NDC Code(s) : 50228-451-90, 50228-451-10, 50228-452-90, 50228-452-05, 50228-453-90, 50228-453-10, 50228-454-90, 50228-454-05
Packager : ScieGen Pharmaceuticals, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| ATORVASTATIN CALCIUMATORVASTATIN CALCIUM TABLET, FILM COATED | ||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| ATORVASTATIN CALCIUMATORVASTATIN CALCIUM TABLET, FILM COATED | ||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| ATORVASTATIN CALCIUMATORVASTATIN CALCIUM TABLET, FILM COATED | ||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| ATORVASTATIN CALCIUMATORVASTATIN CALCIUM TABLET, FILM COATED | ||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| LABELER - ScieGen Pharmaceuticals, Inc.(079391286) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| ScieGen Pharmaceuticals, Inc. | 079391286 | manufacture(50228-451, 50228-452, 50228-453, 50228-454), analysis(50228-451, 50228-452, 50228-453, 50228-454) | |
PRINCIPAL DISPLAY PANEL
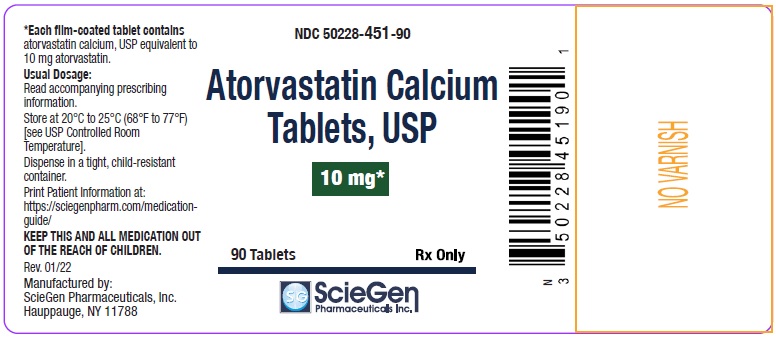
NDC 50228- 451-90
Atorvastatin Calcium
Tablets, USP
10 mg *
90 Tablets Rx Only
ScieGen Pharmaceuticals Inc.
NDC 50228- 451-10
Atorvastatin Calcium
Tablets, USP
10 mg *
1,000 Tablets Rx Only
ScieGen Pharmaceuticals Inc.

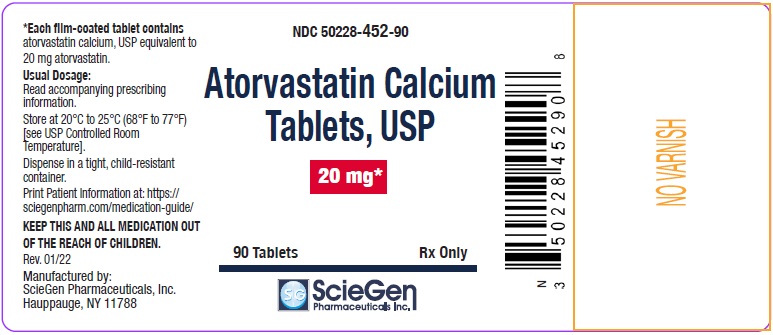
NDC 50228- 452-90
Atorvastatin Calcium
Tablets, USP
20 mg *
90 Tablets Rx Only
ScieGen Pharmaceuticals Inc.
NDC 50228- 452-05
Atorvastatin Calcium
Tablets, USP
20 mg *
500 Tablets Rx Only
ScieGen Pharmaceuticals Inc.

NDC 50228- 453-90
Atorvastatin Calcium
Tablets, USP
40 mg *
90 Tablets Rx Only
ScieGen Pharmaceuticals Inc.

NDC 50228- 453-10
Atorvastatin Calcium
Tablets, USP
40 mg *
1,000 Tablets Rx Only
ScieGen Pharmaceuticals Inc.

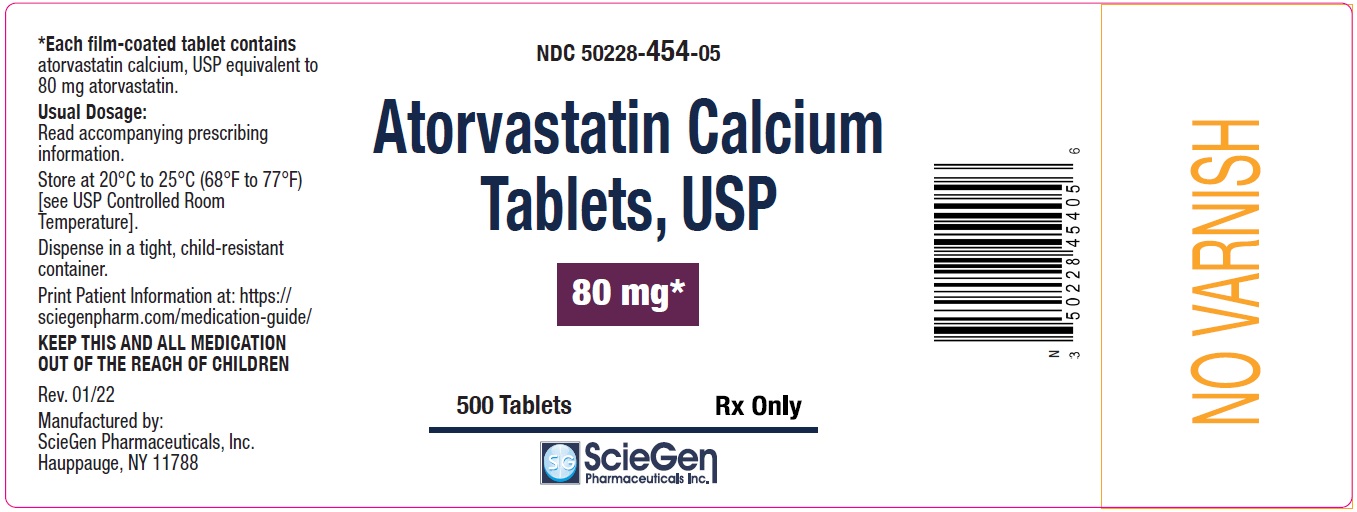
NDC 50228- 454-90
Atorvastatin Calcium
Tablets, USP
80 mg *
90 Tablets Rx Only
ScieGen Pharmaceuticals Inc.

NDC 50228- 454-05
Atorvastatin Calcium
Tablets, USP
80 mg *
500 Tablets Rx Only
ScieGen Pharmaceuticals Inc.