NDC Code(s) : 45802-946-94, 45802-946-96
Packager : Perrigo New York Inc
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Perrigo Sodium Sulfacetamide and SulfurSodium Sulfacetamide, Sulfur LOTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
Sodium Sulfacetamide 10% and Sulfur 5% Lotion
Rx Only
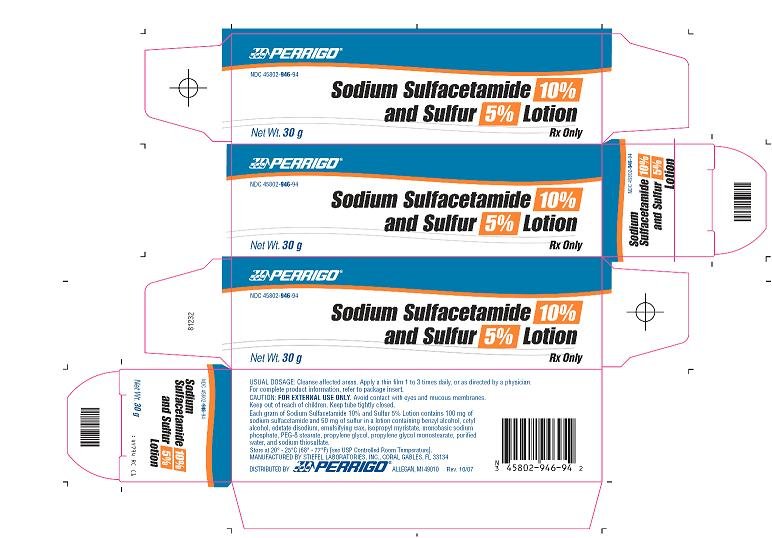 Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 30 g Carton
Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 30 g Carton
PRINCIPAL DISPLAY PANEL
Sodium Sulfacetamide 10% and Sulfur 5% Lotion
Rx Only
 Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 30 g Tube
Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 30 g Tube
PRINCIPAL DISPLAY PANEL
Sodium Sulfacetamide 10% and Sulfur 5% Lotion
Rx Only
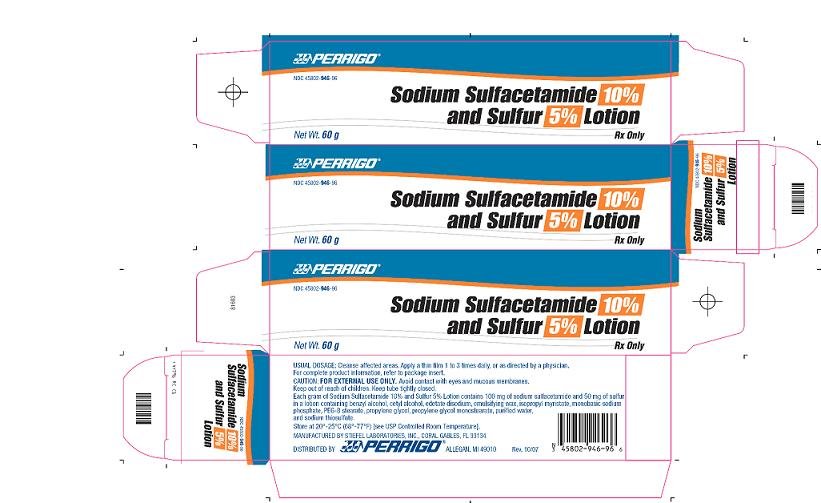 Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 60 g Carton
Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 60 g Carton
PRINCIPAL DISPLAY PANEL
Sodium Sulfacetamide 10% and Sulfur 5% Lotion
Rx Only
 Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 60 g Tube
Sodium Sulfacetamide 10% and Sulfur 5% Lotion - 60 g Tube








