NDC Code(s) : 0527-2580-32, 0527-2580-37
Packager : Lannett Company, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Fluvastatin SodiumFluvastatin Sodium TABLET, FILM COATED, EXTENDED RELEASE | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| LABELER - Lannett Company, Inc.(002277481) |
| REGISTRANT - Lannett Company, Inc.(002277481) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Beijing Sciecure Pharmaceutical Co., Ltd. | 547707195 | manufacture(0527-2580) | |
PRINCIPAL DISPLAY PANEL
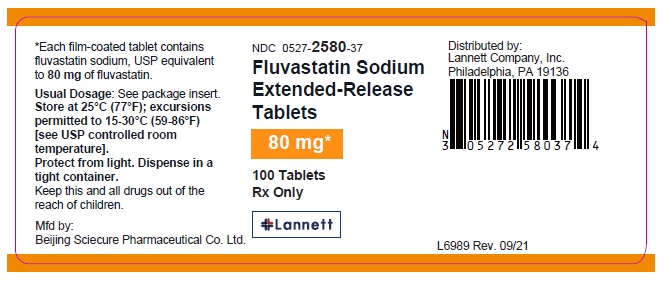
PRINCIPAL DISPLAY PANEL – 80 mg
NDC 0527- 2580-32
Fluvastatin Sodium
Extended-Release
Tablets
80 mg*
30 Tablets
Rx only
*Each film-coated tablet contains fluvastatin
sodium, USP equivalent to
80 mg of fluvastatin.
Usual Dosage: See package insert.
Store at 25°C (77°F); excursions permitted to
15-30°C (59-86°F)
[see USP controlled room temperature].
Protect from light. Dispense in a tight container.
Keep this and all drugs out of the reach of children.
Mfd by:
Beijing Sciecure Pharmaceutical Co. Ltd.
Distributed by:
Lannett Company, Inc.
Philadelphia, PA 19136
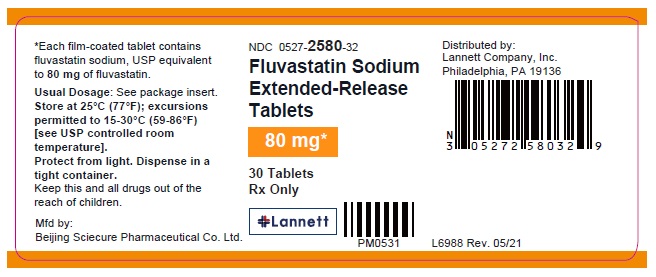
NDC 0527- 2580-37
100 Tablets
Rx only