NDC Code(s) : 70748-261-01, 70748-261-30, 70748-261-60
Packager : Lupin Pharmaceuticals, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| formoterol fumarate formoterol fumarate SOLUTION | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| LABELER - Lupin Pharmaceuticals, Inc.(089153071) |
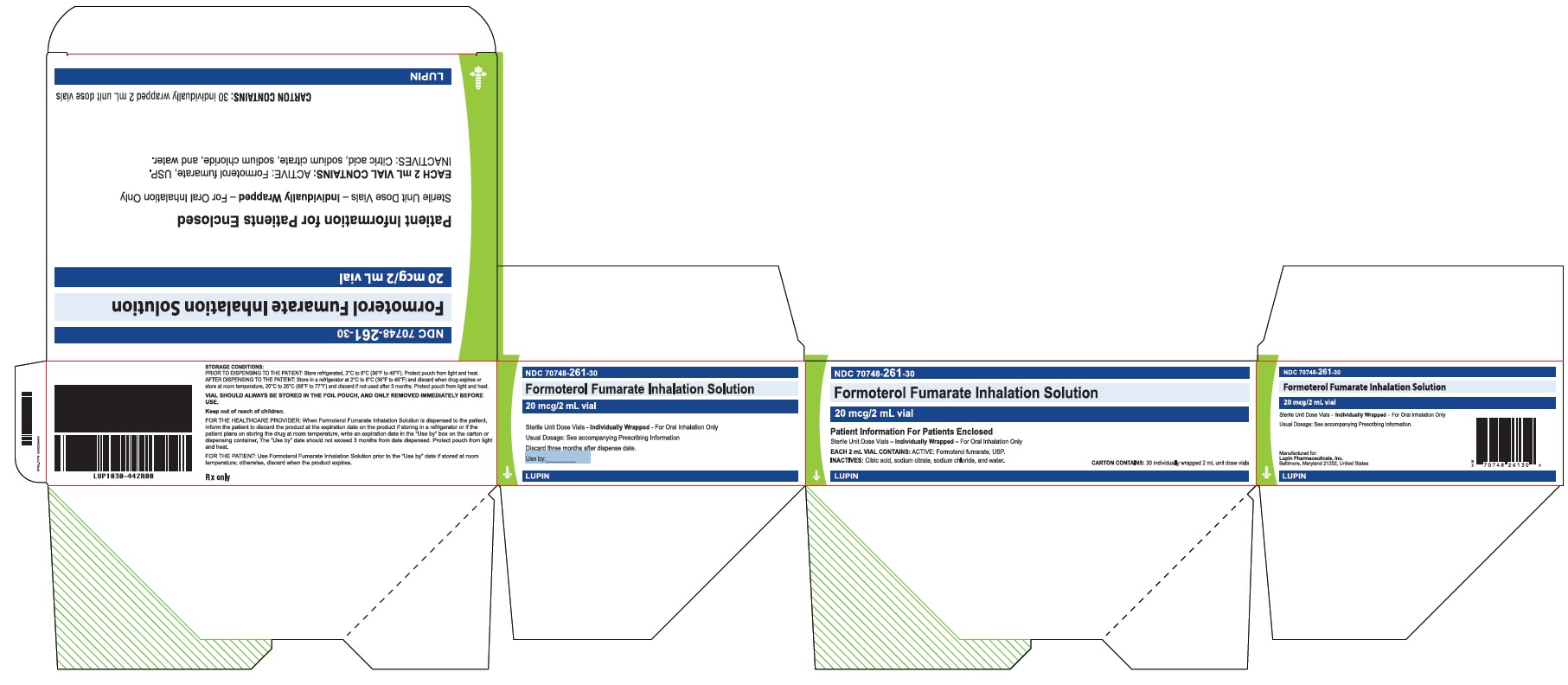
PRINCIPAL DISPLAY PANEL
Formoterol Fumarate Inhalation Solution
20 mcg/2 mL Vial
Rx Only
Sterile Unit Dose Vials
For Oral Inhalation Only.
Each 2 mL Vial Contains:
ACTIVE: Formoterol fumarate, USP
INACTIVES: Citric acid, sodium citrate, sodium chloride, and water.
NDC 70748-261-01 - Pouch Label

Formoterol Fumarate Inhalation Solution
20 mcg/2 mL Vial
Rx Only
Patient Information for Patient Enclosed
Sterile Unit Dose Vials - Individually Wrapped - For Oral Inhalation Only.
Each 2 mL Vial Contains: ACTIVE: Formoterol fumarate, USP
INACTIVES: Citric acid, sodium citrate, sodium chloride, and water.
NDC 70748-261-30
Carton contains: 30 individual wrapped 2 mL unit dose vials