NDC Code(s) : 69844-001-01, 69844-001-02, 69844-001-03, 69844-001-04, 69844-002-01, 69844-002-02, 69844-002-03, 69844-002-04, 69844-003-01, 69844-003-02, 69844-003-03, 69844-003-04, 69844-004-01, 69844-004-02, 69844-004-03, 69844-004-04
Packager : Graviti Pharmaceuticals Private Limited
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Atorvastatin Calcium atorvastatin calcium, film coated TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Atorvastatin Calcium atorvastatin calcium, film coated TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Atorvastatin Calcium atorvastatin calcium, film coated TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Atorvastatin Calcium atorvastatin calcium, film coated TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| LABELER - Graviti Pharmaceuticals Private Limited(650884781) |
| REGISTRANT - Graviti Pharmaceuticals Private Limited(650884781) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Graviti Pharmaceuticals Private Limited | 650884781 | MANUFACTURE(69844-001, 69844-002, 69844-003, 69844-004), ANALYSIS(69844-001, 69844-002, 69844-003, 69844-004) | |
PRINCIPAL DISPLAY PANEL
NDC 69844-001-01
30 Tablets
Rx only
Atorvastatin Calcium Tablets, USP
10 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

NDC 69844-001-04
100 Tablets (10 x 10 Unit-Dose)
Rx only
Atorvastatin Calcium Tablets, USP
10 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

PRINCIPAL DISPLAY PANEL
NDC 69844-002-01
30 Tablets
Rx only
Atorvastatin Calcium Tablets, USP
20 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

NDC 69844-002-04
100 Tablets (10 x 10 Unit-Dose)
Rx only
Atorvastatin Calcium Tablets, USP
20 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

PRINCIPAL DISPLAY PANEL
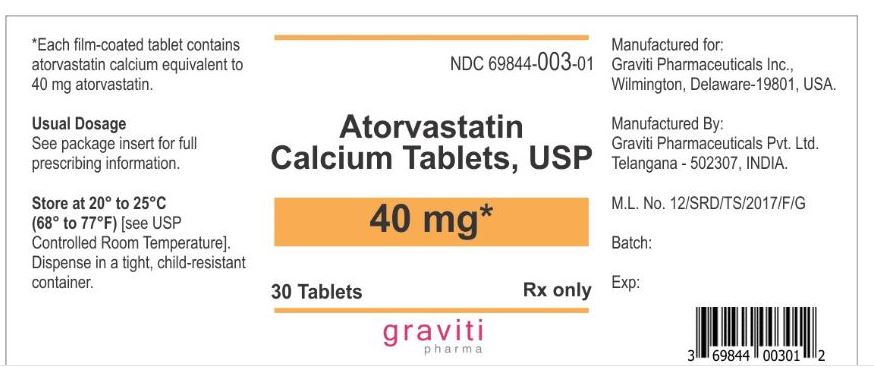
NDC 69844-003-01
30 Tablets
Rx only
Atorvastatin Calcium Tablets, USP
40 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.
NDC 69844-003-04
100 Tablets (10 x 10 Unit-Dose)
Rx only
Atorvastatin Calcium Tablets, USP
40 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

PRINCIPAL DISPLAY PANEL
NDC 69844-004-01
30 Tablets
Rx only
Atorvastatin Calcium Tablets, USP
80 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.

NDC 69844-004-04
100 Tablets (10 x 10 Unit-Dose)
Rx only
Atorvastatin Calcium Tablets, USP
80 mg*
Manufactured for Graviti Pharmaceuticals Inc.
Made in India.









