NDC Code(s) : 53270-3120-1, 53270-3500-1, 53270-3100-1, 53270-3000-1, 53270-3300-1
Packager : Cangene BioPharma, LLC
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| WINRHOrho (d) immune globulin LIQUID | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WINRHOrho (d) immune globulin LIQUID | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WINRHOrho (d) immune globulin LIQUID | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WINRHOrho (d) immune globulin LIQUID | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WINRHOrho (d) immune globulin LIQUID | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
PRINCIPAL DISPLAY PANEL

WinRho ® SDF Liquid
600 IU (120µg)
Target Vol.: 0.5 mL
PRINCIPAL DISPLAY PANEL
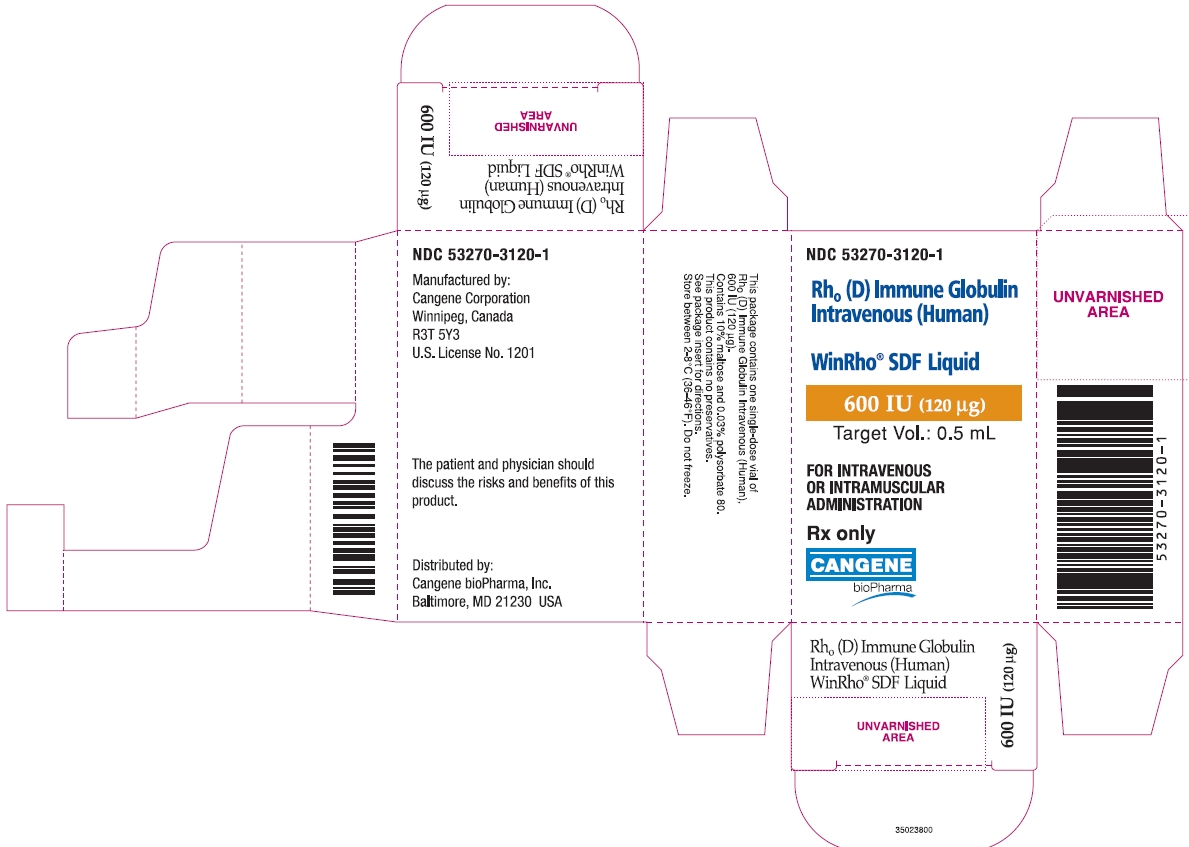
Rh o(D) Immune Globulin Intravenous (Human)
WinRho ® SDF Liquid
600 IU (120µg)
Target Vol.: 0.5 mL
For Intravenous or Intramuscular Administration
Rx Only
PRINCIPAL DISPLAY PANEL

WinRho ® SDF Liquid
1500 IU (300µg)
Target Vol.: 1.3 mL
PRINCIPAL DISPLAY PANEL
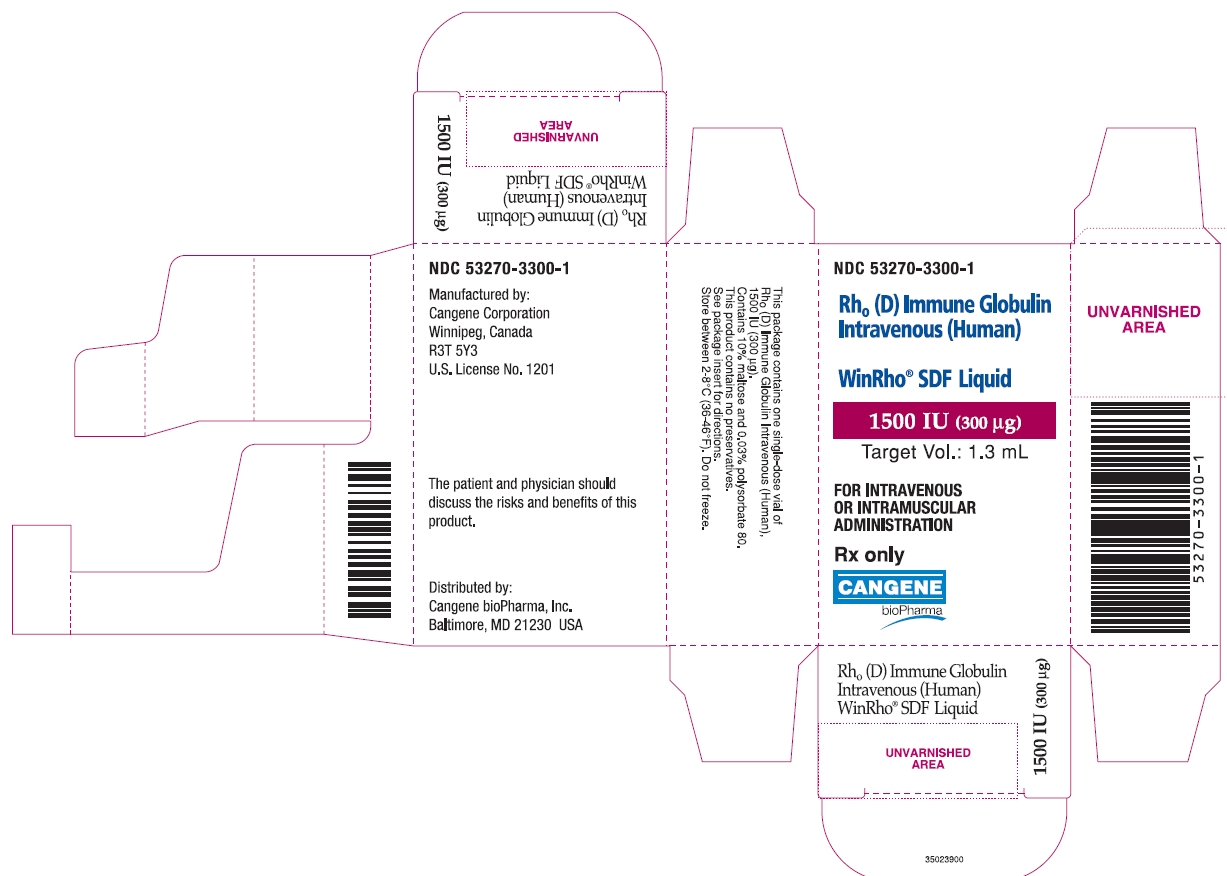
Rh o(D) Immune Globulin Intravenous (Human)
WinRho ® SDF Liquid
1500 IU (300µg)
Target Vol.: 1.3 mL
For Intravenous or Intramuscular Administration
Rx Only
PRINCIPAL DISPLAY PANEL

WinRho ® SDF Liquid
2500 IU (500µg)
Target Vol.: 2.2 mL
PRINCIPAL DISPLAY PANEL
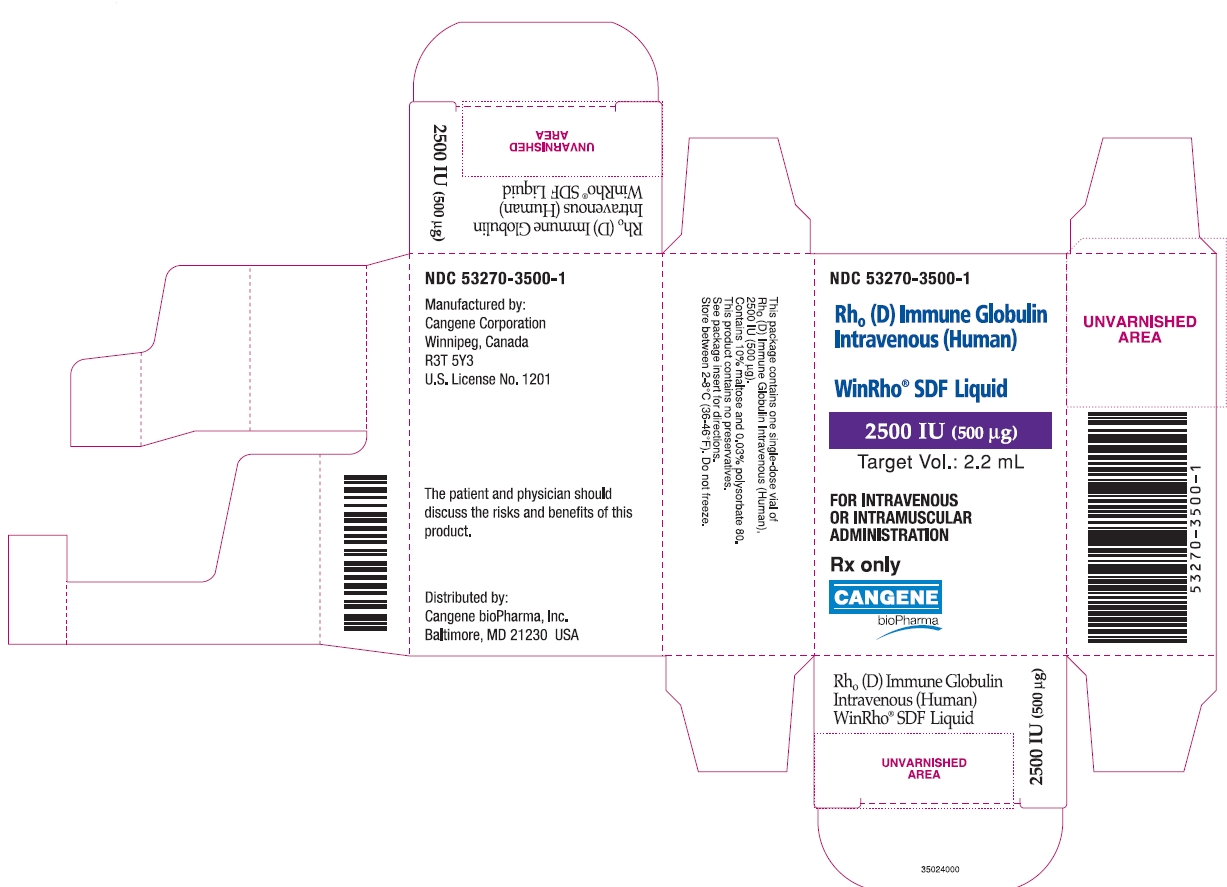
Rh o(D) Immune Globulin Intravenous (Human)
WinRho ® SDF Liquid
2500 IU (500µg)
Target Vol.: 2.2 mL
For Intravenous or Intramuscular Administration
Rx Only
PRINCIPAL DISPLAY PANEL
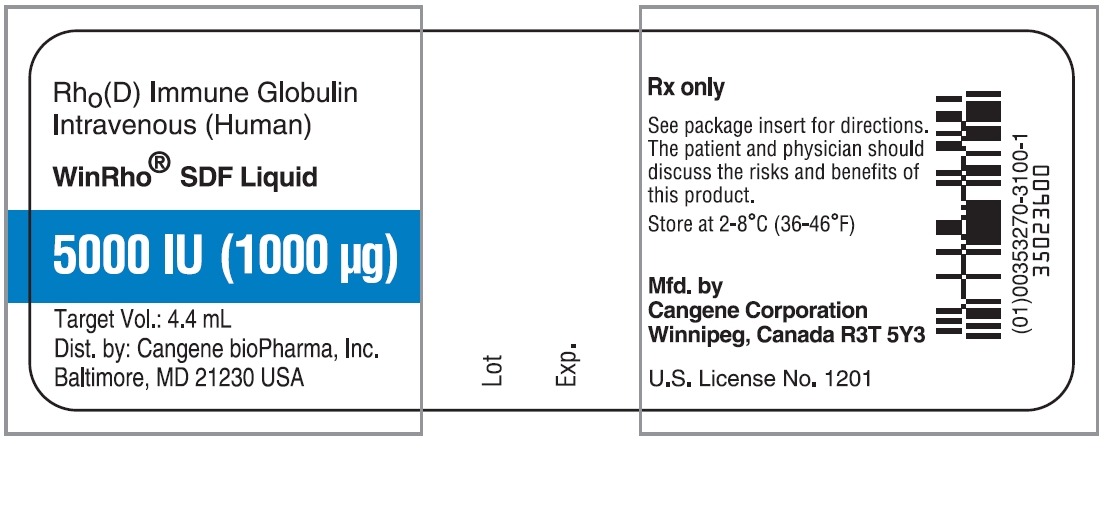
WinRho ® SDF Liquid
5000 IU (1000µg)
Target Vol.: 4.4 mL
PRINCIPAL DISPLAY PANEL
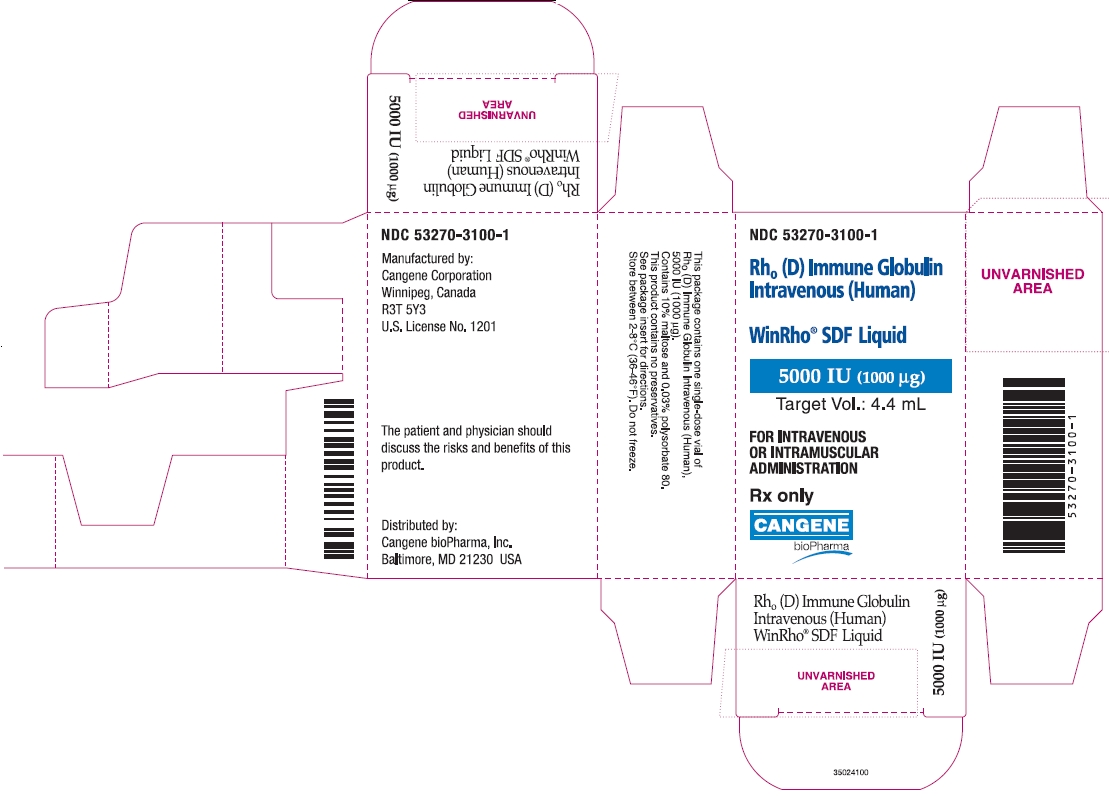
Rh o(D) Immune Globulin Intravenous (Human)
WinRho ® SDF Liquid
5000 IU (1000µg)
Target Vol.: 4.4 mL
For Intravenous or Intramuscular Administration
Rx Only
PRINCIPAL DISPLAY PANEL

WinRho ® SDF Liquid
15000 IU (3000µg)
Target Vol.: 13.0 mL
PRINCIPAL DISPLAY PANEL

Rh o(D) Immune Globulin Intravenous (Human)
WinRho ® SDF Liquid
15000 IU (3000µg)
Target Vol.: 13.0 mL
For Intravenous or Intramuscular Administration
Rx Only








