NDC Code(s) : 57297-201-01, 57297-201-08, 57297-201-10, 57297-201-25, 57297-208-08, 57297-208-11, 57297-208-02, 57297-203-10, 57297-203-08, 57297-203-11, 57297-203-02, 57297-205-10, 57297-205-08, 57297-205-11, 57297-205-02, 57297-206-01, 57297-206-02, 57297-206-03, 57297-206-06, 57297-206-05, 57297-206-04, 57297-207-02, 57297-207-03, 57297-207-04
Packager : LUPIN LIMITED
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime TABLET | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime CAPSULE | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime TABLET, CHEWABLE | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime TABLET, CHEWABLE | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime POWDER, FOR SUSPENSION | |||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
INGREDIENTS AND APPEARANCE
| SUPRAXcefixime POWDER, FOR SUSPENSION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
SUPRAX® CEFIXIME TABLETS USP
400 mg
Rx only
NDC 27437-201-10: Bottle of 10 Tablets
NDC 27437-201-25: Bottle of 2 Tablets [Physician Sample Pack]
NDC 27437-201-25: Carton for 2 Tablets [Physician Sample Pack]



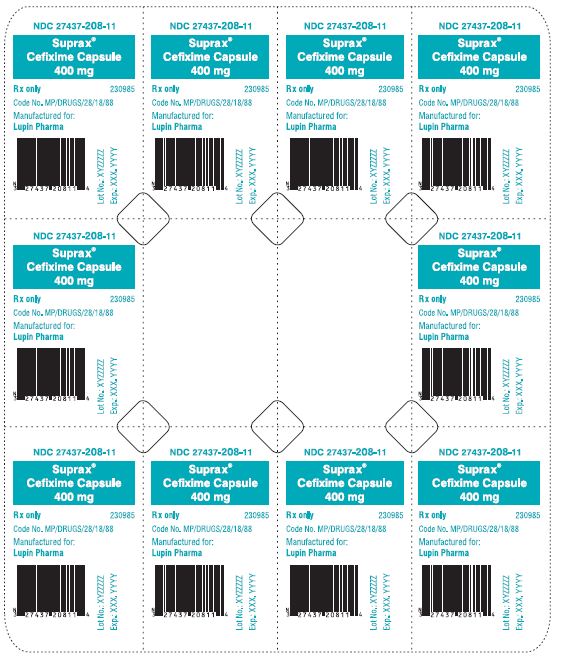
SUPRAX® CEFIXIME CAPSULES
400 mg
Rx only
NDC 27437-208-08: Bottle of 50 Capsules
NDC 27437-208-11: Unit Dose Package of 10 (1 Blister of 10 Capsules)
NDC 27437-208-02: Single Dose Package of 1 (Blister of 1 Capsule)




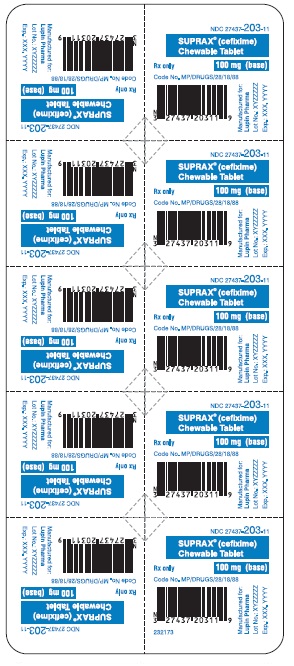
SUPRAX® CEFIXIME CHEWABLE TABLETS
100 mg
Rx only
NDC 27437-203-10: Bottle of 10 Tablets
NDC 27437-203-11: Unit Dose Package of 10 (1 Blister of 10 Tablets)
NDC 27437-203-02: Unit Dose Package of 1 (1 Blister of 1 Tablet)




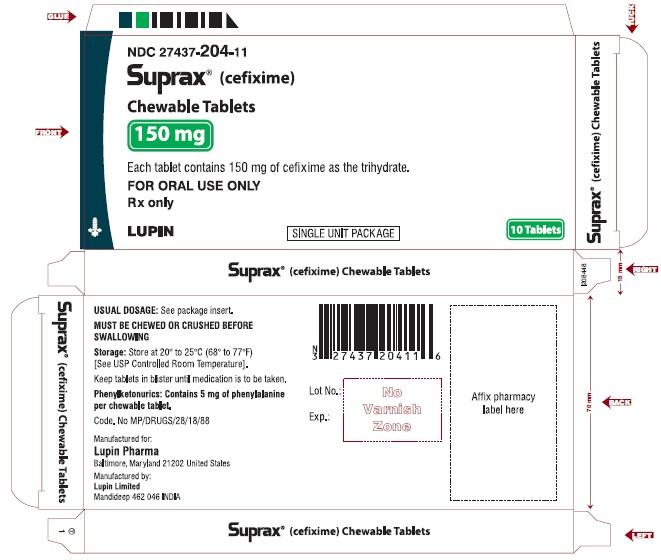
SUPRAX® CEFIXIME CHEWABLE TABLETS
150 mg
Rx only
NDC 27437-204-10: Bottle of 10 Tablets
NDC 27437-204-11: Unit Dose Package of 10 (1 Blister of 10 Tablets)


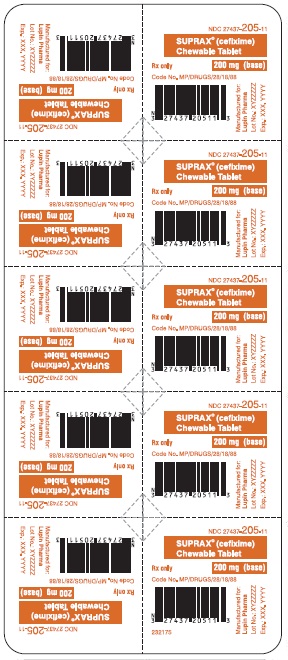
SUPRAX® CEFIXIME CHEWABLE TABLETS
200 mg
Rx only
NDC 27437-205-10: Bottle of 10 Tablets
NDC 27437-205-11: Unit Dose Package of 10 (1 Blister of 10 Tablets)
NDC 27437-205-02: Single Dose Package (Blister of 1 Tablet)

SUPRAX® CEFIXIME FOR ORAL SUSPENSION USP
200 mg/5 mL
Rx only
NDC 27437-206-05: Bottle of 25 mL
NDC 27437-206-04: Bottle of 10 mL [Physician Sample Pack]
NDC 27437-206-04: Carton for 10 mL [Physician Sample Pack]
SUPRAX® CEFIXIME FOR ORAL SUSPENSION USP
500 mg/5 mL
Rx only
NDC 27437-207-02: Bottle of 10 mL
NDC 27437-207-02: Carton for 10 mL
NDC 27437-207-04: Bottle of 10 mL [Physician Sample Pack]
NDC 27437-207-04: Carton for 10 mL [Physician Sample Pack]








