NDC Code(s) : 0591-2222-15, 0591-2223-15, 0591-2224-55
Packager : Actavis Pharma, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| CycloSPORINECycloSPORINE CAPSULE, LIQUID FILLED | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| CycloSPORINECycloSPORINE CAPSULE, LIQUID FILLED | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| CycloSPORINECycloSPORINE SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
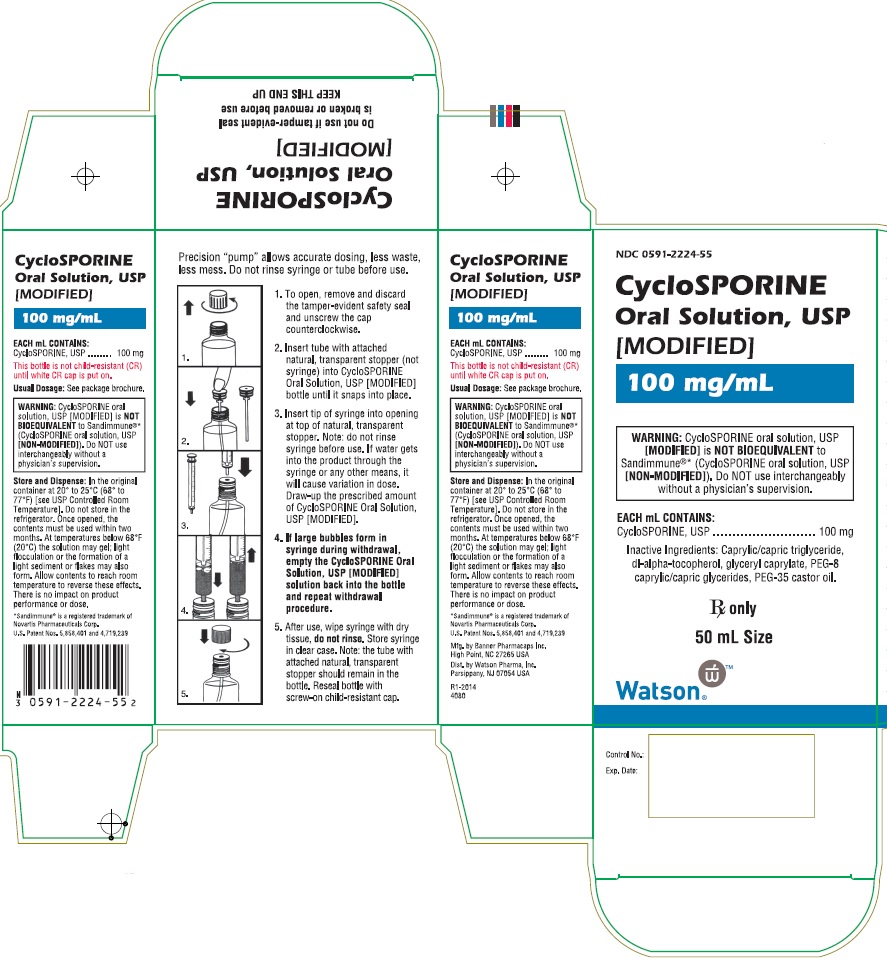
PRINCIPAL DISPLAY PANEL
NDC 0591-2222-15
CycloSPORINE Capsules, USP
[MODIFIED]
25 mg
WARNING: CycloSPORINE capsules, USP [MODIFIED] is NOT BIOEQUIVALENT to Sandimmune®* (CycloSPORINE capsules, USP [NON-MODIFIED]). Do NOT use interchangeably without a physician’s supervision.
This unit-dose package is not child-resistant.
Rx only
*Sandimmune® is a registered trademark of
Novartis Pharmaceuticals Corporation.
30 Soft Gelatin Capsules
Watson
®
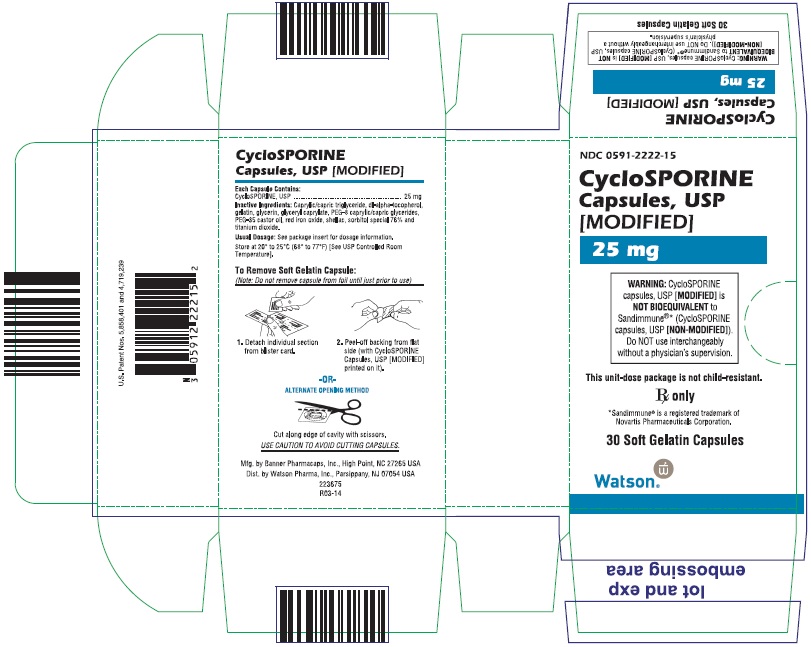
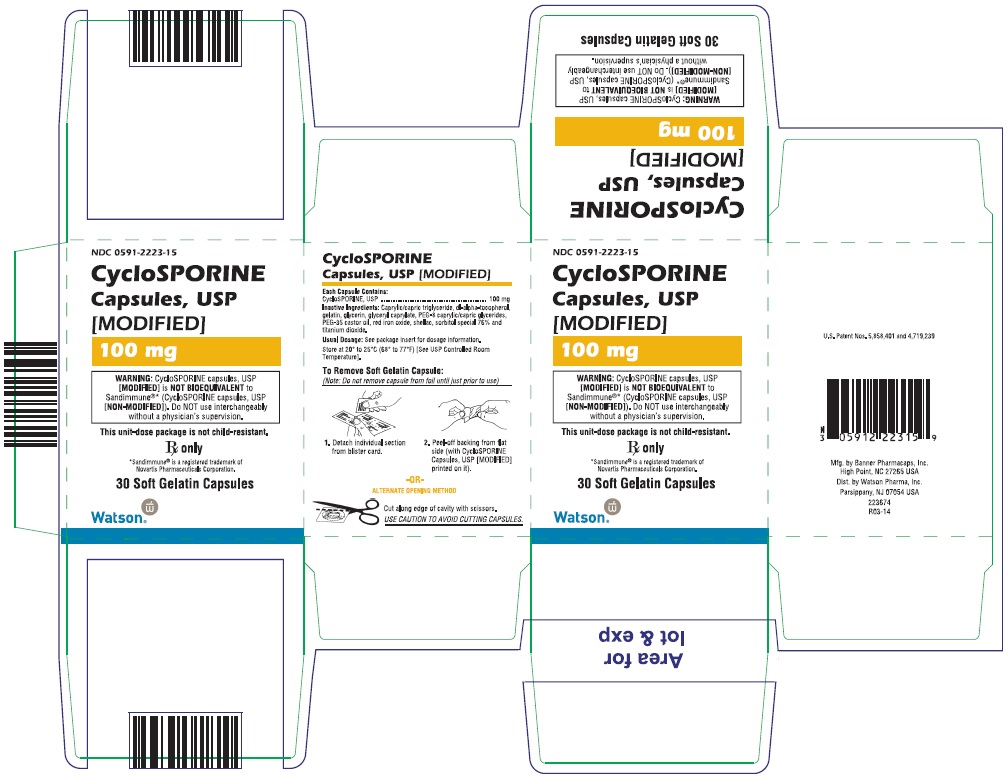
PRINCIPAL DISPLAY PANEL
NDC 0591-2223-15
CycloSPORINE Capsules, USP
[MODIFIED]
100 mg
WARNING: CycloSPORINE capsules, USP [MODIFIED] is NOT BIOEQUIVALENT to Sandimmune®* (CycloSPORINE capsules, USP [NON-MODIFIED]). Do NOT use interchangeably without a physician’s supervision.
This unit-dose package is not child-resistant.
Rx only
*Sandimmune® is a registered trademark of
Novartis Pharmaceuticals Corporation.
30 Soft Gelatin Capsules
Watson
®
PRINCIPAL DISPLAY PANEL
NDC 0591-2224-55
CycloSPORINE Oral Solution, USP
[MODIFIED]
100 mg/mL
WARNING: CycloSPORINE oral solution, USP [MODIFIED] is NOT BIOEQUIVALENT to Sandimmune®* (CycloSPORINE oral solution, USP [NON-MODIFIED]). Do NOT use interchangeably without a physician’s supervision.
EACH mL CONTAINS: CycloSPORINE, USP………………………………………………100 mg
Inactive Ingredients: Caprylic/capric triglyceride, dl-alpha-tocopherol, glyceryl caprylate, PEG-8 caprylic/capric glycerides, PEG-35 castor oil.
Rx only
50 mL Size
Watson
®