NDC Code(s) : 68453-375-10, 68453-850-75, 68453-777-03, 68453-900-02
Packager : Victory Pharma, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| NAPRELANnaproxen sodium TABLET, FILM COATED, EXTENDED RELEASE | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| NAPRELANnaproxen sodium TABLET, FILM COATED, EXTENDED RELEASE | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| NAPRELANnaproxen sodium TABLET, FILM COATED, EXTENDED RELEASE | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| NAPRELANnaproxen sodium KIT | |||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 375 MG BOTTLE
NDC 68453-375-10
NAPRELAN ®
(naproxen sodium)
CONTROLLED-RELEASE TABLETS
375 mg
Each tablet contains 412.5 mg naproxen sodium
equivalent to 375 mg naproxen
Rx only
100 Tablets
Victory Pharma

PRINCIPAL DISPLAY PANEL
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 500 MG BOTTLE
NDC 68453-850-75
NAPRELAN ®
(naproxen sodium)
CONTROLLED-RELEASE TABLETS
500 mg
Each tablet contains 550 mg naproxen sodium
equivalent to 500 mg naproxen
Rx only
75 Tablets
Victory Pharma

PRINCIPAL DISPLAY PANEL
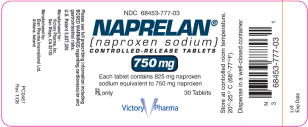
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 750 MG BOTTLE
NDC 68453-777-03
NAPRELAN ®
(naproxen sodium)
CONTROLLED-RELEASE TABLETS
750 mg
Each tablet contains 825 mg naproxen sodium
equivalent to 750 mg naproxen
Rx only
30 Tablets
Victory Pharma
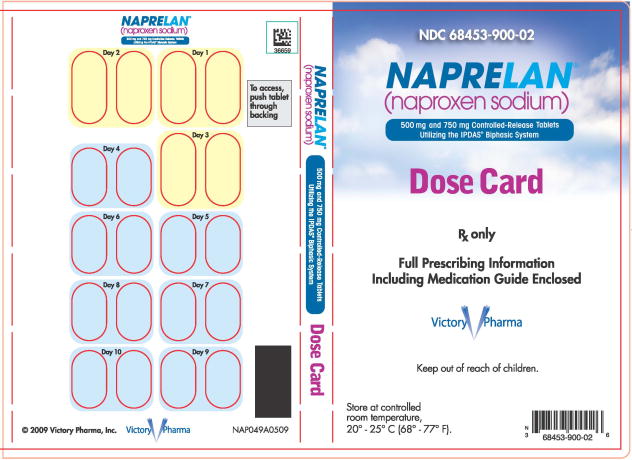
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – DOSE CARD
NDC 68453-900-02
NAPRELAN ®
(naproxen sodium)
500 mg and 750 mg Controlled-Release Tablets
Utilizing the IPDAS® Biphasic System
Dose Card
Rx only
Full Prescribing Information
Including Medication Guide Enclosed
Victory Pharma
Keep out of reach of children
Store at controlled
room temperature,
20°-25° C (68°-77°F).