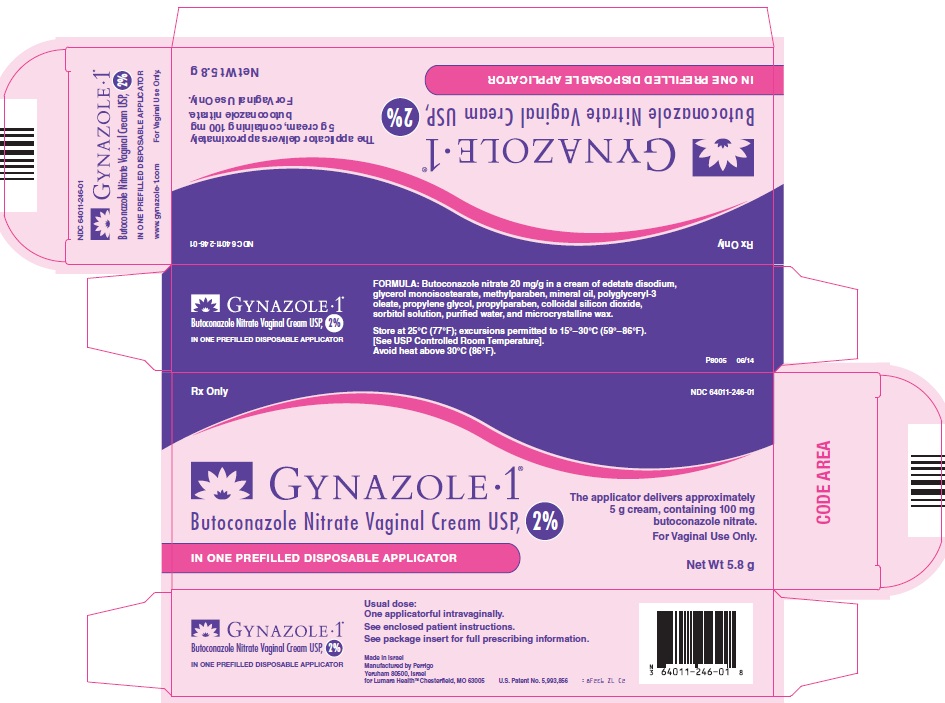
NDC Code(s) : 64011-246-01
Packager : Lumara Health, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| gynazole 1Butoconazole Nitrate CREAM | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
Rx Only
GYNAZOLE • 1®
Butoconazole Nitrate Vaginal Cream USP, 2%
The applicator delivers approximately 5 g cream, containing 100 mg butoconazole nitrate.
For Vaginal Use Only.
IN ONE PREFILLED DISPOSABLE APPLICATOR
Net Wt 5.8 g