NDC Code(s) : 58602-730-36, 58602-730-07, 58602-730-14, 58602-730-21, 58602-730-29, 58602-730-34, 58602-730-35, 58602-730-67, 58602-730-76, 58602-730-40, 58602-730-41, 58602-730-94, 58602-730-44, 58602-730-28
Packager : Aurohealth LLC
Category : Human OTC Drug Label
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Acetaminophen Acetaminophen TABLET, EXTENDED RELEASE | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LABELER - Aurohealth LLC(078728447) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Aurobindo Pharma Limited | 650381903 | ANALYSIS(58602-730), MANUFACTURE(58602-730) | |
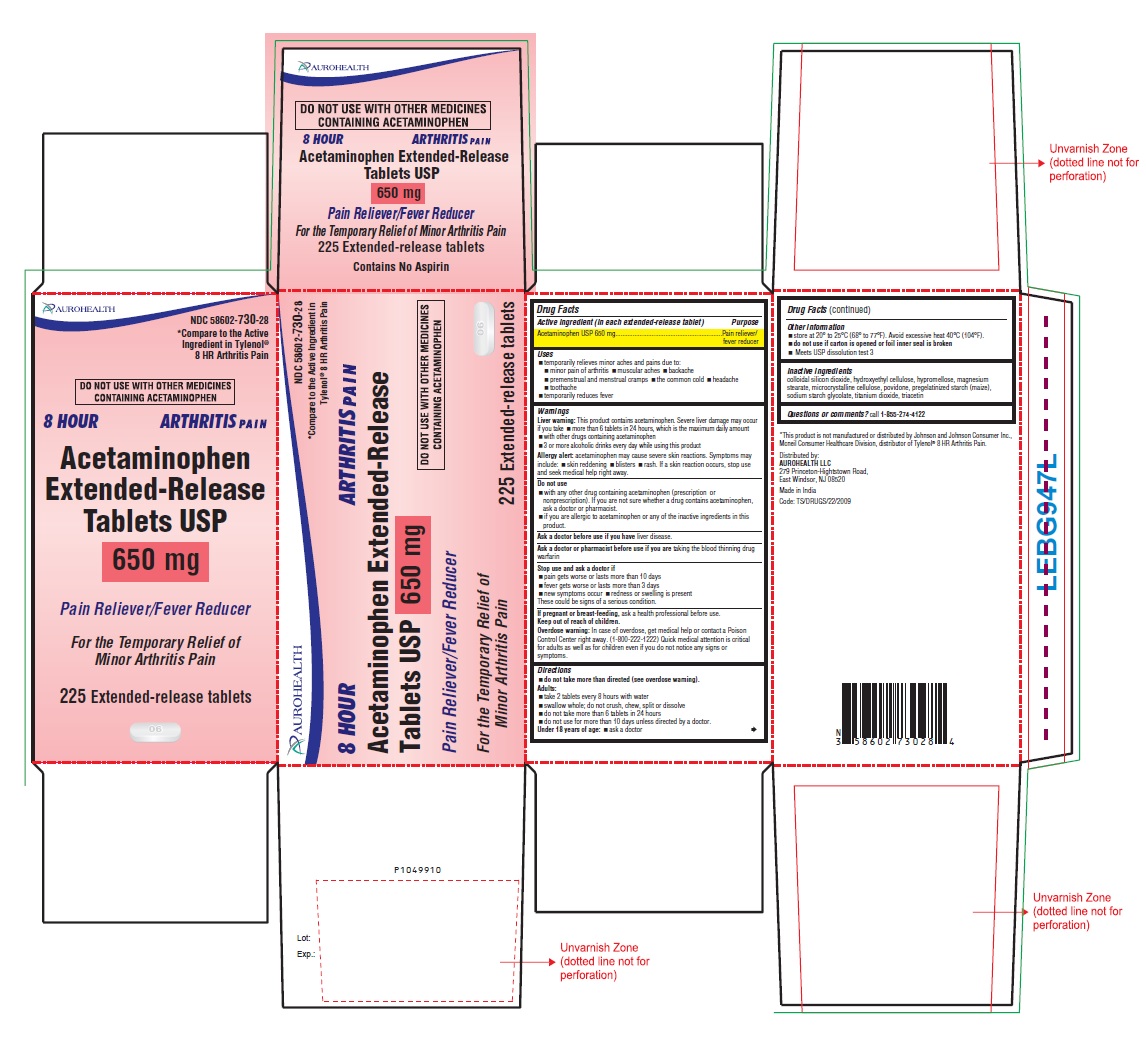
PRINCIPAL DISPLAY PANEL
AUROHEALTH
TO OPEN: 1. PUSH DOWN NDC 58602-730-28
2. TURN CAP
DO NOT USE WITH OTHER MEDICINES
CONTAINING ACETAMINOPHEN
8 HOUR ARTHRITIS PAIN
Acetaminophen
Extended-Release
Tablets USP
650 mg
Pain Reliever/Fever Reducer
For the Temporary Relief of
Minor Arthritis Pain
225 Extended-release tablets
PRINCIPAL DISPLAY PANEL
AUROHEALTH
NDC 58602-730-28
*Compare to the Active
Ingredient in Tylenol®
8 HR Arthritis Pain
DO NOT USE WITH OTHER MEDICINES
CONTAINING ACETAMINOPHEN
8 HOUR ARTHRITIS PAIN
Acetaminophen
Extended-Release
Tablets USP
650 mg
Pain Reliever/Fever Reducer
For the Temporary Relief of
Minor Arthritis Pain
225 Extended-release tablets