NDC Code(s) : 21922-009-09
Packager : Encube Ethicals Private Limited
Category : Human Prescription Drug Label
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Diclofenac Sodium Diclofenac Sodium Topical Gel, 1%, GEL | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LABELER - Encube Ethicals Private Limited(915834105) |
| REGISTRANT - Encube Ethicals Private Limited(915834105) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Encube Ethicals Private Limited | 725076298 | ANALYSIS(21922-009), MANUFACTURE(21922-009) | |
PRINCIPAL DISPLAY PANEL
See Medication Guide and Patient Instructions Inside of Carton
Carton Label
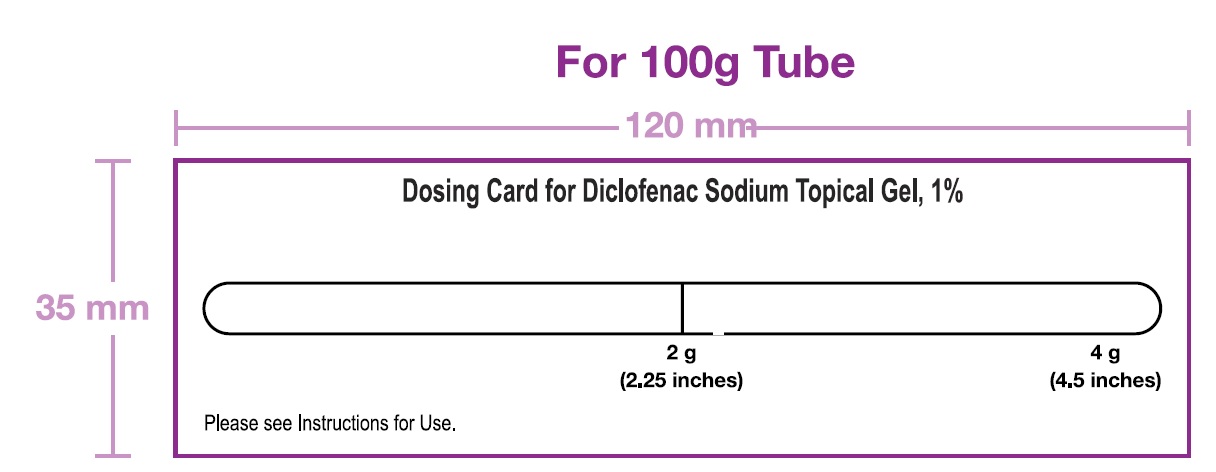
NDC 21922-009-09
Diclofenac sodium topical gel 1%
For Topical Use Only
Rx only
100 g
Use the Dosing Card Attached Inside Carton

Container Label
NDC 21922-009-09
Diclofenac sodium topical gel 1%
For Topical Use Only
Rx only
100 g
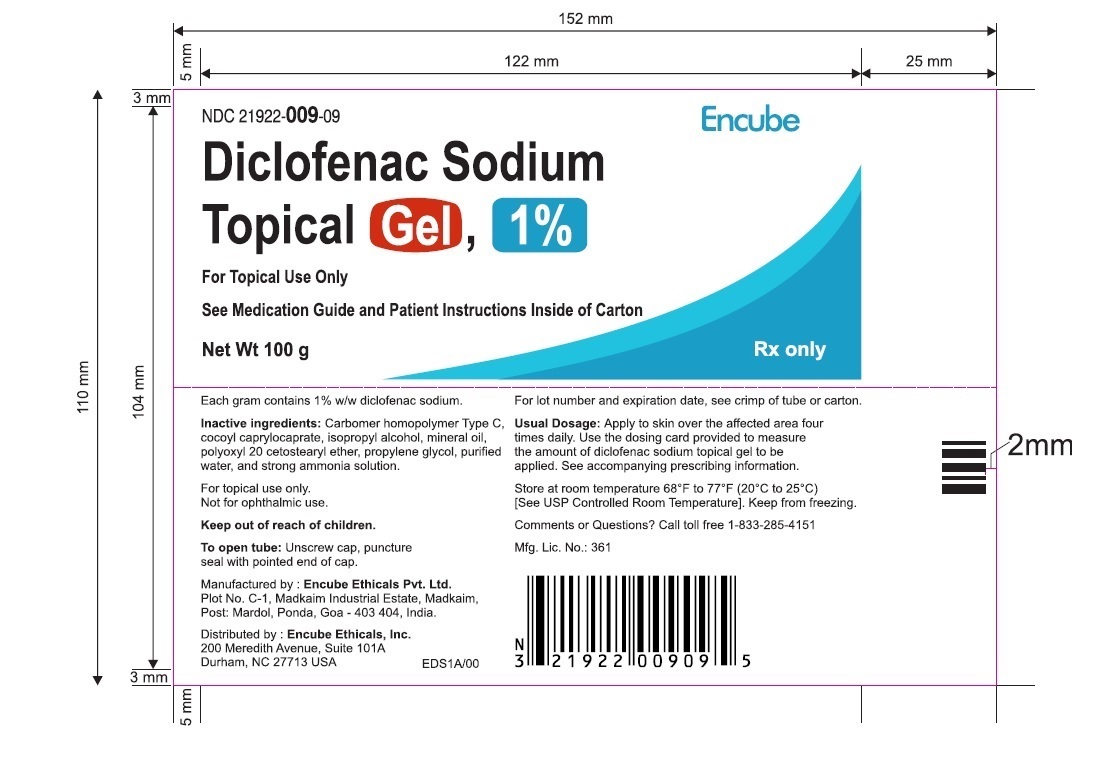
Dosing Card
NDC 21922-009-09
Diclofenac sodium topical gel 1%
For Topical Use Only
Rx only
100 g