NDC Code(s) : 13551-101-05, 13551-101-01
Packager : Avadel Pharmaceuticals (USA), Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Karbinal Carbinoxamine Maleate SUSPENSION, EXTENDED RELEASE | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
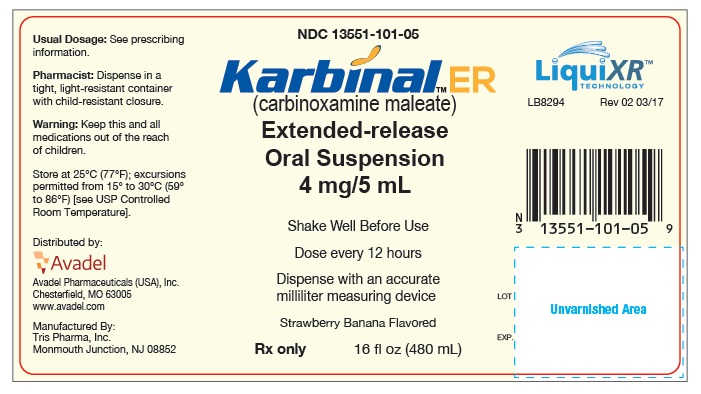
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 480 mL Bottle Label
NDC 13551-101-05
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx only
16 fl oz (480 mL)
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 480 mL Alternate Bottle Label
NDC 13551-101-05
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx only
16 fl oz (480 mL)

PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label
NDC 13551-101-01
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx only
1 fl oz (30 mL)
Physician Sample Not For Sale

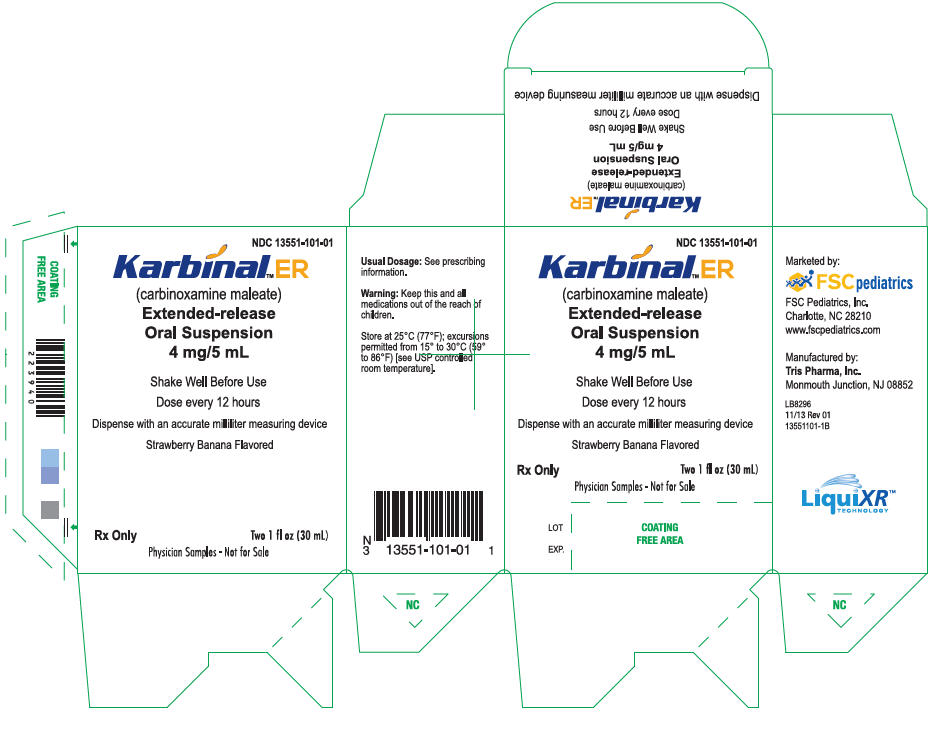
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 30 mL Carton Label
NDC 13551-101-01
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx only
Two 1 fl oz (30 mL)
PHYSICIAN SAMPLES - NOT FOR SALE
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 480 mL Alternate Bottle Label (FSC Laboratories)
NDC 13551-101-05
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx only
16 fl oz (480 mL)

PRINCIPAL DISPLAY PANEL
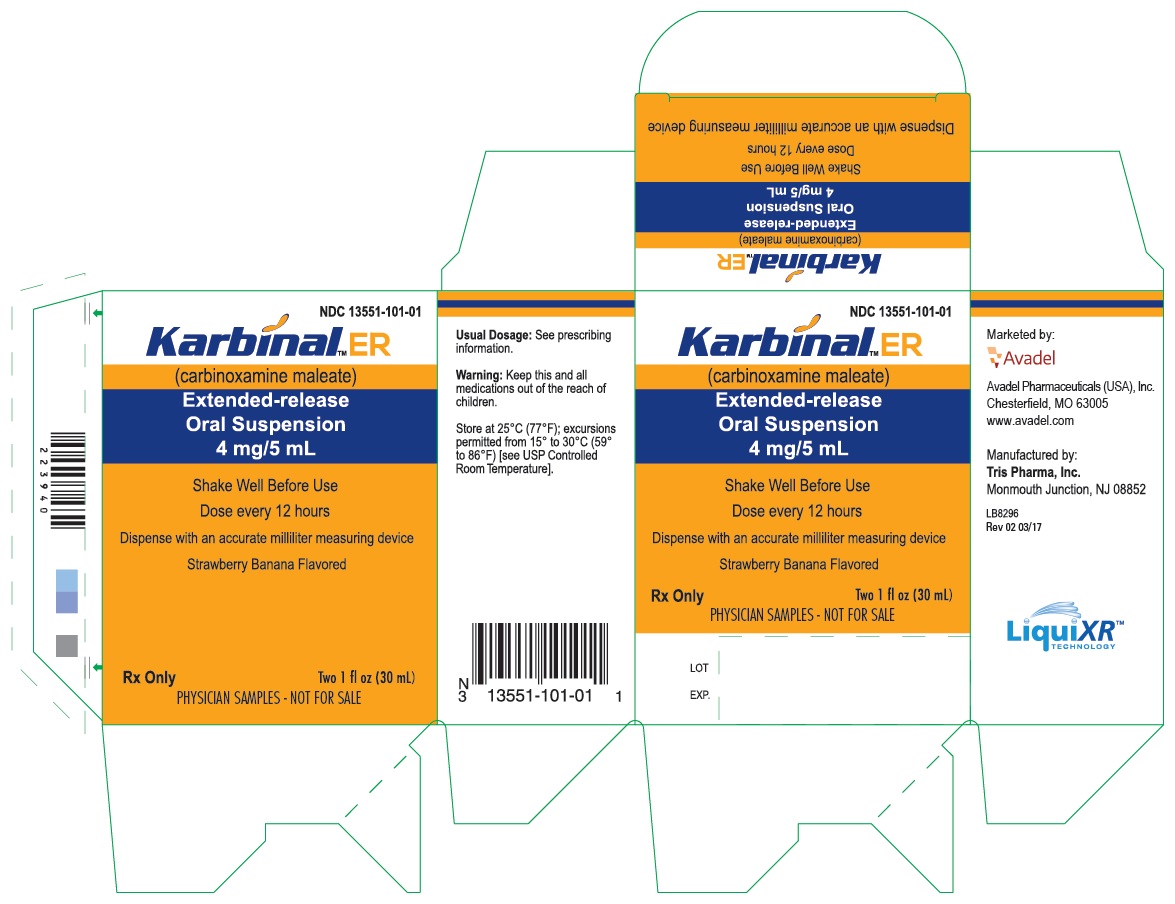
PRINCIPAL DISPLAY PANEL - 30 mL Alternate Carton Label (FSC Laboratories)
NDC 13551-101-01
Karbinal
™ ER
(carbinoxamine maleate)
Extended-release
Oral Suspension
4 mg/5 mL
Shake Well Before Use
Dose every 12 hours
Dispense with an accurate milliliter measuring device
Strawberry Banana Flavored
Rx Only
Two 1 fl oz (30 mL)
Physician Samples - Not for Sale