NDC Code(s) : 43547-280-10, 43547-280-11, 43547-281-10, 43547-281-11, 43547-282-10, 43547-282-11
Packager : SOLCO HEALTHCARE US, LLC
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| EscitalopramEscitalopram TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| EscitalopramEscitalopram TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| EscitalopramEscitalopram TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| LABELER - SOLCO HEALTHCARE US, LLC(828343017) |
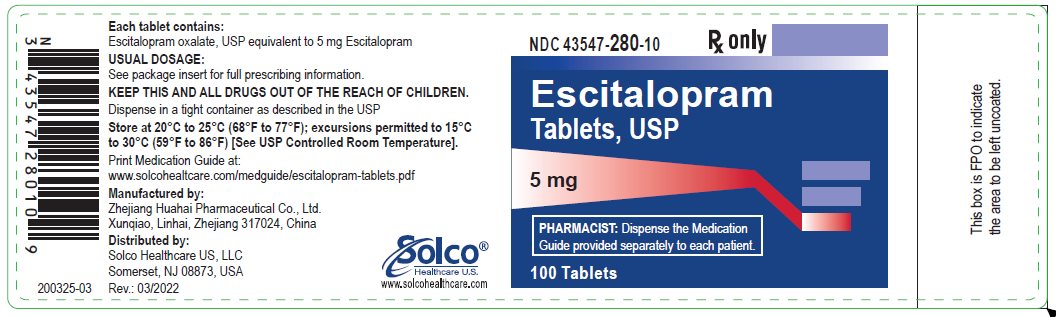
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - 5mg
NDC 43547-280-10
Escitalopram
Tablets, USP
5 mg
PHARMACIST: Dispense the Medication Guide
provided separately to each patient.
Rx only
100 Tablets
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - 10mg
NDC 43547-281-10
Escitalopram
Tablets, USP
10 mg
PHARMACIST: Dispense the Medication Guide
provided separately to each patient.
Rx only
100 Tablets

PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - 20mg
NDC 43547-282-10
Escitalopram
Tablets, USP
20 mg
PHARMACIST: Dispense the Medication Guide
provided separately to each patient.
Rx only
100 Tablets









