NDC Code(s) : 60793-800-01, 60793-801-01, 60793-802-01
Packager : Pfizer Laboratories Div Pfizer Inc
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| CORGARDnadolol TABLET | ||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| CORGARDnadolol TABLET | ||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| CORGARDnadolol TABLET | ||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
PRINCIPAL DISPLAY PANEL
Pfizer
NDC 60793-800-01
Corgard
®
(nadolol tablets, USP)
20 mg
100 Tablets
Rx only

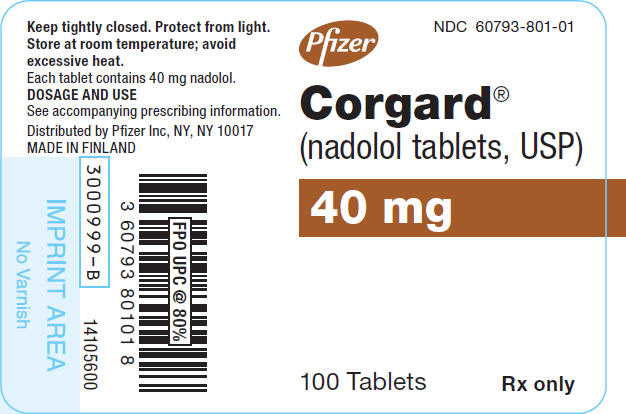
PRINCIPAL DISPLAY PANEL
Pfizer
NDC 60793-801-01
Corgard
®
(nadolol tablets, USP)
40 mg
100 Tablets
Rx only
PRINCIPAL DISPLAY PANEL
Pfizer
NDC 60793-802-01
Corgard
®
(nadolol tablets, USP)
80 mg
100 Tablets
Rx only









