NDC Code(s) : 50419-701-10, 50419-701-05, 50419-702-05, 50419-702-10, 50419-703-05
Packager : Bayer HealthCare Pharmaceuticals Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Adalat CCnifedipine TABLET, FILM COATED | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Adalat CCnifedipine TABLET, FILM COATED | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Adalat CCnifedipine TABLET, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
Adalat 30 mg 1000 Tablet Label
83396521 NDC 50419-701-10
ADALAT© CC
(nifedipine)
Extended Release Tablets
30 mg
1000 Tablets Rx Only
Rx Only

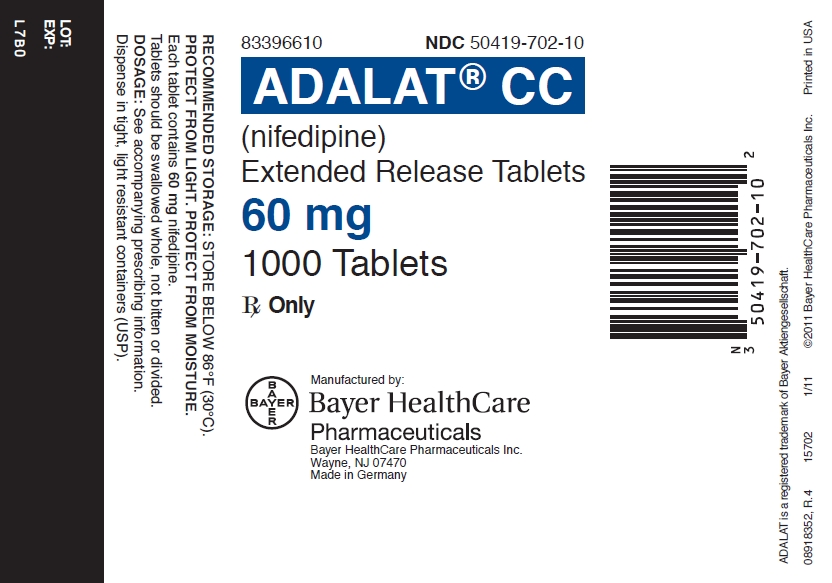
PRINCIPAL DISPLAY PANEL
Adalat 60 mg 1000 Tablet Label
83396610 NDC 50419-702-10
ADALAT© CC
(nifedipine)
Extended Release Tablets
60 mg
1000 Tablets Rx Only
Rx Only
PRINCIPAL DISPLAY PANEL
Adalat 90 mg 1000 Tablet Label
83396645 NDC 50419-703-05
ADALAT© CC
(nifedipine)
Extended Release Tablets
90 mg
100 Tablets Rx Only
Rx Only









