NDC Code(s) : 76385-110-01, 76385-110-50, 76385-111-01, 76385-111-50, 76385-112-01, 76385-112-50, 76385-113-01, 76385-113-50
Packager : Bayshore Pharmaceuticals LLC
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| CarvedilolCarvedilol Tablet, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| CarvedilolCarvedilol Tablet, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| CarvedilolCarvedilol Tablet, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| CarvedilolCarvedilol Tablet, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| LABELER - Bayshore Pharmaceuticals LLC(968737416) |
| REGISTRANT - Beximco Pharmaceuticals USA Inc(731557430) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Beximco Pharmaceuticals Ltd | 731579053 | manufacture(76385-110, 76385-111, 76385-112, 76385-113) | |
PRINCIPAL DISPLAY PANEL
NDC 76385-110-01 Rx only
Carvedilol Tablet
Tablet USP
3.125 mg
100 Tablet
Bayshore

PRINCIPAL DISPLAY PANEL
NDC 76385-111-01 Rx only
Carvedilol Tablet
Tablet USP
6.25 mg
100 Tablet
Bayshore
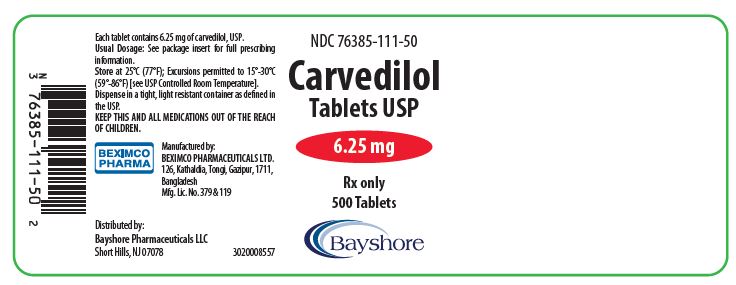
PRINCIPAL DISPLAY PANEL
NDC 76385-112-01 Rx only
Carvedilol Tablet
Tablet USP
12.5 mg
100 Tablet
Bayshore
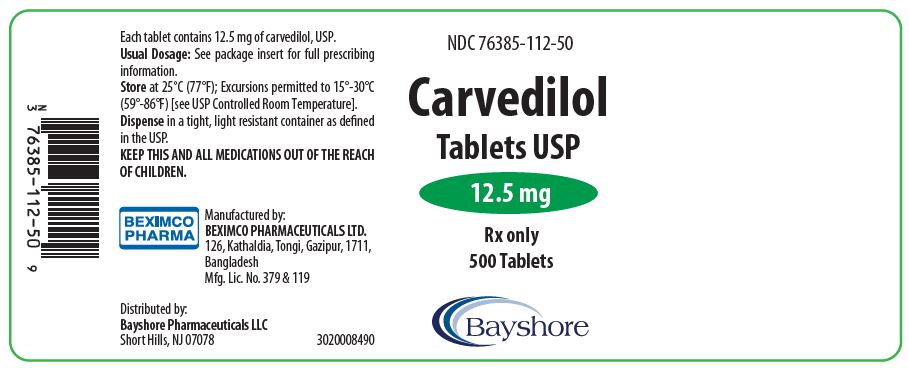
PRINCIPAL DISPLAY PANEL
NDC 76385-113-01 Rx only
Carvedilol Tablet
Tablet USP
25 mg
100 Tablet
Bayshore









