NDC Code(s) : 71288-007-75
Packager : Meitheal Pharmaceuticals Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Ampicillin and SulbactamAmpicillin Sodium and Sulbactam Sodium INJECTION, POWDER, FOR SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LABELER - Meitheal Pharmaceuticals Inc.(080548348) |
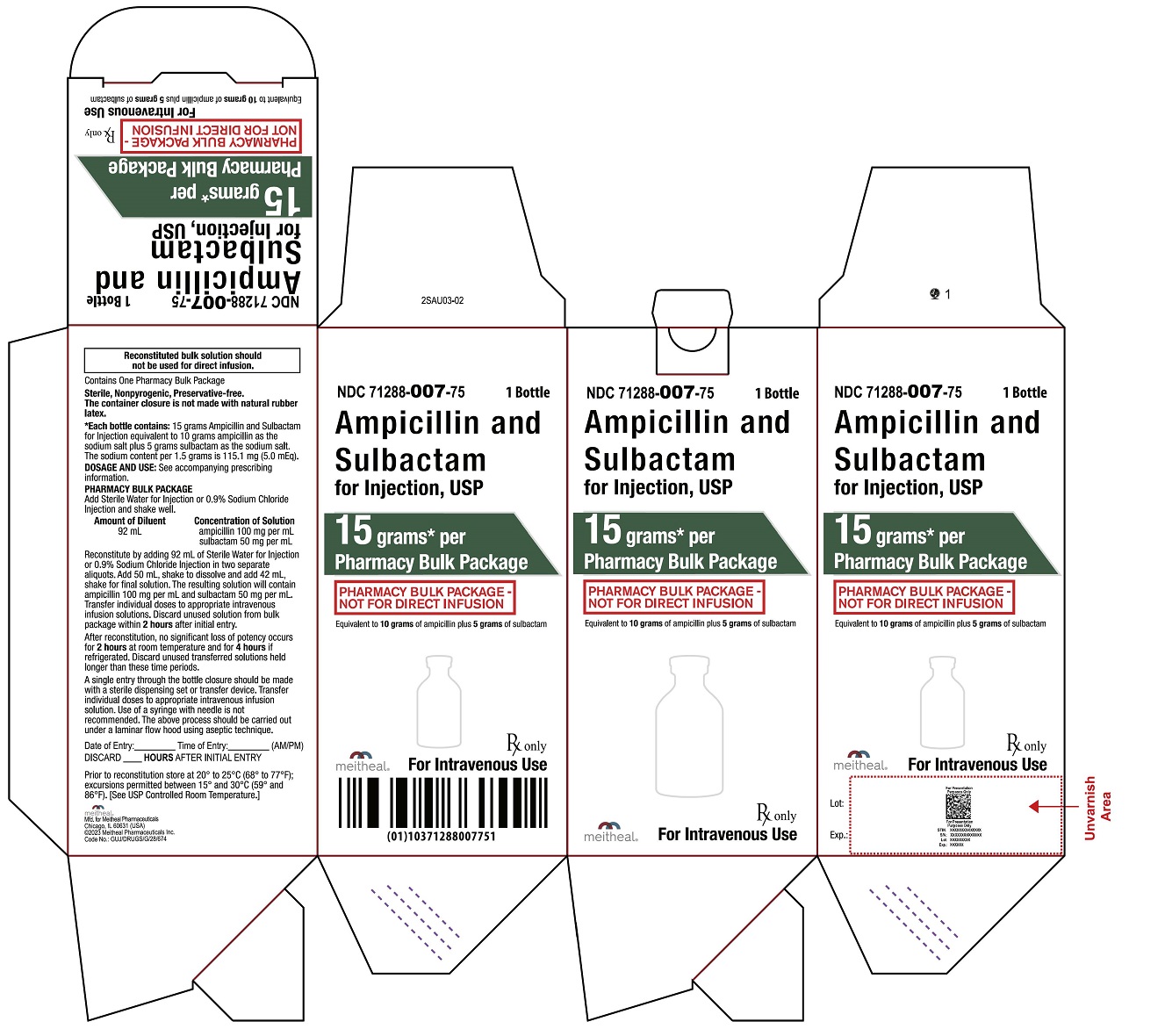
PRINCIPAL DISPLAY PANEL
NDC 71288-007-75
Rx only
Ampicillin and Sulbactam for Injection, USP
15 grams* per Pharmacy Bulk Package
PHARMACY BULK PACKAGE –
NOT FOR DIRECT INFUSION
Equivalent to 10 grams of ampicillin
plus 5 grams of sulbactam
For Intravenous Use

PRINCIPAL DISPLAY PANEL
NDC 71288-007-75
1 Bottle
Ampicillin and Sulbactam for Injection, USP
15 grams* per Pharmacy Bulk Package
PHARMACY BULK PACKAGE –
NOT FOR DIRECT INFUSION
Equivalent to 10 grams of ampicillin
plus 5 grams of sulbactam
Rx only
For Intravenous Use