NDC Code(s) : 57664-376-83, 57664-376-88, 57664-376-08, 57664-376-13, 57664-376-18, 57664-378-83, 57664-378-88, 57664-378-08, 57664-378-13, 57664-378-18, 57664-379-83, 57664-379-88, 57664-379-08, 57664-379-13, 57664-379-18
Packager : Sun Pharmaceutical Industries, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : CII
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Dexmethylphenidate hydrochlorideDexmethylphenidate hydrochloride TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Dexmethylphenidate hydrochlorideDexmethylphenidate hydrochloride TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Dexmethylphenidate hydrochlorideDexmethylphenidate hydrochloride TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| LABELER - Sun Pharmaceutical Industries, Inc.(146974886) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Ohm Laboratories Inc. | 184769029 | manufacture(57664-376, 57664-378, 57664-379) | |
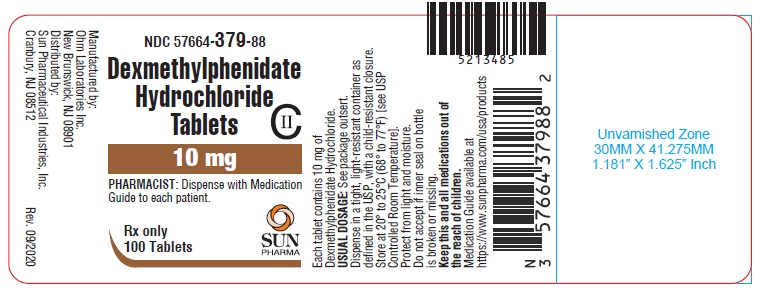
PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL
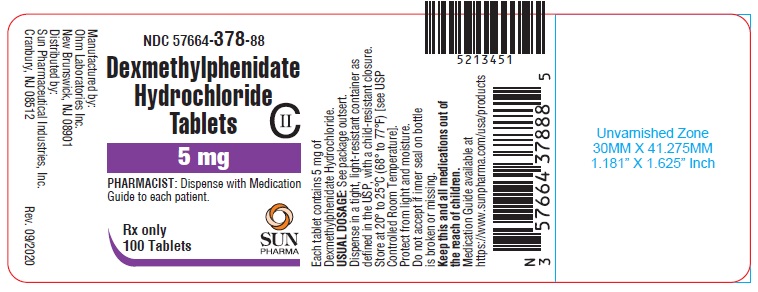
PRINCIPAL DISPLAY PANEL