NDC Code(s) : 0310-6540-01, 0310-6540-04, 0310-6540-85
Packager : AstraZeneca Pharmaceuticals LP
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| BYDUREON BCiseexenatide INJECTION, SUSPENSION, EXTENDED RELEASE | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| LABELER - AstraZeneca Pharmaceuticals LP(054743190) |
| REGISTRANT - AstraZeneca PLC(230790719) |
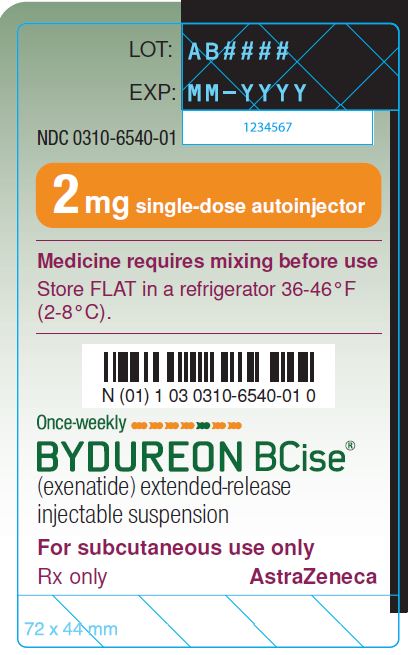
PRINCIPAL DISPLAY PANEL
NDC 0310-6540-01
2 mg single-dose autoinjector
Medication requires mixing before use
Store FLAT in a refrigerator 36-46°F
(2-8°C)
Once-weekly
BYDUREON BCise®
(exenatide) extended-release
injectable suspension
For subcutaneous use only
Rx only AstraZeneca
PRINCIPAL DISPLAY PANEL
NDC 0310-6540-04
Once-Weekly
BYDUREON BCise® 2 mg single-dose autoinjector
(exenatide) extended-release
injectable suspension
For subcutaneous use only
Dispense with enclosed Medication Guide
to each patient.
Store FLAT in refrigerator 36-46°F (2-8°C).
Total Quantity: 4 x Single-Dose Autoinjectors
Use one autoinjector every week
Each autoinjector delivers a 2 mg dose
Rx only AstraZeneca









