NDC Code(s) : 70377-039-11, 70377-040-11
Packager : Biocon Pharma Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Mycophenolic AcidMycophenolic Acid TABLET, DELAYED RELEASE | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Mycophenolic AcidMycophenolic Acid TABLET, DELAYED RELEASE | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| LABELER - Biocon Pharma Inc.(080000063) |
| REGISTRANT - Biocon Pharma Limited(871412155) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Biocon Pharma Limited | 871412155 | analysis(70377-039, 70377-040), manufacture(70377-039, 70377-040), pack(70377-039, 70377-040) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Biocon Limited | 650173537 | api manufacture(70377-039, 70377-040) | |
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL
NDC 70377-039-11
Mycophenolic Acid
Delayed-Release Tablets, USP
180 mg*
*as Mycophenolate sodium
Rx Only
PHARMACIST:Dispense the accompanying
Medication Guide to each patient.
120 Tablets

PRINCIPAL DISPLAY PANEL
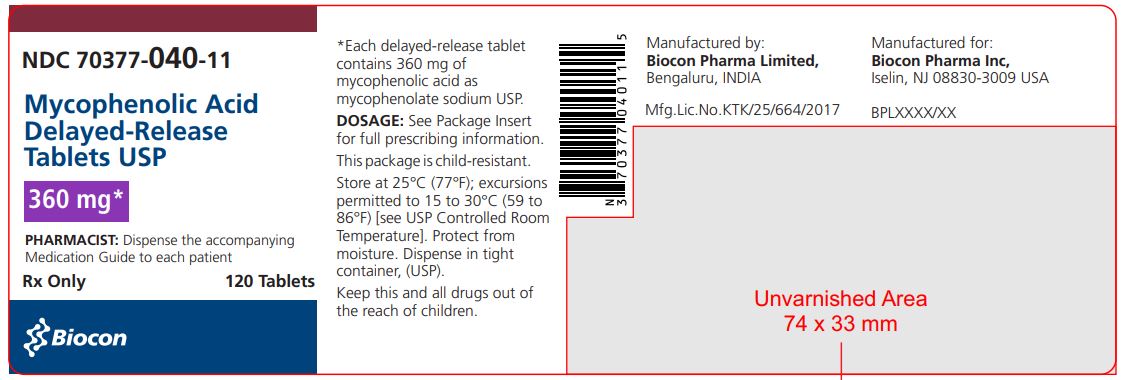
PRINCIPAL DISPLAY PANEL
NDC 70377-040-11
Mycophenolic Acid
Delayed-Release Tablets, USP
360 mg*
*as Mycophenolate sodium
Rx Only
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
120 Tablets