NDC Code(s) : 72485-649-71
Packager : ARMAS PHARMACEUTICALS INC.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| BromfenacBromfenac SOLUTION/ DROPS | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LABELER - ARMAS PHARMACEUTICALS INC.(098405973) |
| REGISTRANT - SENTISS AG(486920486) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Ophtapharm AG | 482198285 | manufacture(72485-649) | |
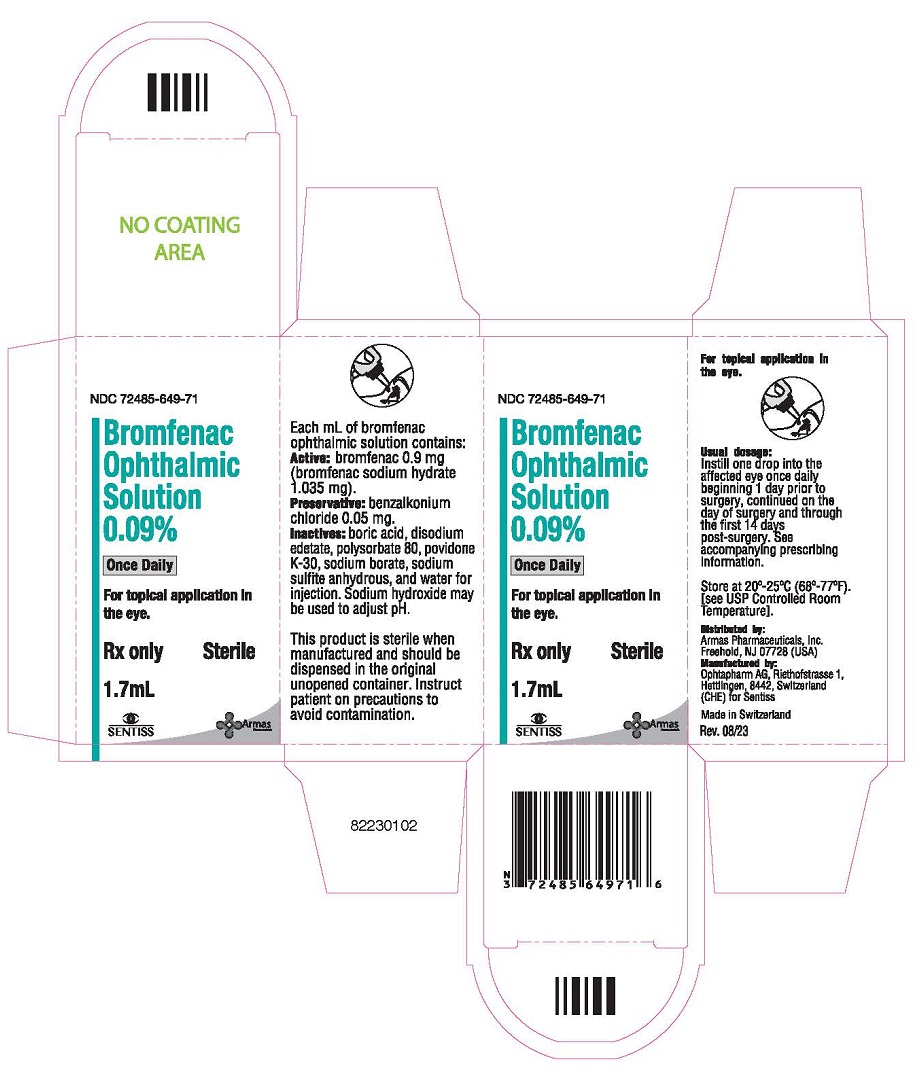
PRINCIPAL DISPLAY PANEL
NDC 72485-649-71
Bromfenac Ophthalmic Solution 0.09%
Once Daily
For topical application in the eye.
Rx only Sterile
1.7 mL