NDC Code(s) : 60505-0678-0, 60505-0678-1, 60505-0678-4, 60505-0834-0, 60505-0834-1, 60505-0834-4, 60505-0681-0, 60505-0681-1, 60505-0681-4
Packager : Apotex Corporation
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| CEFEPIMECefepime INJECTION, POWDER, FOR SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| CEFEPIMECefepime INJECTION, POWDER, FOR SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| CEFEPIMECefepime INJECTION, POWDER, FOR SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
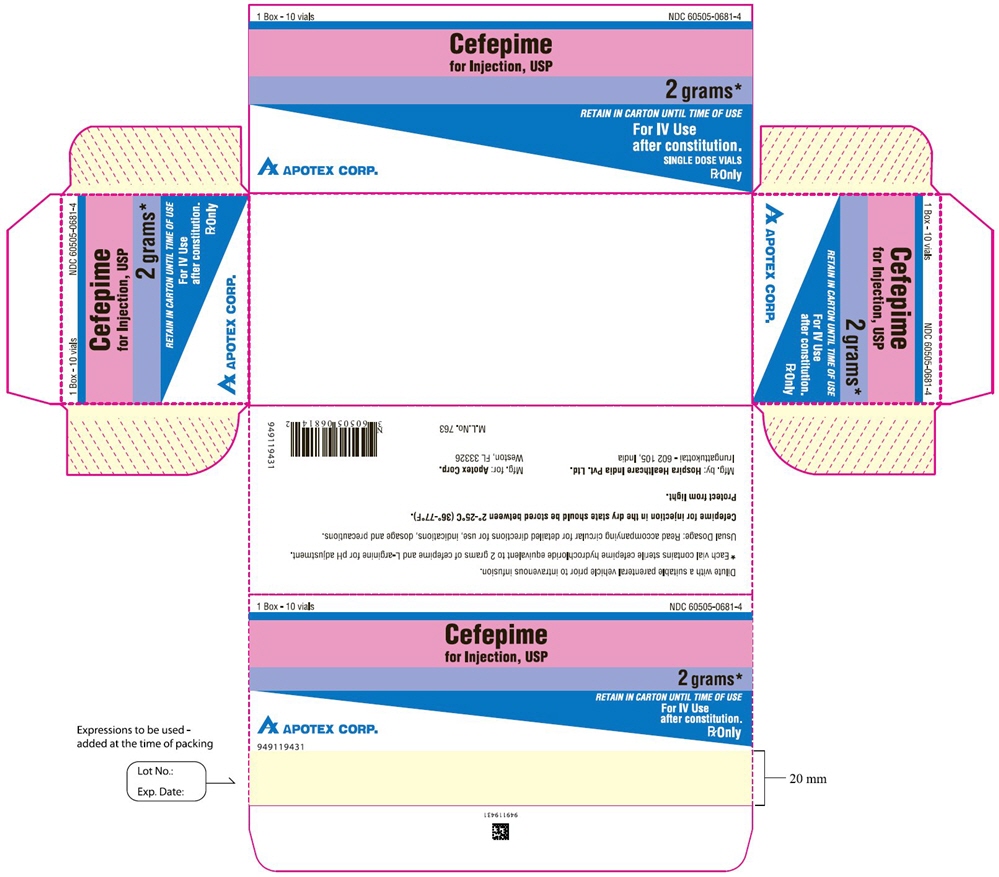
PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL
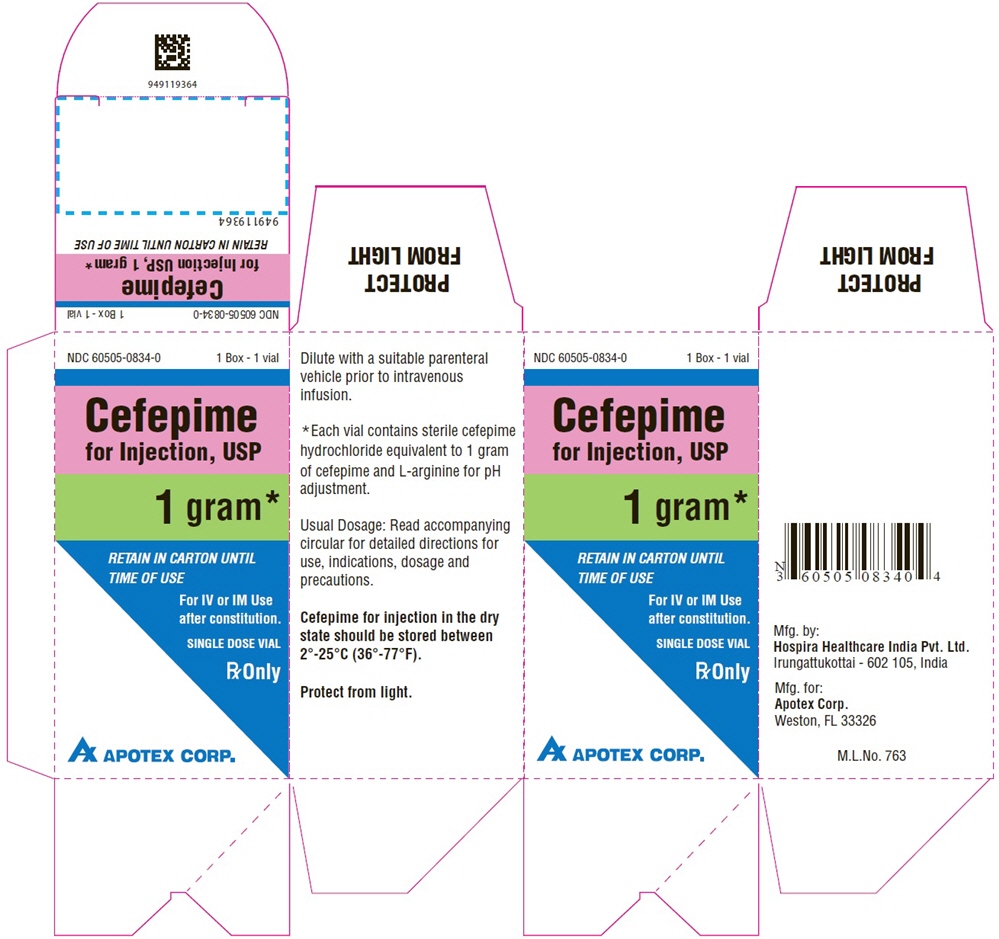
Single-Dose Vial
NDC 60505-0834-1
Cefepime
for Injection, USP
1 gram*
For IV or IM Use
after constitution.
Rx Only
APOTEX CORP.

PRINCIPAL DISPLAY PANEL
949119430

PRINCIPAL DISPLAY PANEL
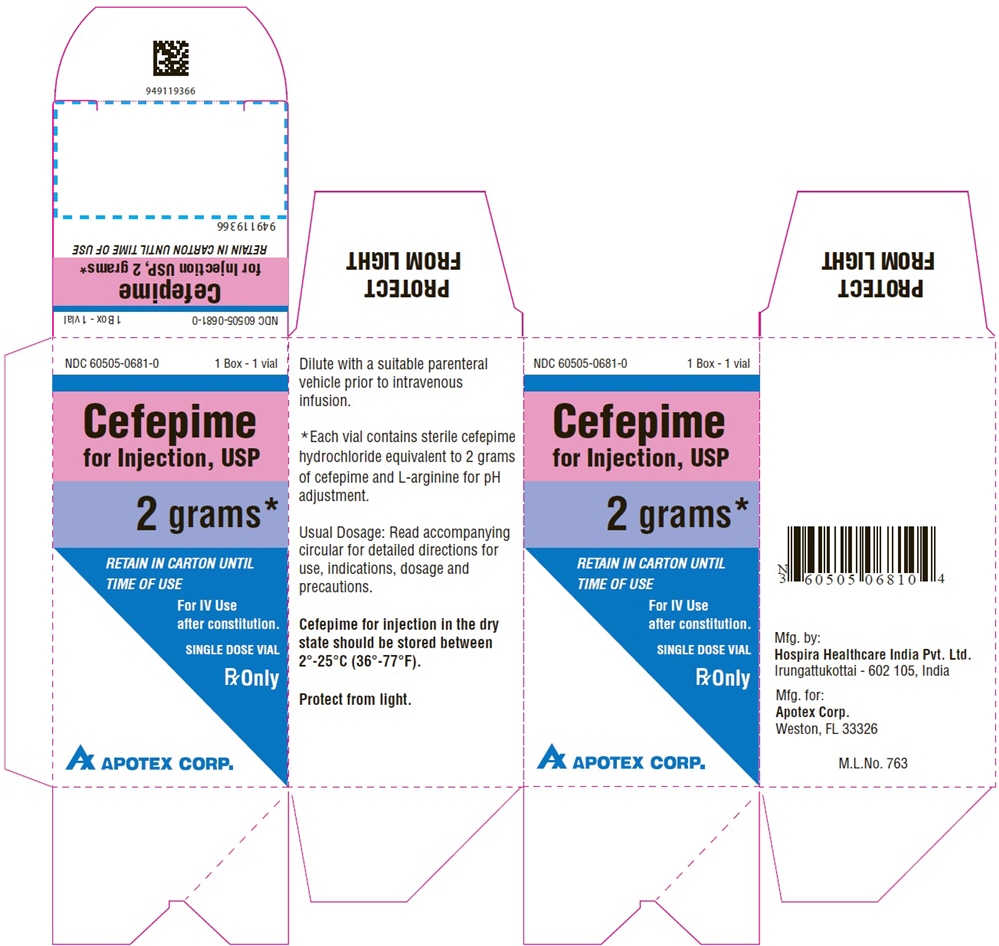
Single-Dose Vial
NDC 60505-0681-1
Cefepime
for Injection, USP
2 grams*
For IV Use
after constitution.
Rx Only
APOTEX CORP.

PRINCIPAL DISPLAY PANEL
949119431