NDC Code(s) : 59676-030-56, 59676-030-84, 59676-040-28, 59676-040-56, 59676-050-28
Packager : Janssen Products LP
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| BALVERSAErdafitinib TABLET, FILM COATED | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| BALVERSAErdafitinib TABLET, FILM COATED | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| BALVERSAErdafitinib TABLET, FILM COATED | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| LABELER - Janssen Products LP(804684207) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Cilag AG | 483237103 | api manufacture(59676-030, 59676-040, 59676-050) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Johnson & Johnson Private Limited | 677603030 | analysis(59676-030, 59676-040, 59676-050) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Janssen Cilag SpA | 542797928 | manufacture(59676-030, 59676-040, 59676-050), analysis(59676-030, 59676-040, 59676-050), pack(59676-030, 59676-040, 59676-050) | |
PRINCIPAL DISPLAY PANEL
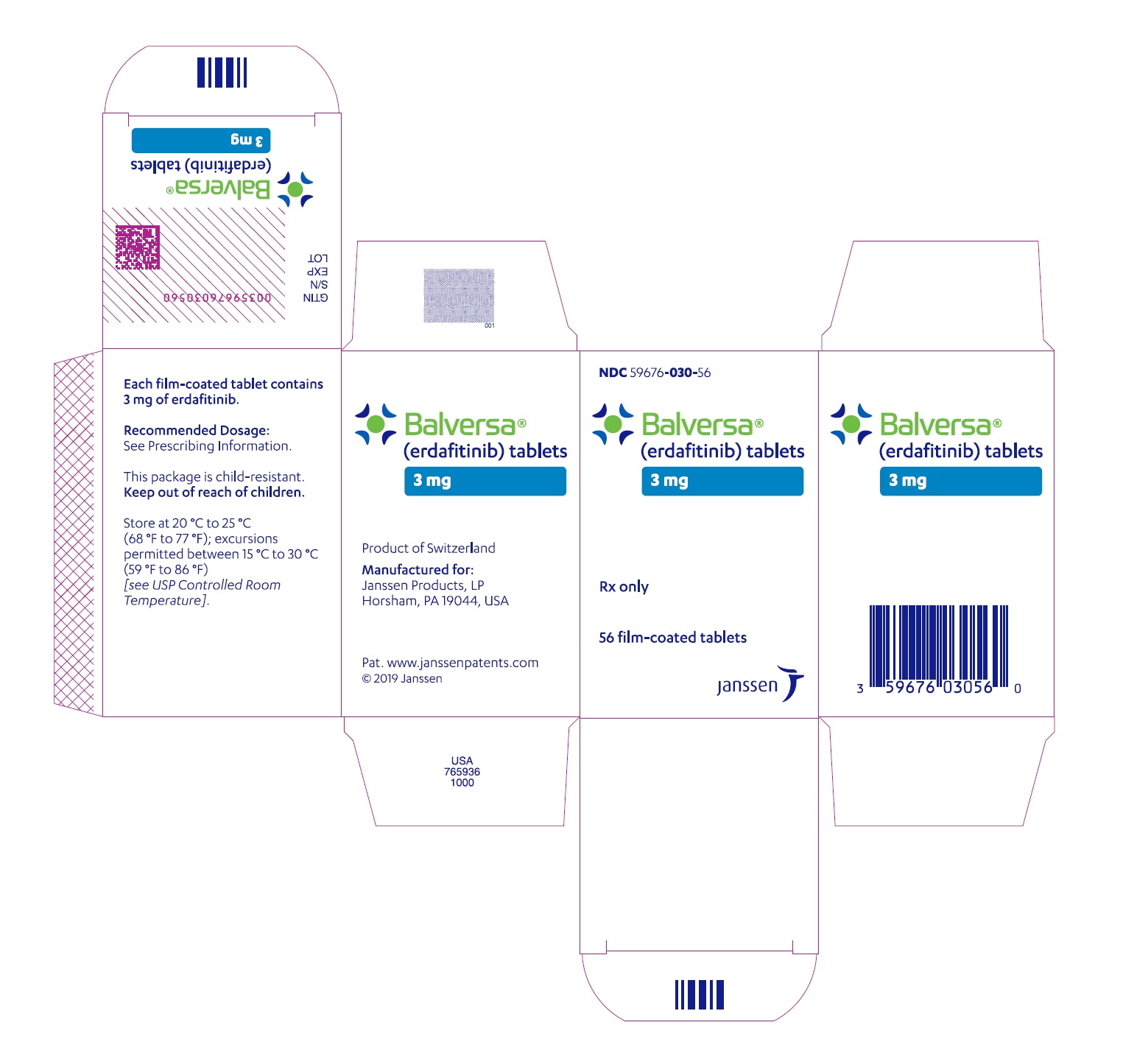 NDC 59676-030-56
NDC 59676-030-56
Balversa
®
(erdafitinib) tablets
3 mg
Each film-coated tablet
contains 3 mg of erdafitinib.
Rx only
56 film-coated tablets
PRINCIPAL DISPLAY PANEL
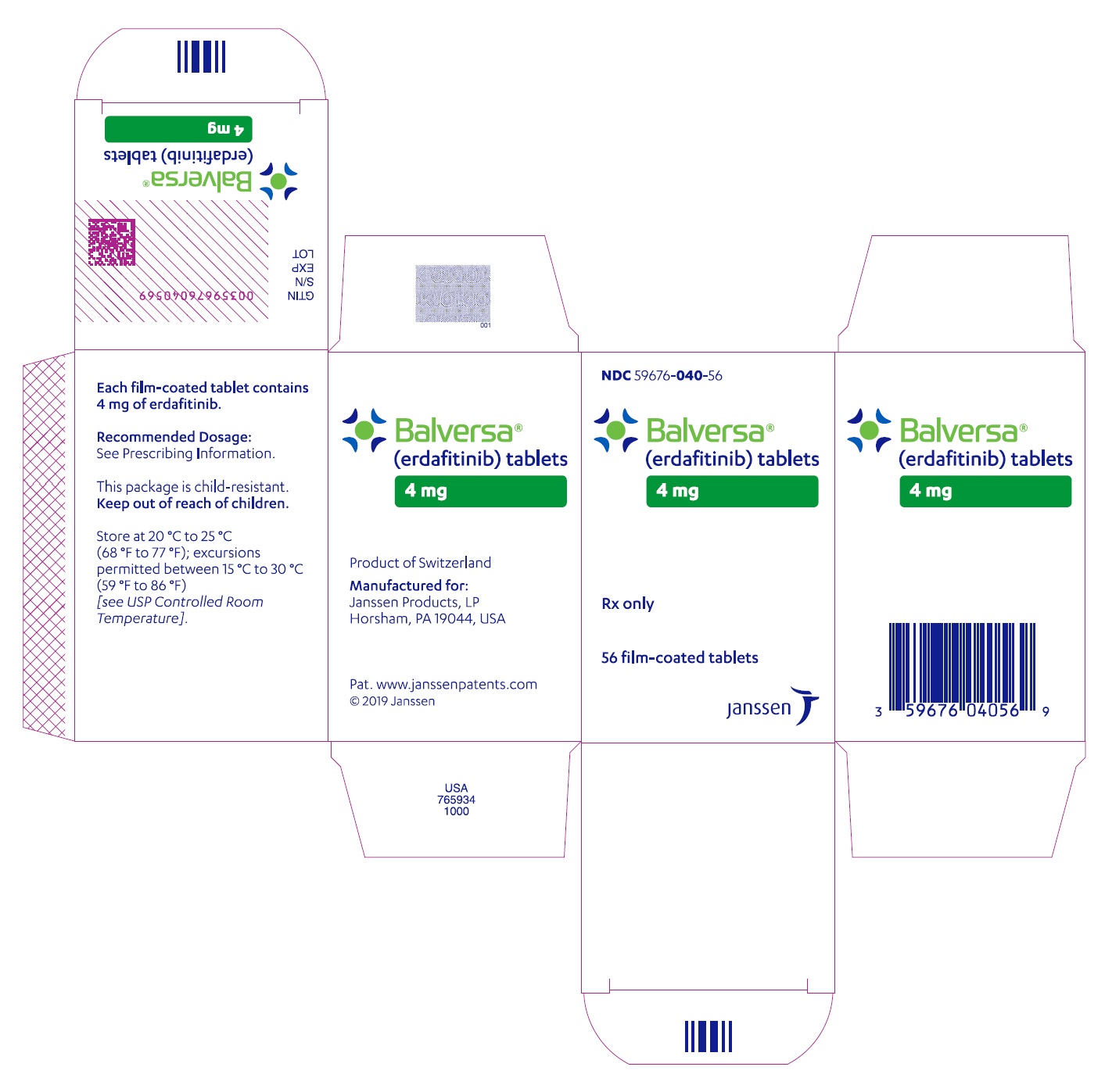 NDC 59676-040-56
NDC 59676-040-56
Balversa
®
(erdafitinib) tablets
4 mg
Each film-coated tablet
contains 4 mg of erdafitinib.
Rx only
56 film-coated tablets
PRINCIPAL DISPLAY PANEL
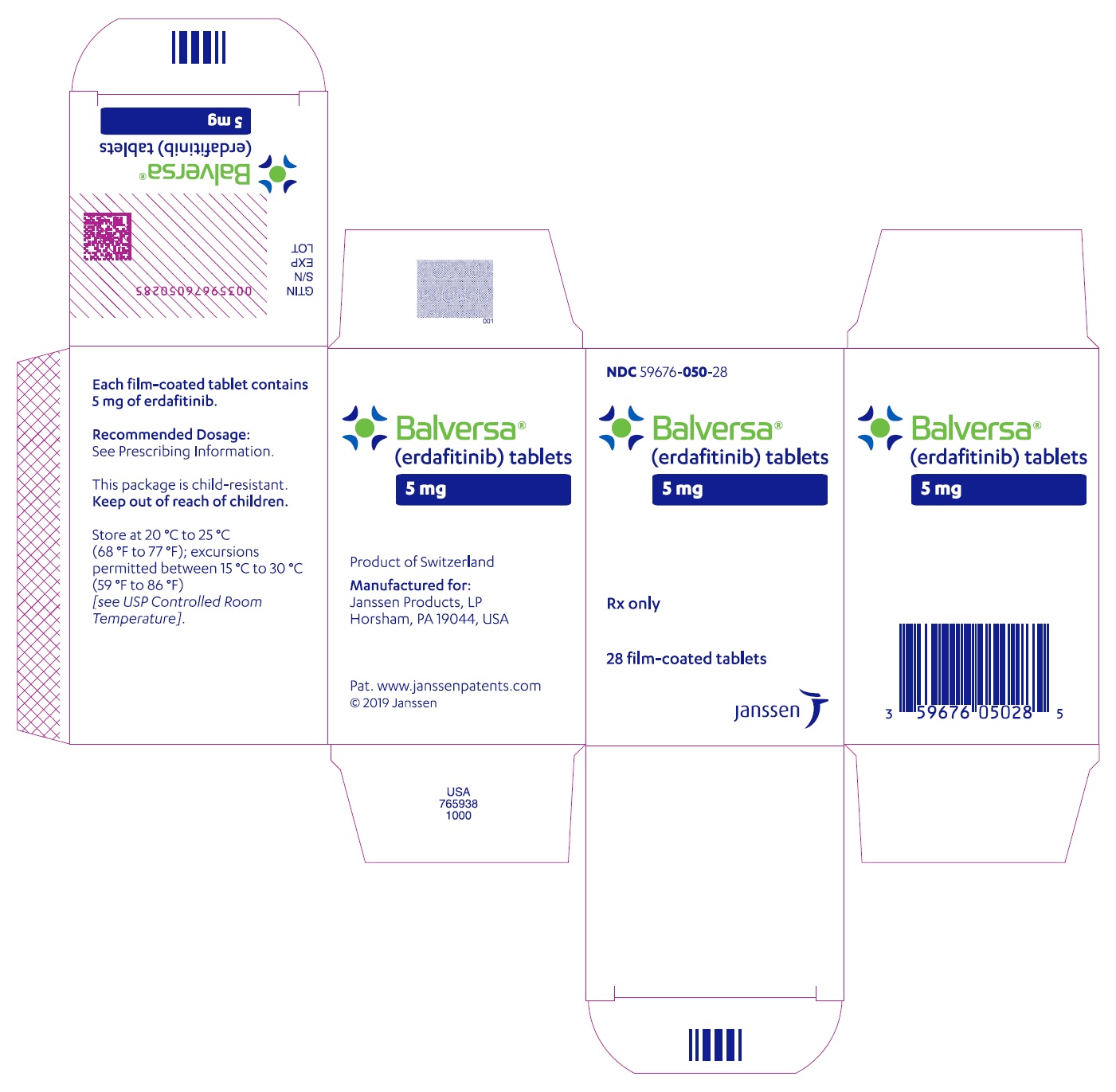 NDC 59676-050-28
NDC 59676-050-28
Balversa
®
(erdafitinib) tablets
5 mg
Each film-coated tablet
contains 5 mg of erdafitinib.
Rx only
28 film-coated tablets








