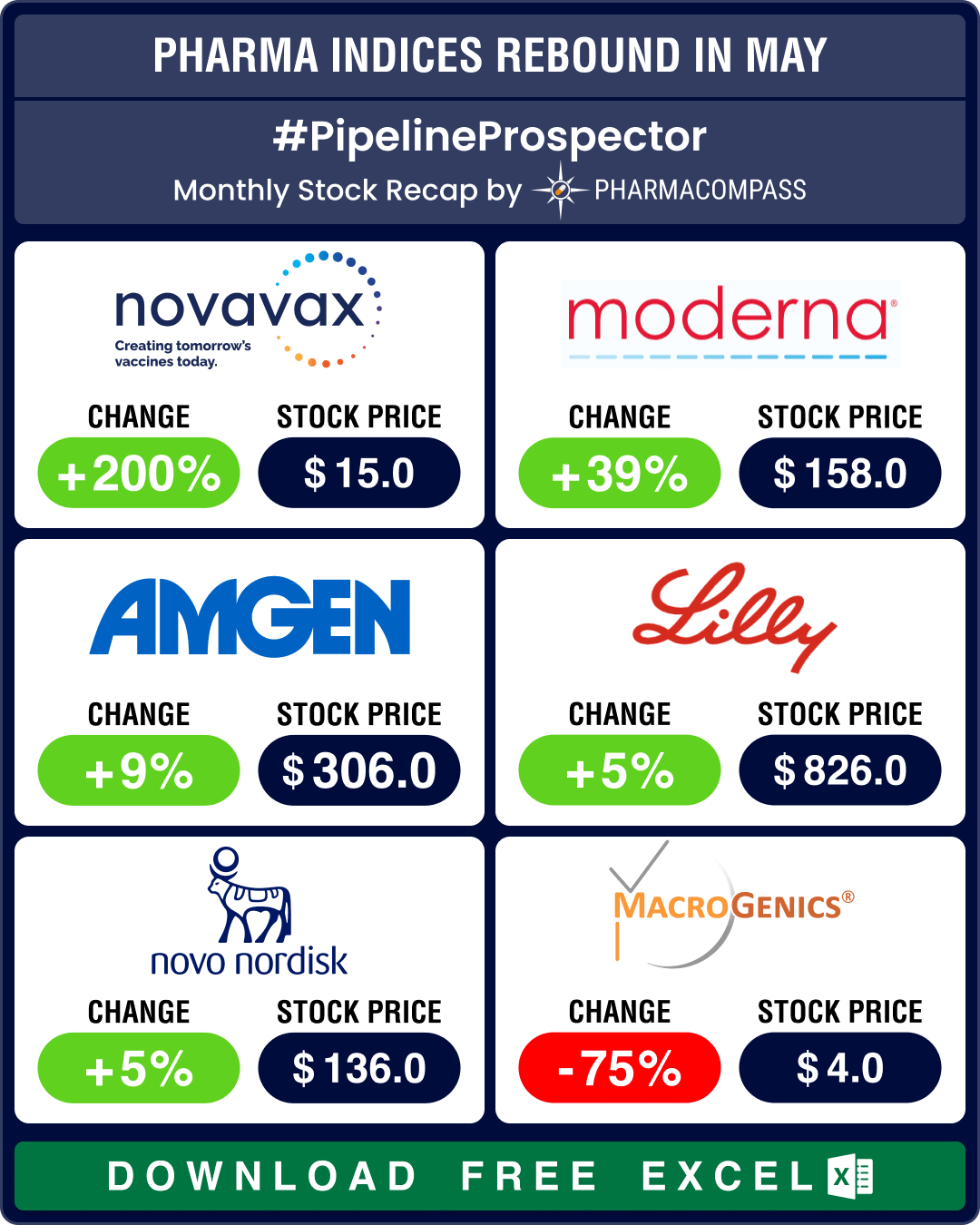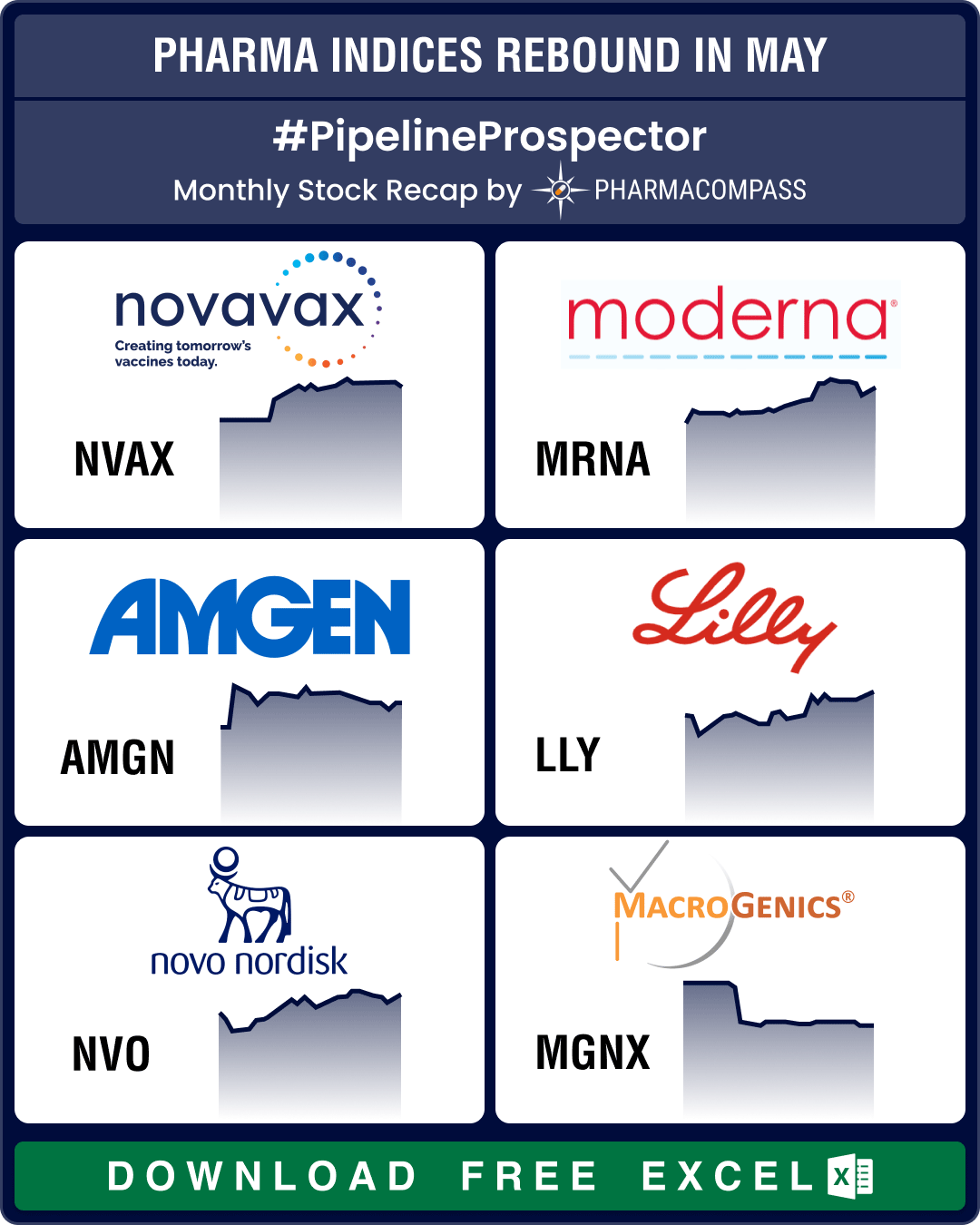
Stock Recap #PipelineProspector
Pipeline Prospector February 2026 highlights: Gilead buys Arcellx for US$ 7.8 bn; FDA okays Vanda’s psychotic pill
February
was a month of upheavals. In the US, the Supreme Court ruled that President Donald Trump
Pipeline Prospector Jan 2026 highlights: Astra, CSPC sign up to US$ 18.5 bn obesity deal; Wegovy’s pill version debuts in US
The
year 2026 began amid heightened geopolitical tensions, particularly over
Greenland. The US Pre
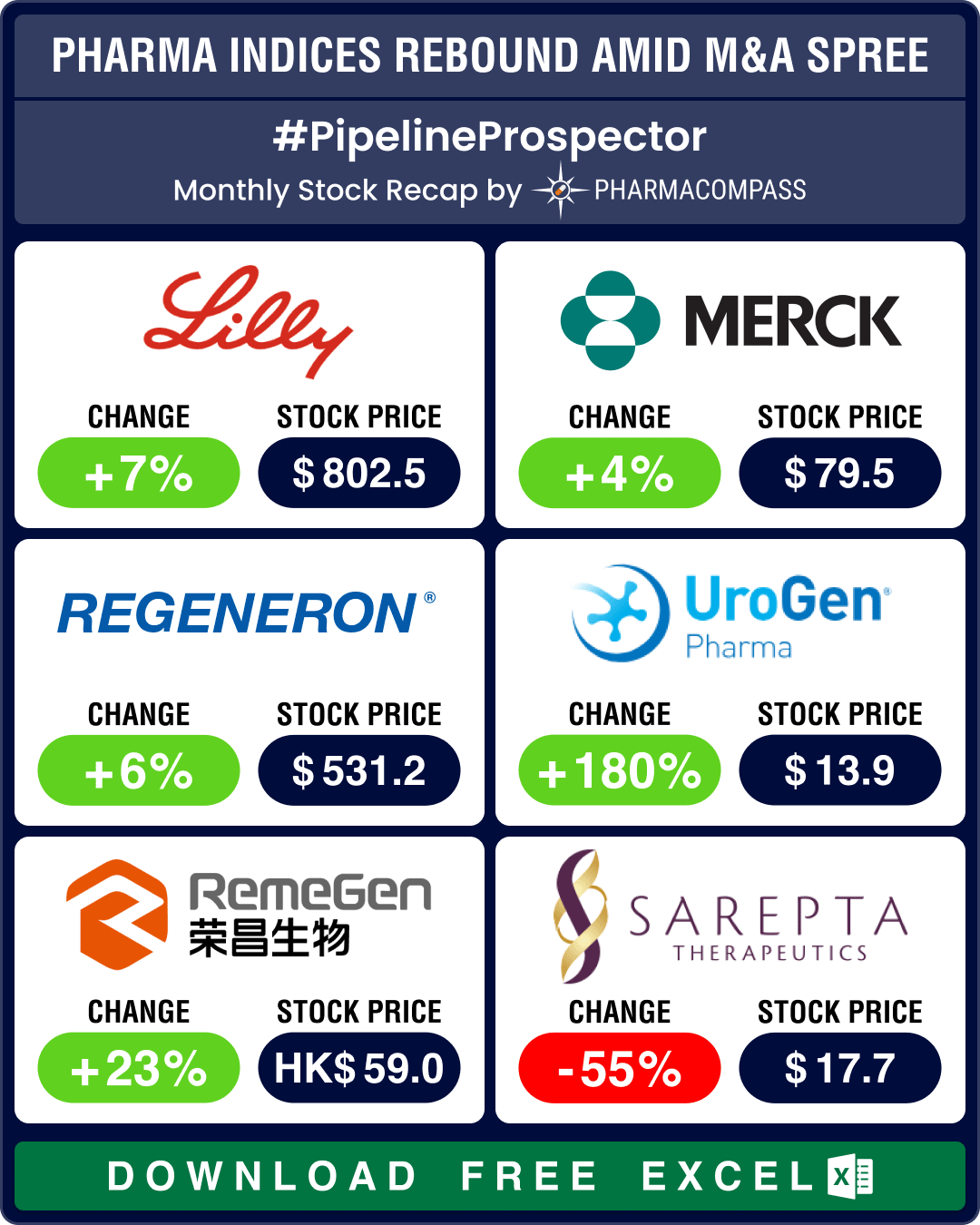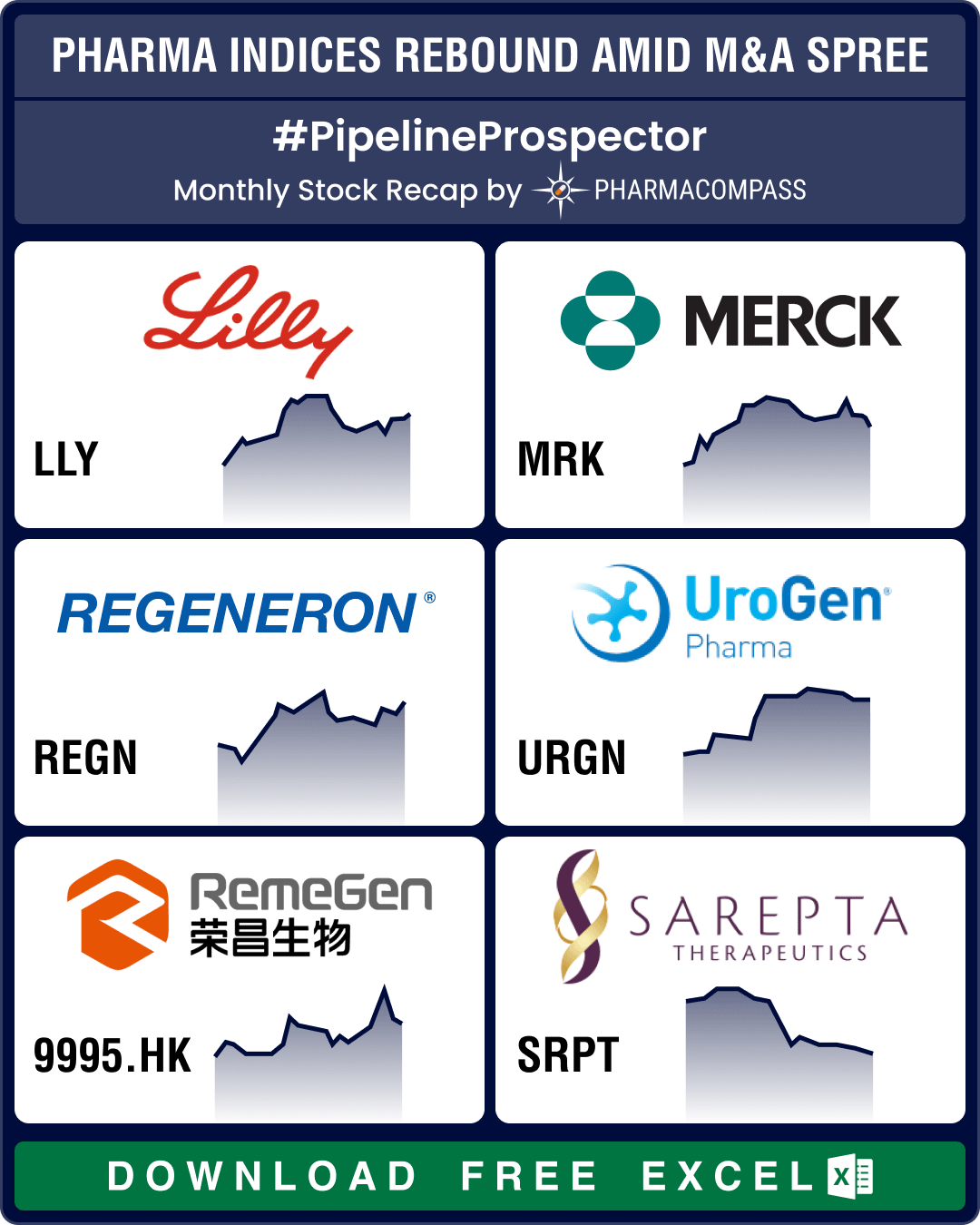
Pipeline Prospector June 2025: Sanofi, AbbVie, Eli Lilly, BioNTech lead pharma M&A spree; BMS, Astra ink major collaborations
In June, the pharmaceutical industry continued to witness more merger and acquisitions, deals, and g
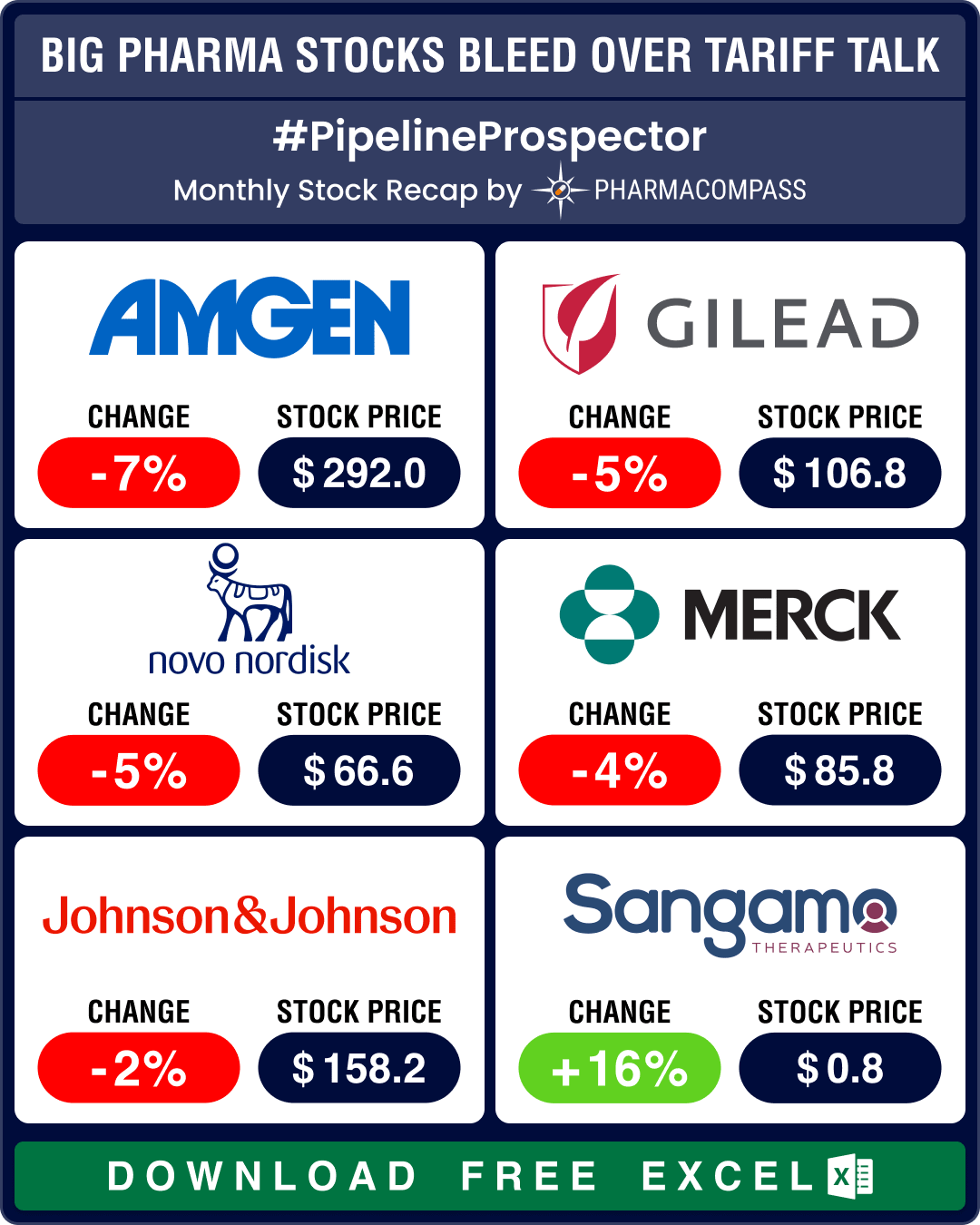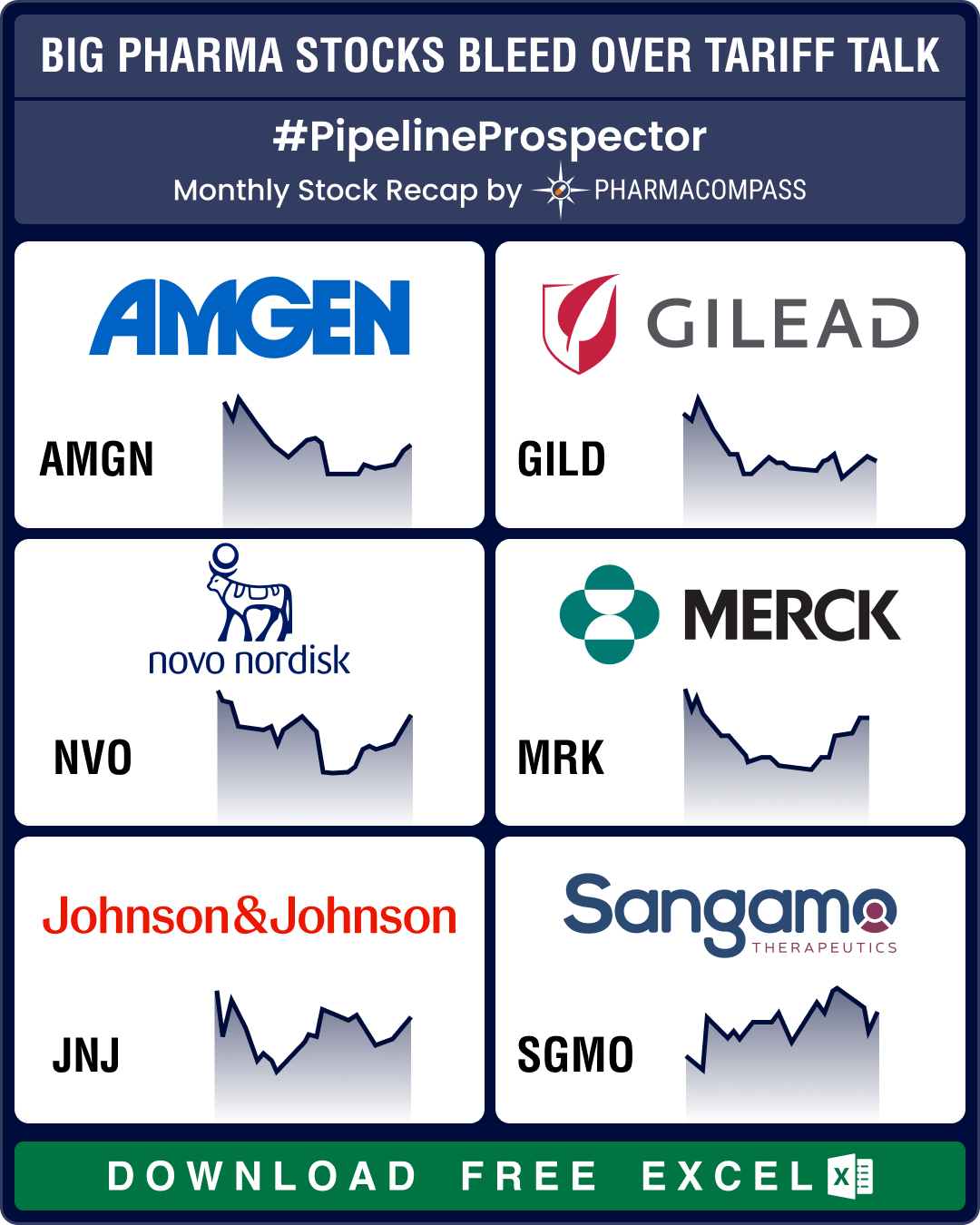
Pipeline Prospector April 2025: Merck KGaA buys SpringWorks for US$ 3.9 bn; Swiss giants lead pharma migration to US soil
April witnessed considerable volatility in pharmaceutical stocks as the US President Donald Trump&rs
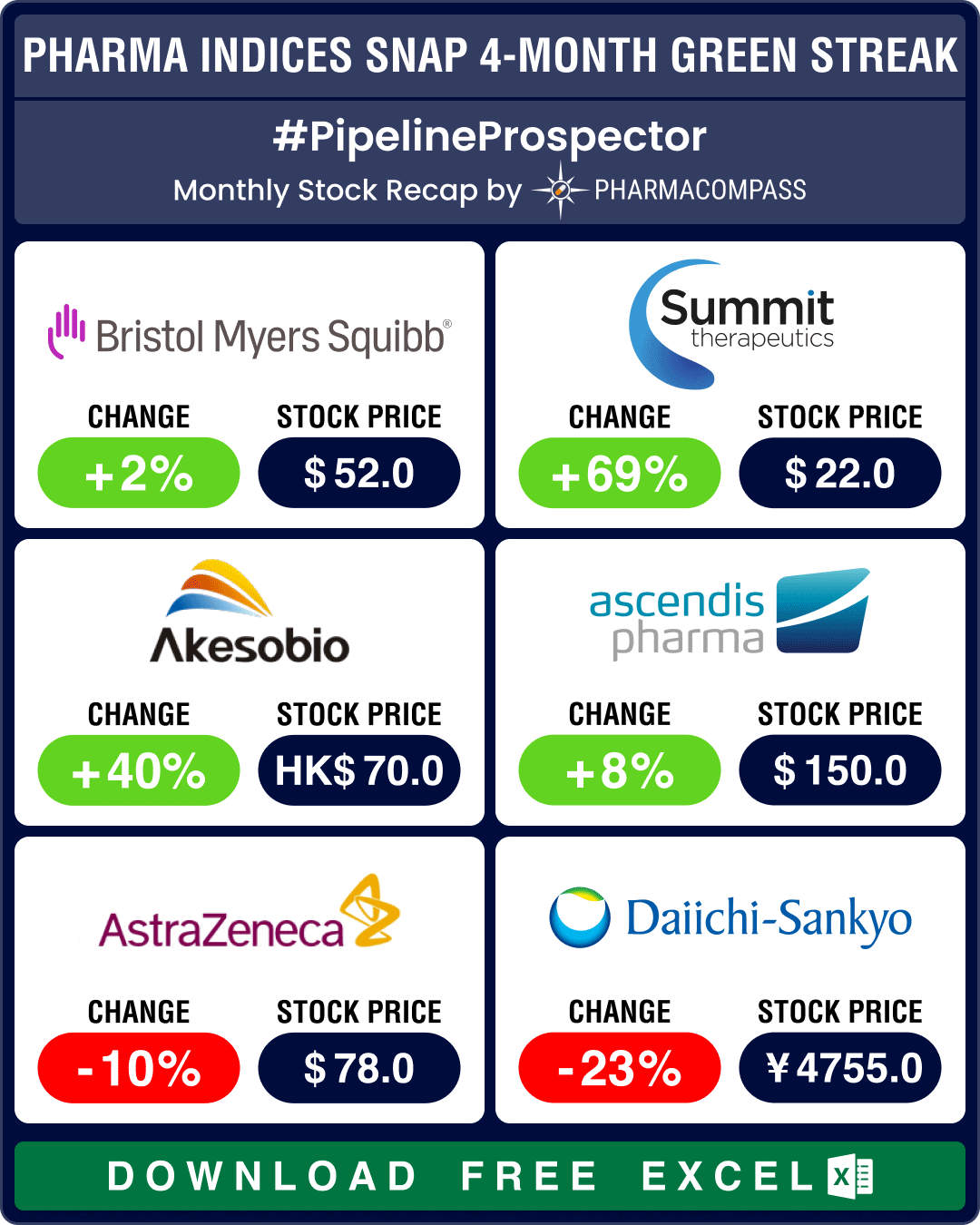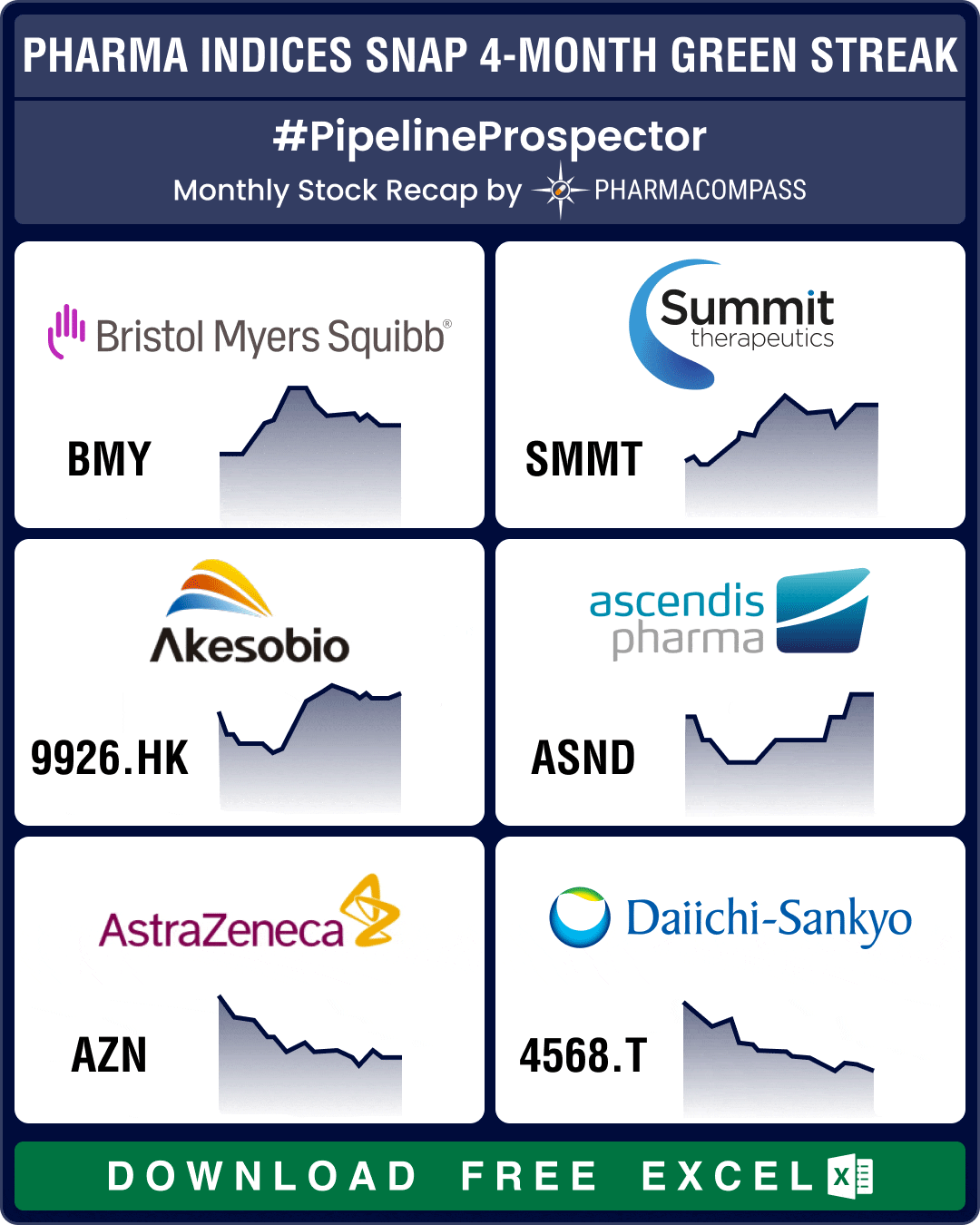
Pipeline Prospector Sept 2024: BMS wins landmark FDA approval for schizophrenia med; Sanofi’s Dupixent okayed for COPD
Pharma indices settled slightly lower in September after four months of solid gains. The three major
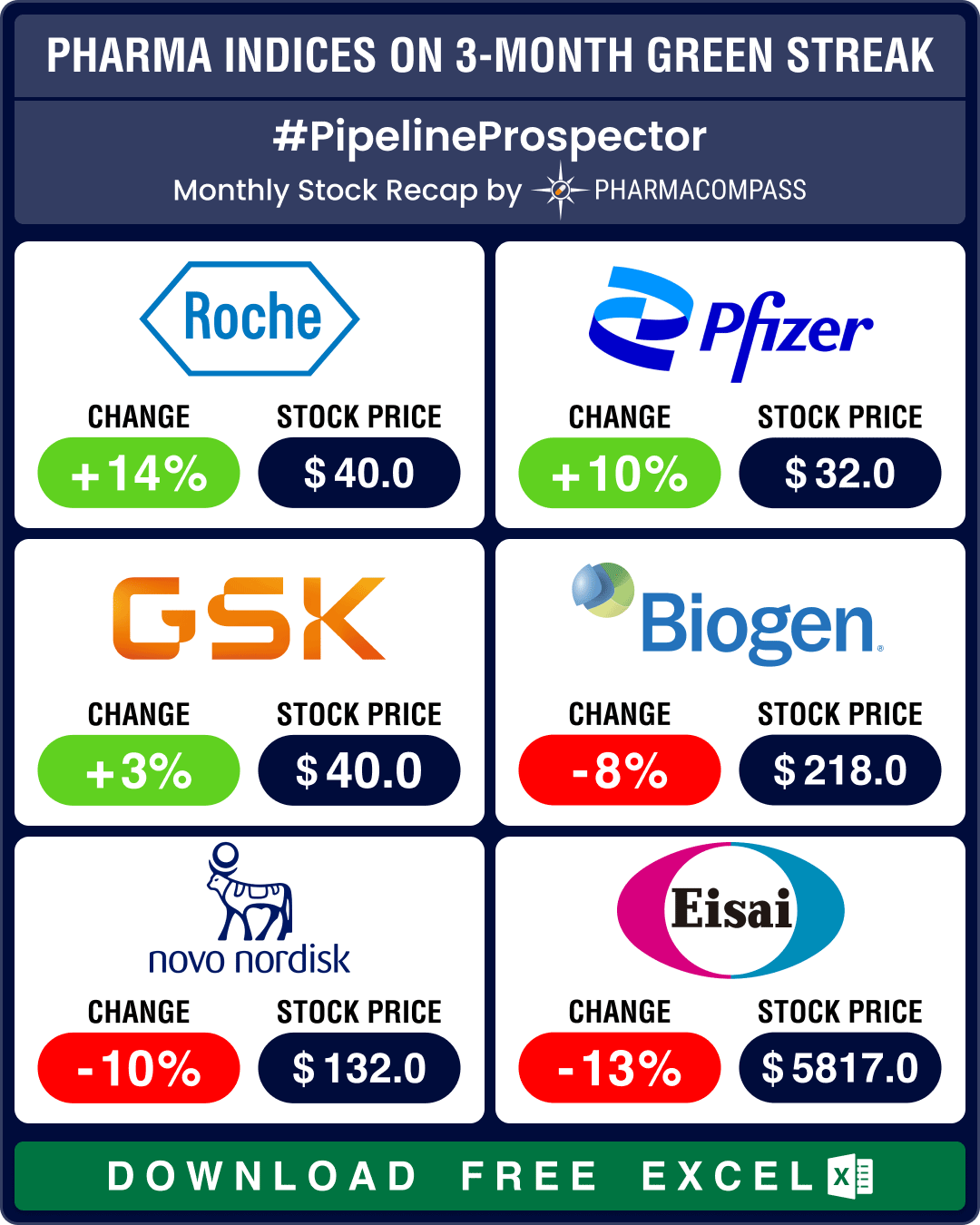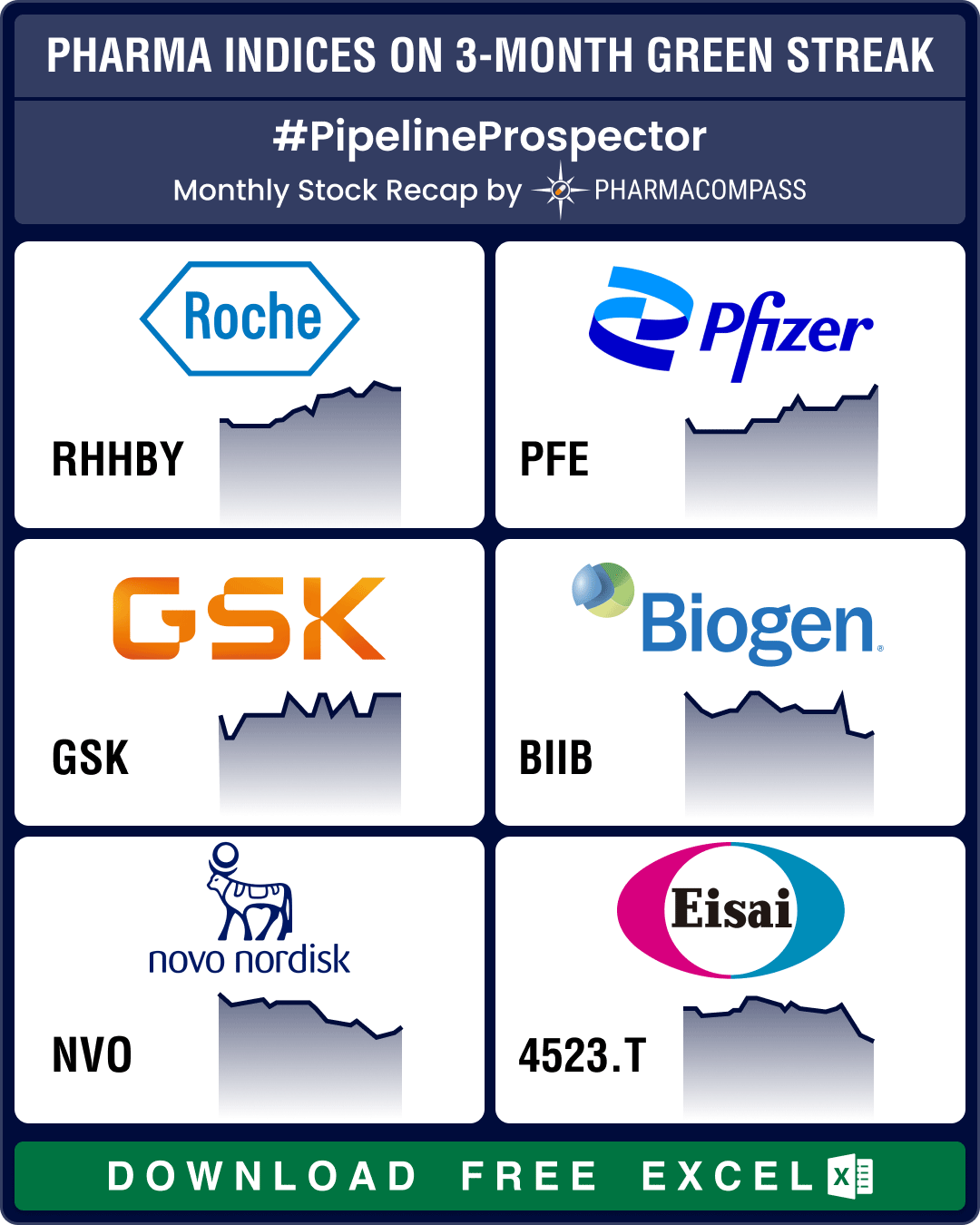
Pipeline Prospector July 2024: Indices continue to climb; Lilly buys Morphic for US$ 3.2 bn, Kisunla bags FDA nod
The biotechnology sector ended in the green for the third month in a row in July, significantly outp