NDC Code(s) : 0409-1181-30, 0409-1176-03, 0409-1176-30, 0409-1178-03, 0409-1178-30, 0409-1179-03, 0409-1179-30, 0409-1180-59, 0409-1180-69, 0409-1362-11, 0409-1362-01, 0409-1418-11, 0409-1418-01
Packager : Hospira, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : CII
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEMEROLMEPERIDINE HYDROCHLORIDE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LABELER - Hospira, Inc.(141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Hospira, Inc. | 030606222 | ANALYSIS(0409-1181, 0409-1176, 0409-1178, 0409-1179, 0409-1180, 0409-1362, 0409-1418), MANUFACTURE(0409-1181, 0409-1176, 0409-1178, 0409-1179, 0409-1180, 0409-1362, 0409-1418), PACK(0409-1181, 0409-1176, 0409-1178, 0409-1179, 0409-1180, 0409-1362, 0409-1418), LABEL(0409-1181, 0409-1176, 0409-1178, 0409-1179, 0409-1180, 0409-1362, 0409-1418) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Hospira, Inc. | 827731089 | ANALYSIS(0409-1181, 0409-1176, 0409-1178, 0409-1179, 0409-1180, 0409-1362, 0409-1418) | |
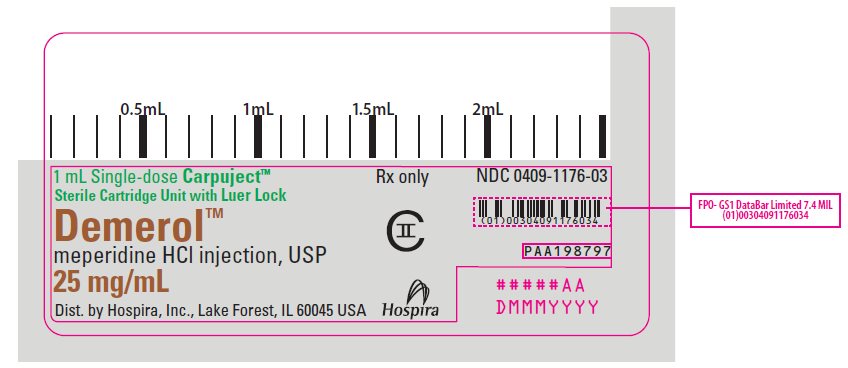
PRINCIPAL DISPLAY PANEL
1 mL Single-dose Carpuject™
Sterile Cartridge Unit with Luer Lock
Rx only
NDC 0409-1176-03
Demerol™
meperidine HCl injection, USP
25 mg/mL
CII
Dist. by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
PAA198797
#####AA
DMMMYYYY
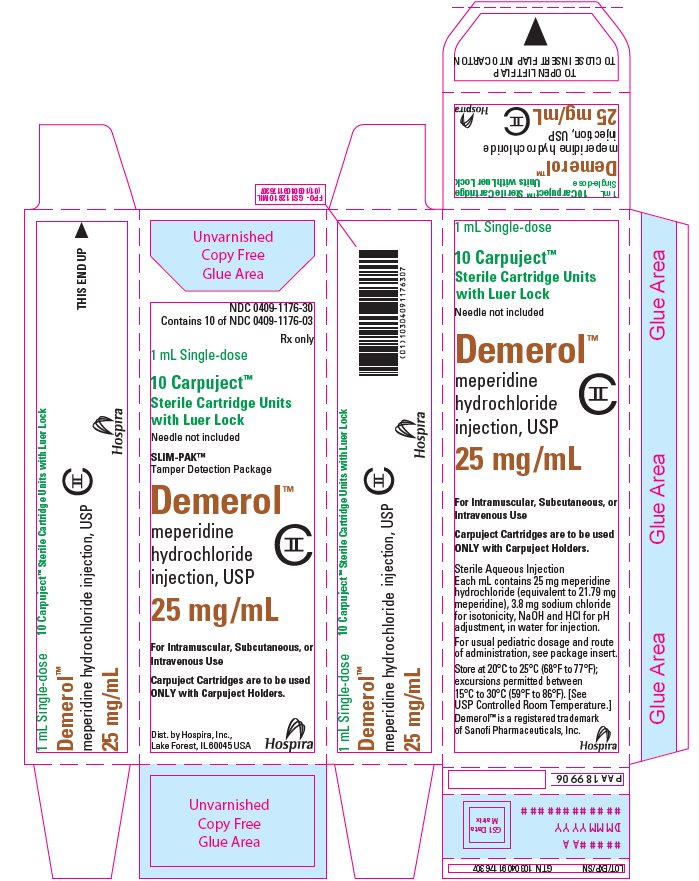
PRINCIPAL DISPLAY PANEL
NDC 0409-1176-30
Contains 10 of NDC 0409-1176-03
Rx only
1 mL Single-dose
10 Carpuject™
Sterile Cartridge Units
with Luer Lock
Needle not included
SLIM-PAK™
Tamper Detection Package
Demerol™
meperidine
hydrochloride
injection, USP
25 mg/mL
CII
For Intramuscular, Subcutaneous, or
Intravenous Use
Carpuject Cartridges are to be used
ONLY with Carpuject Holders.
Dist. by Hospira, Inc.,
Lake Forest, IL60045 USA
Hospira
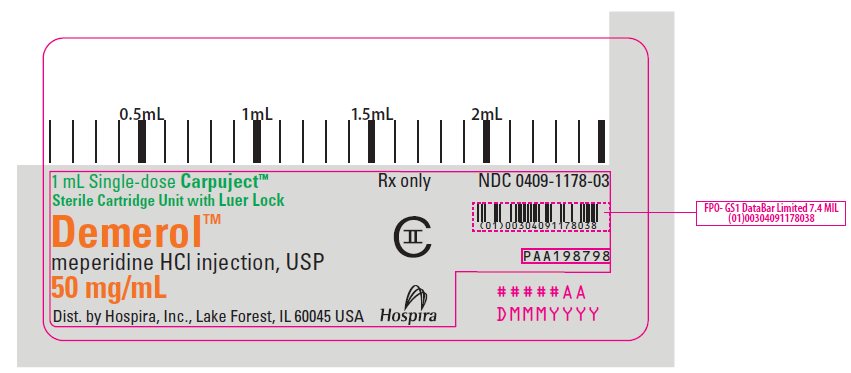
PRINCIPAL DISPLAY PANEL
1 mL Single-dose Carpuject™
Sterile Cartridge Unit with Luer Lock
Rx only
NDC 0409-1178-03
Demerol™
meperidine HCl injection, USP
50 mg/mL
CII
Dist. by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
PAA198798
#####AA
DMMMYYYY
PRINCIPAL DISPLAY PANEL
NDC 0409-1178-30
Contains 10 of NDC 0409-1178-03
Rx only
1 mL Single-dose
10 Carpuject™
Sterile Cartridge Units
with Luer Lock
Needle not included
SLIM-PAK™
Tamper Detection Package
Demerol™
meperidine
hydrochloride
injection, USP
50 mg/mL
CII
For Intramuscular, Subcutaneous, or
Intravenous Use
Carpuject Cartridges are to be used
ONLY with Carpuject Holders.
Dist. by Hospira, Inc.
Lake Forest, IL 60045 USA
Hospira
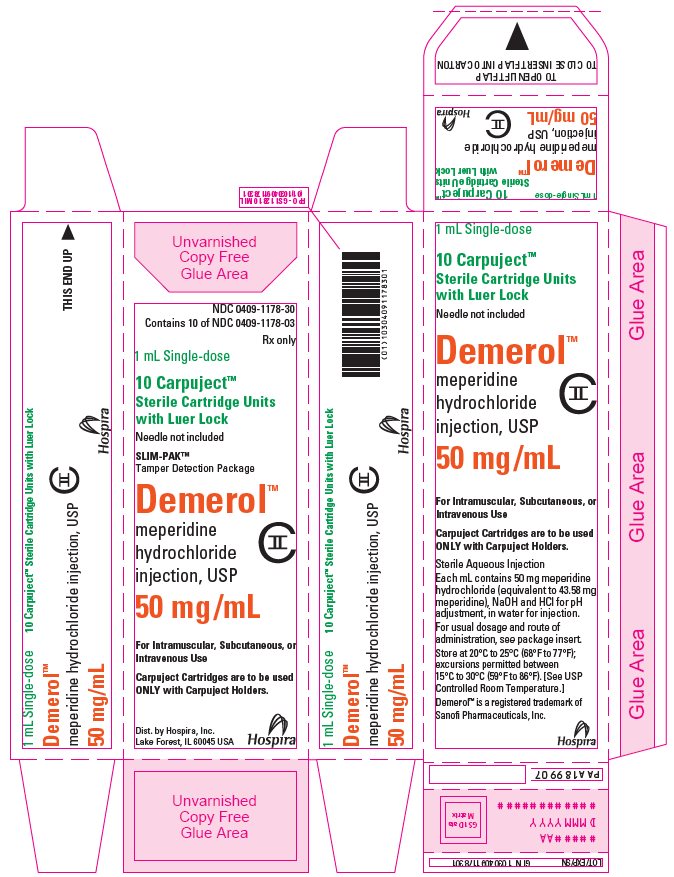
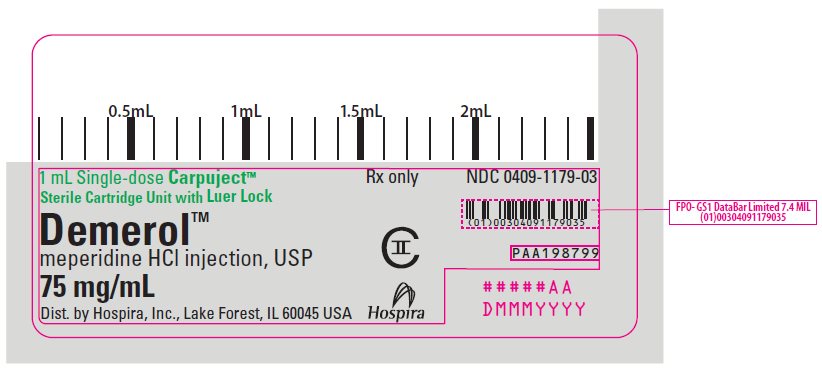
PRINCIPAL DISPLAY PANEL
1 mL Single-dose Carpuject™
Sterile Cartridge Unit with Luer Lock
Rx only
NDC 0409-1179-03
Demerol™
meperidine HCl injection, USP
75 mg/mL
CII
Dist. by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
PAA198799
#####AA
DMMMYYYY
PRINCIPAL DISPLAY PANEL
NDC 0409-1179-30
Contains 10 of NDC 0409-1179-03
Rx only
1 mL Single-dose
10 Carpuject™
Sterile Cartridge Units
with Luer Lock
Needle not included
SLIM-PAK™
Tamper Detection Package
Demerol™
meperidine
hydrochloride
injection, USP
75 mg/mL
CII
For Intramuscular, Subcutaneous, or
Intravenous Use
Carpuject Cartridges are to be used
ONLY with Carpuject Holders.
Dist. by Hospira, Inc.
Lake Forest, IL 60045 USA
Hospira
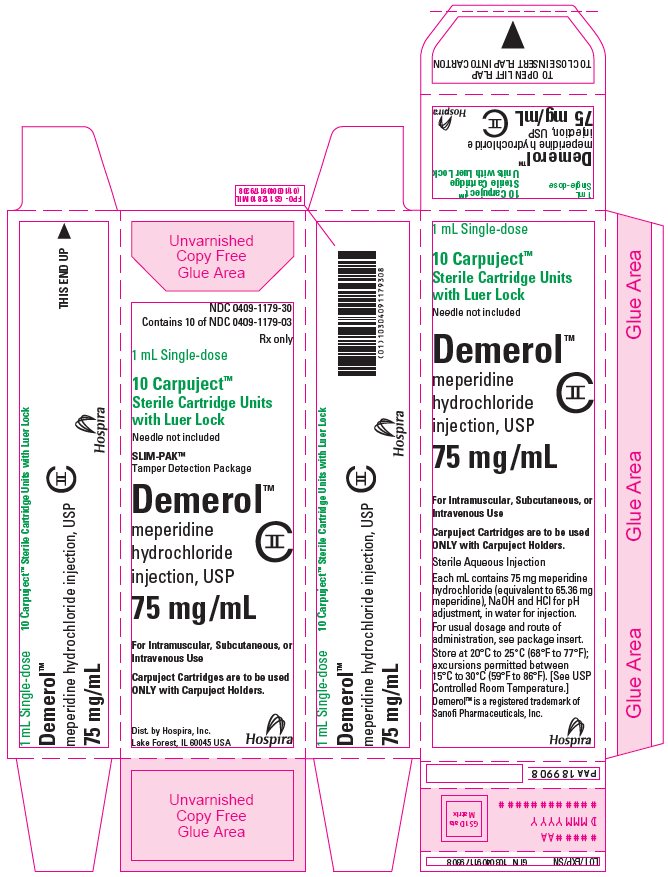
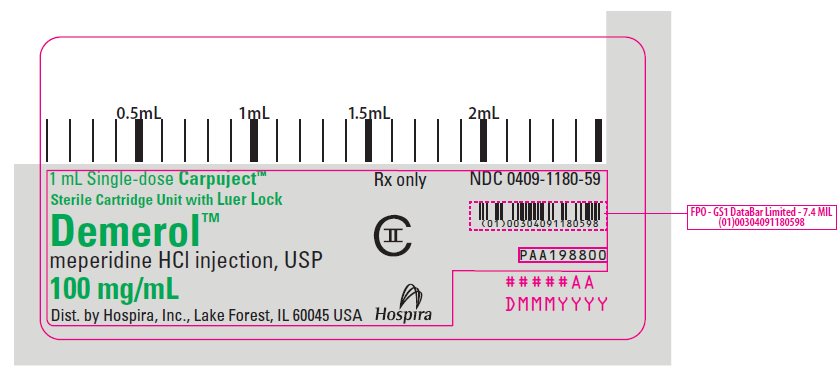
PRINCIPAL DISPLAY PANEL
1 mL Single-dose Carpuject™
Sterile Cartridge Unit with Luer Lock
Rx only
NDC 0409-1180-59
Demerol™
meperidine HCl injection, USP
100 mg/mL
CII
Dist. by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
PAA198800
#####AA
DMMMYYYY
PRINCIPAL DISPLAY PANEL
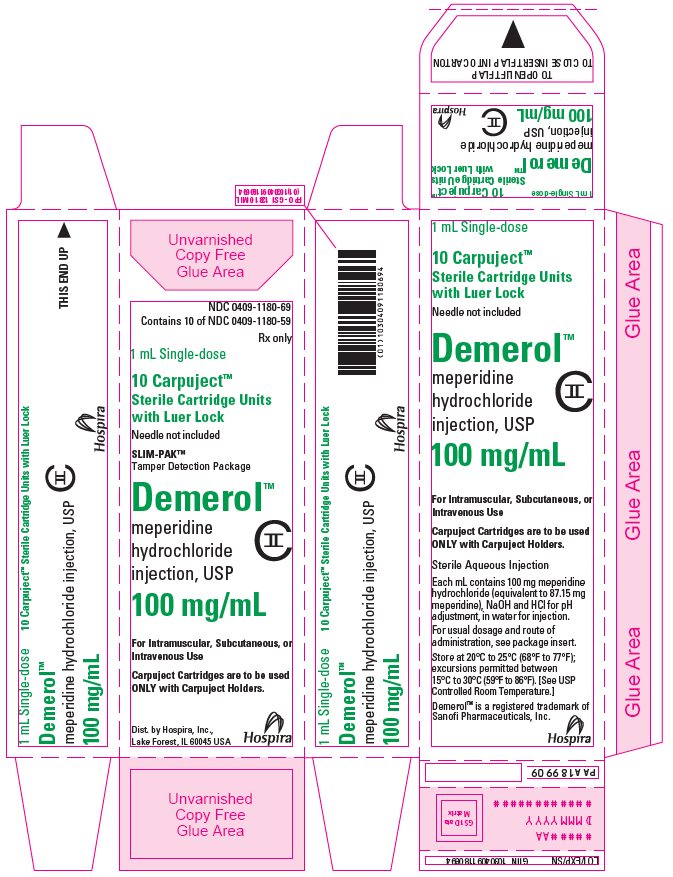
NDC 0409-1180-69
Contains 10 of NDC 0409-1180-59
Rx only
1 mL Single-dose
10 Carpuject™
Sterile Cartridge Units
with Luer Lock
Needle not included
SLIM-PAK™
Tamper Detection Package
Demerol™
meperidine
hydrochloride
injection, USP
100 mg/mL
CII
For Intramuscular, Subcutaneous, or
Intravenous Use
Carpuject Cartridges are to be used
ONLY with Carpuject Holders.
Dist. by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
PRINCIPAL DISPLAY PANEL
30 mL
Multiple-dose vial
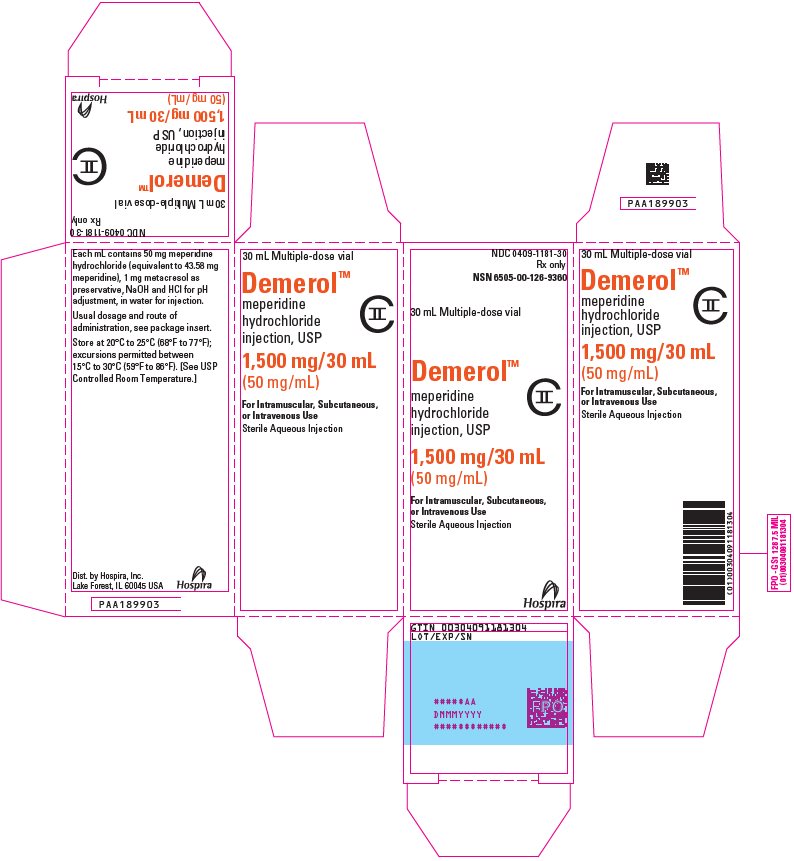
NDC 0409-1181-30
Rx only
Sterile Aqueous Injection
Demerol™
meperidine hydrochloride injection, USP
1500 mg/30 mL (50 mg/mL)
CII
For Intramuscular, Subcutaneous, or Intravenous Use
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL
NDC 0409-1181-30
Rx only
NSN 6505-00-126-9360
30 mL Multiple-dose vial
Demerol™
meperidine
hydrochloride
injection, USP
1500 mg/30 mL
(50 mg/mL)
CII
For Intramuscular, Subcutaneous,
or Intravenous Use
Sterile Aqueous Injection
Hospira
PRINCIPAL DISPLAY PANEL
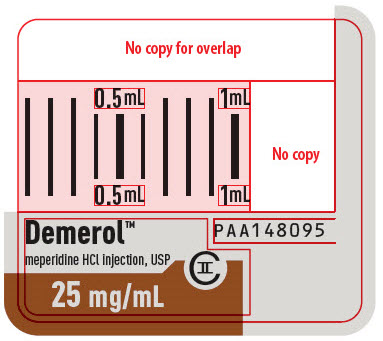
Demerol™
meperidine HCl injection, USP
CII
25 mg/mL
PAA148095
PRINCIPAL DISPLAY PANEL
TWIST & PULL
Tamper Seal
NDC 0409-1362-11
CII
Demerol™
meperidine HCl injection, USP
25 mg/mL
Intramuscular,
Subcutaneous, or
Intravenous Use
Rx only
1 mL Single-dose syringe
PAA148096
LOT #####AA
EXP DMMMYYYY

PRINCIPAL DISPLAY PANEL
NDC 0409-1362-01
Demerol™
CII
meperidine hydrochloride
injection, USP
25 mg / mL
For Intramuscular, Subcutaneous, or Intravenous Use
10 NexJect™ 1 mL Single-dose syringes with luer lock
Rx only
Needle not included
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between
15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.]
Sterile Aqueous Injection
Each mL contains 25 mg meperidine hydrochloride (equivalent to 21.79 mg
meperidine), 3.8 mg sodium chloride for isotonicity, NaOH and HCl for pH
adjustment, in water for injection.
For usual pediatric dosage and route of administration, see package insert.
Demerol™ is a registered trademark of Sanofi Pharmaceuticals, Inc.
PAA189910
Distributed by Hospira, Inc.
Lake Forest, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL
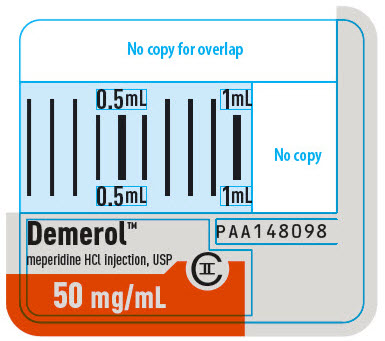
Demerol™
meperidine HCl injection, USP
CII
50 mg/mL
PAA148098
PRINCIPAL DISPLAY PANEL
TWIST & PULL
Tamper Seal
NDC 0409-1418-11
CII
Demerol™
meperidine HCl injection, USP
50 mg/mL
Intramuscular,
Subcutaneous, or
Intravenous Use
Rx only
1 mL Single-dose syringe
PAA148099
LOT #####AA
EXP DMMMYYYY

PRINCIPAL DISPLAY PANEL
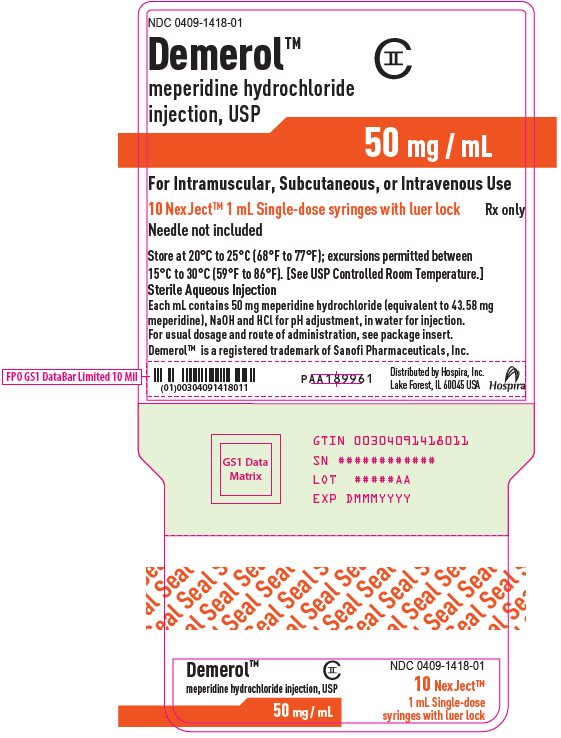
NDC 0409-1418-01
Demerol™
CII
meperidine hydrochloride
injection, USP
50 mg / mL
For Intramuscular, Subcutaneous, or Intravenous Use
10 NexJect™ 1 mL Single-dose syringes with luer lock
Rx only
Needle not included
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between
15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.]
Sterile Aqueous Injection
Each mL contains 50 mg meperidine hydrochloride (equivalent to 43.58 mg
meperidine), NaOH and HCl for pH adjustment, in water for injection.
For usual dosage and route of administration, see package insert.
Demerol™ is a registered trademark of Sanofi Pharmaceuticals, Inc.
PAA189961
Distributed by Hospira, Inc.
Lake Forest, IL 60045 USA
Hospira