NDC Code(s) : 47335-582-83, 47335-582-81, 47335-582-18, 47335-583-83, 47335-583-81, 47335-583-18, 47335-584-83, 47335-584-81, 47335-584-18, 47335-585-83, 47335-585-81, 47335-585-18
Packager : Sun Pharmaceutical Industries, Inc.
Category : Human Prescription Drug Label
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Rosuvastatin CalciumRosuvastatin Calcium TABLET, FILM COATED | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Rosuvastatin CalciumRosuvastatin Calcium TABLET, FILM COATED | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Rosuvastatin CalciumRosuvastatin Calcium TABLET, FILM COATED | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Rosuvastatin CalciumRosuvastatin Calcium TABLET, FILM COATED | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| LABELER - Sun Pharmaceutical Industries, Inc.(146974886) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Sun Pharmaceutical Industries Limited | 650445203 | ANALYSIS(47335-582, 47335-583, 47335-584, 47335-585), MANUFACTURE(47335-582, 47335-583, 47335-584, 47335-585) | |
PRINCIPAL DISPLAY PANEL
NDC 47335-582-81
Rosuvastatin Tablets
5 mg
PHARMACIST: Dispense with Patient Information
Rx only
90 Tablets
SUN PHARMA

PRINCIPAL DISPLAY PANEL
NDC 47335-583-81
Rosuvastatin Tablets
10 mg
PHARMACIST: Dispense with Patient Information
Rx only
90 Tablets
SUN PHARMA

PRINCIPAL DISPLAY PANEL
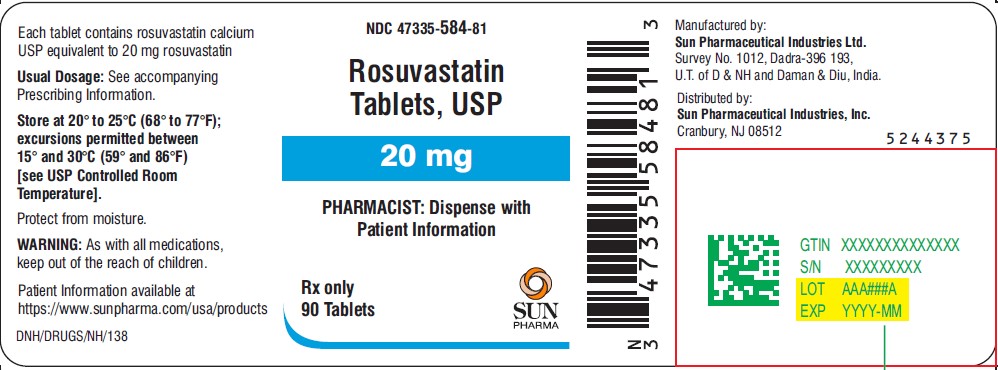
NDC 47335-584-81
Rosuvastatin Tablets
20 mg
PHARMACIST: Dispense with Patient Information
Rx only
90 Tablets
SUN PHARMA
PRINCIPAL DISPLAY PANEL
NDC 47335-585-81
Rosuvastatin Tablets
40 mg
PHARMACIST: Dispense with Patient Information
Rx only
90 Tablets
SUN PHARMA









