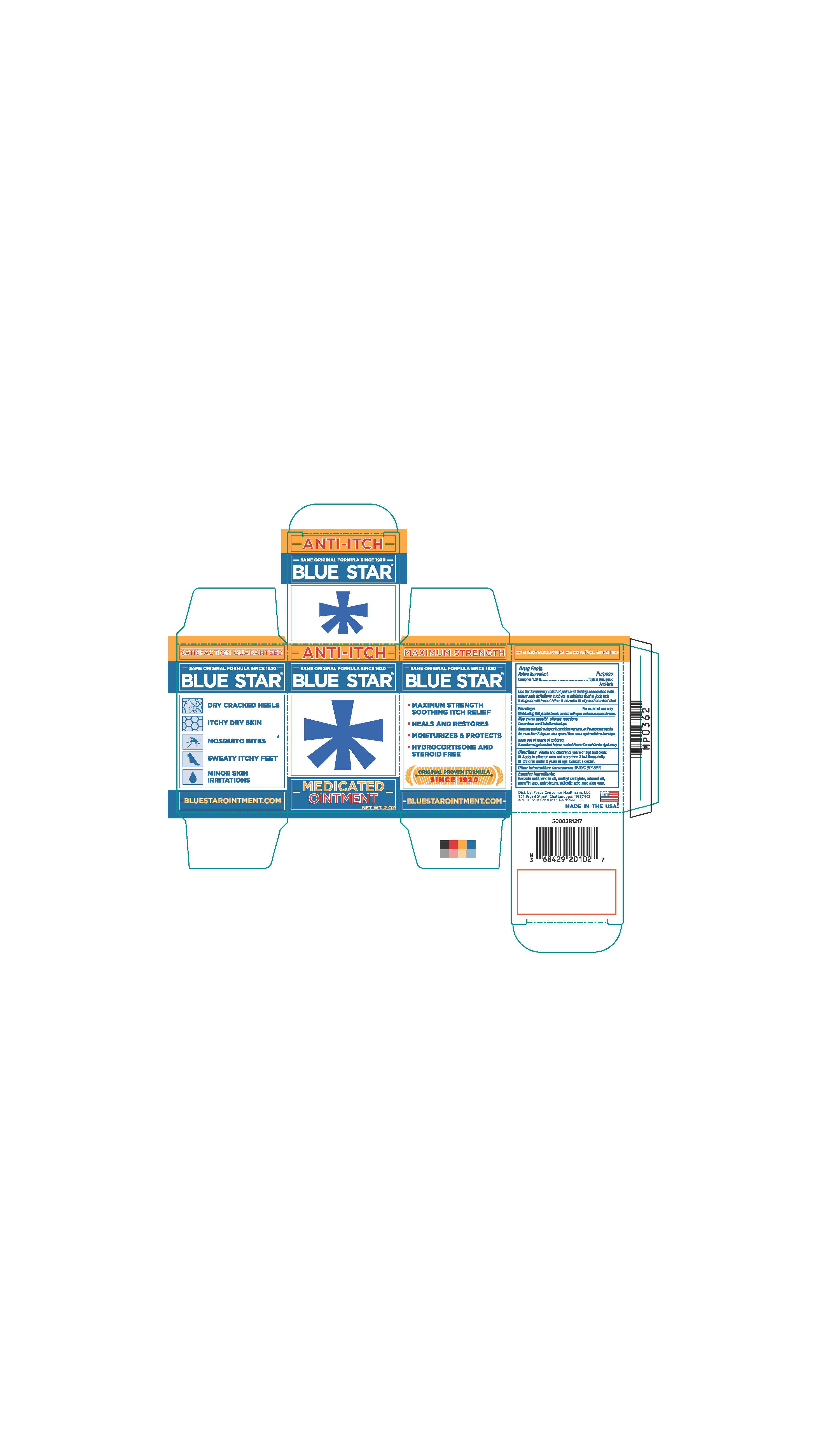
NDC Code(s) : 71687-2010-2
Packager : Focus Consumer Healthcare, LLC
Category : HUMAN OTC DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Blue Star Medicatedcamphor OINTMENT | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LABELER - Focus Consumer Healthcare, LLC(080743737) |
PRINCIPAL DISPLAY PANEL