NDC Code(s) : 0430-0112-24, 0430-0112-96, 0430-0115-95, 0430-0115-20, 0430-0114-20, 0430-0114-95
Packager : Allergan, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| DORYXdoxycycline hyclate TABLET, DELAYED RELEASE | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| DORYXdoxycycline hyclate TABLET, DELAYED RELEASE | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| DORYXdoxycycline hyclate TABLET, DELAYED RELEASE | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL

N 0430-0112-24
DORYX®
(Doxycycline Hyclate Delayed-Release Tablets, USP)
100 mg
Rx only
Each tablet contains specially coated pellets of doxycycline hyclate equivalent to 100 mg of doxycycline.
100 TABLETS

US Patent No. 6,958,161
PRINCIPAL DISPLAY PANEL
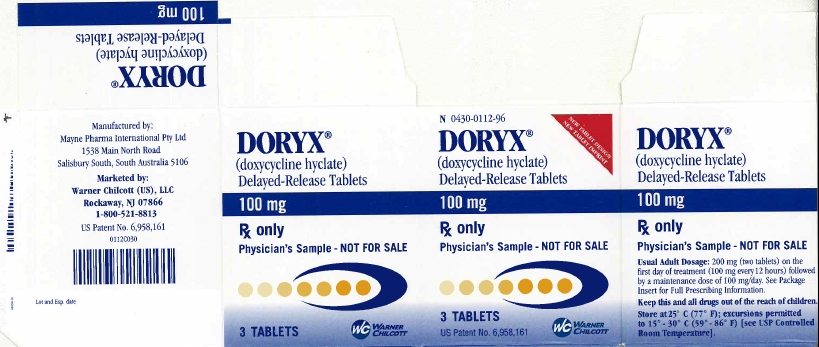
N 0430-0112-96
NEW TABLET DESIGN
NEW TABLET IMPRINT
DORYX®
(doxycycline hyclate)
Delayed-Release Tablets
100 mg
Rx only
Physician's Sample - NOT FOR SALE
3 Tablets
US Patent No. 6,958,161
PRINCIPAL DISPLAY PANEL
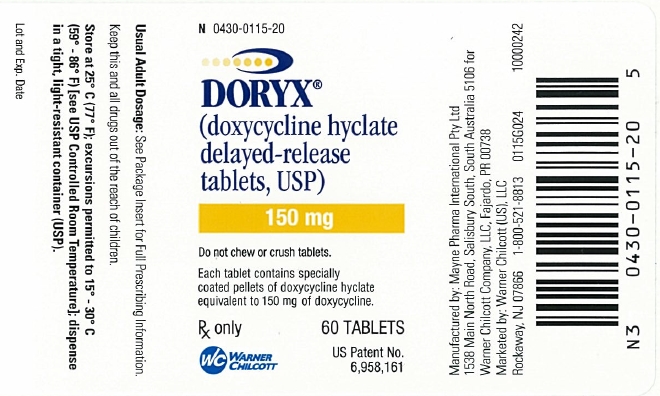
N 0430-0115-20
DORYX®
(doxycycline hyclate delayed-release tablets, USP)
150 mg
Do not chew or crush tablets.
Each tablet contains specially coated pellets of doxycycline hyclate equivalent to 150 mg of doxycycline.
Rx only
60 TABLETS
US Patent No. 6,958,161
PRINCIPAL DISPLAY PANEL

N 0430-0115-95
DORYX®
(doxycycline hyclate delayed-release tablets, USP)
150 mg per tablet
Take with adequate amounts of fluid
Do not chew or crush tablets
Rx only
Physician's Sample - NOT FOR SALE
3 TABLETS
US Patent No. 6,958,161
PRINCIPAL DISPLAY PANEL

N 0430-0114-20
NEW STRENGTH
DORYX®
(doxycycline hyclate delayed-release tablets)
200 mg
Rx only
Each tablet contains specially coated pellets of doxycycline hyclate equivalent to 200 mg of doxycycline.
60 TABLETS
To report SUSPECTED ADVERSE REACTIONS, contact Warner Chilcott at 1-800-521-8813 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
US Patent No. 6,958,161
PRINCIPAL DISPLAY PANEL
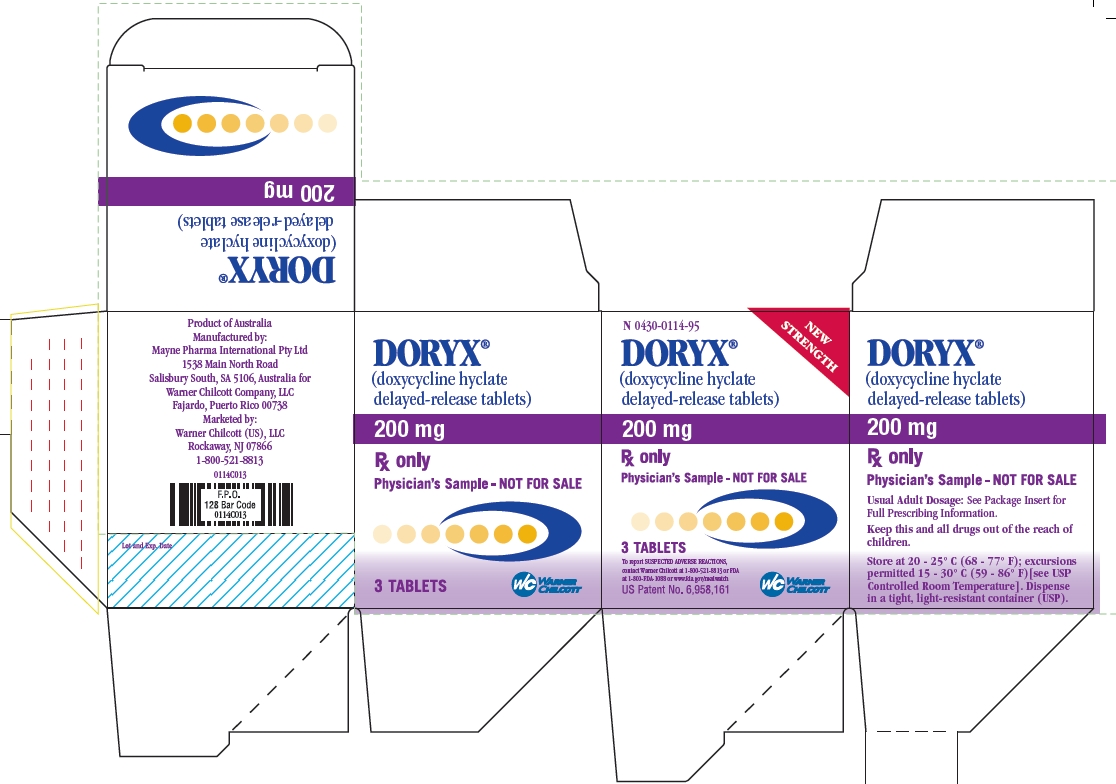
N 0430-0114-95
NEW STRENGTH
DORYX®
(doxycycline hyclate delayed-release tablets)
200 mg
Rx only
Physician's Sample - NOT FOR SALE
3 TABLETS
To report SUSPECTED ADVERSE REACTIONS, contact Warner Chilcott at 1-800-521-8813 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
US Patent No. 6,958,161








