NDC Code(s) : 0169-3201-11, 0169-3201-90, 0169-3204-15, 0169-3204-90, 0169-3204-97, 0169-3205-11, 0169-3205-15, 0169-3205-91, 0169-3205-95, 0169-3206-11, 0169-3206-15, 0169-3206-91, 0169-3206-95
Packager : Novo Nordisk
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Fiaspinsulin aspart injection INJECTION, SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Fiaspinsulin aspart injection INJECTION, SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Fiaspinsulin aspart injection INJECTION, SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Fiaspinsulin aspart INJECTION, SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| LABELER - Novo Nordisk(622920320) |
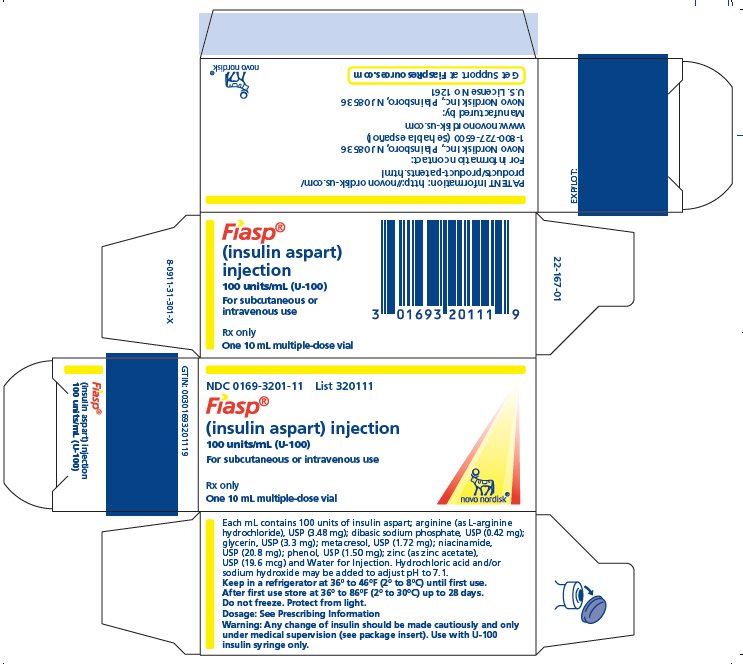
PRINCIPAL DISPLAY PANEL
NDC 0169-3201-11 List 320111
FIASP ®
(insulin apart) injection
100 units/mL (U-100)
For subcutaneous or intravenous use
- Rx only One 10 mL multi-dose vial
PRINCIPAL DISPLAY PANEL
NDC 0169-3204-15 List 320415
FIASP ®FlexTouch ®
(insulin aspart) injection
For Single-Patient Use Only
100 units/mL (U-100)
For subcutaneous use only
Rx Only
Recommended for use with NovoFine® or
NovoFine® Plus disposable needles.
Keep in a refrigerator at 36° to 46°F (2° to 8°C) until first use.
After first use store at 36° to 86°F (2° to 30°C) up to 28 days.
Do not freeze.
Protect from light.
Dispense in this sealed carton.
5×3 mL Prefilled Pens

PRINCIPAL DISPLAY PANEL
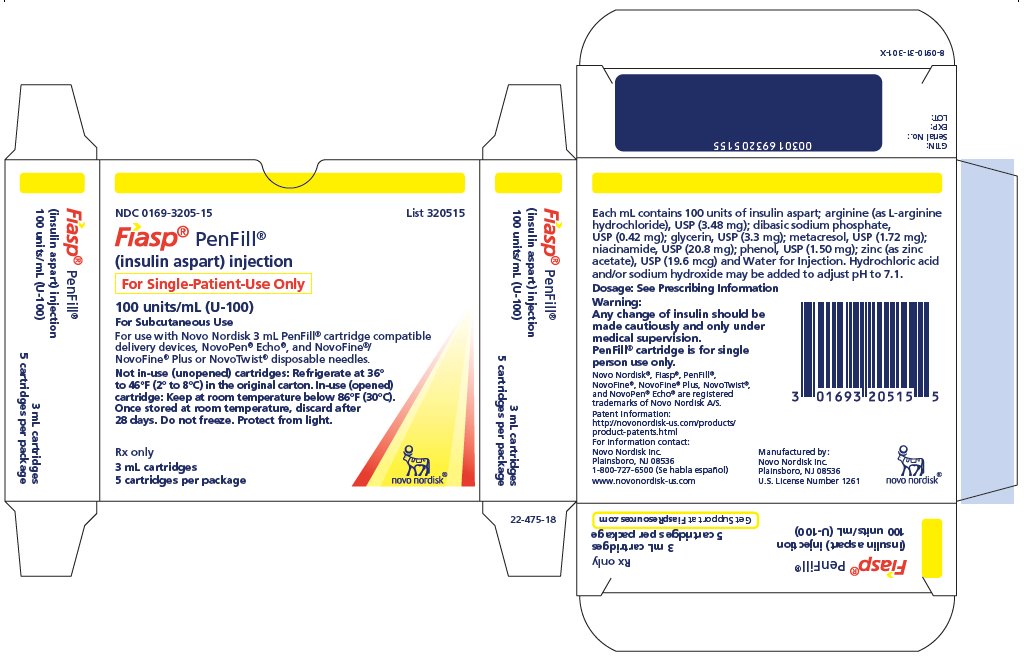
- NDC 0169-3205-15 List 320515
FIASP® PenFill®
(insulin aspart) injection
For Single-Patient Use Only
100 units/mL (U-100)
For Subcutaneous Use
For use with Novo Nordisk 3 mL PenFill® cartridge compatible delivery devices, NovoPen® Echo®, and NovoFine®/NovoFine® Plus disposable needles.
Not in-use (unopened) cartridges: Refrigerate at 36°F to 46°F (2°C to 8°C) in the original carton. In-use (opened) cartridge: Keep at room temperature below 86°F (30°C). Once stored at room temperature, discard after 28 days. Do not freeze. Protect from light.
Rx only
3 mL cartridges
5 cartridges per package
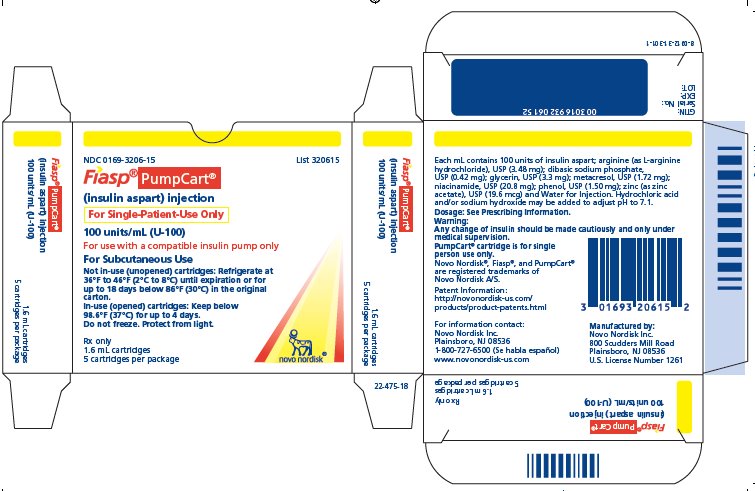
PRINCIPAL DISPLAY PANEL
NDC 0169-3206-15 List 320615
- Fiasp ® PumpCart®
(insulin aspart) injection
For Single-Patient Use Only
100 units/mL (U-100)
For use with a compatible insulin pump only
For Subcutaneous Use
Not in-use (unopened) cartridges: Refrigerate at 36°F to 46°F (2°C to 8°C) until expiration or for up to 18 days below 86°F (30°C) in the original carton.
In-use (opened) cartridges: Keep below 98.6°F (37°C) for up to 4 days.
Do not freeze. Protect from light.
Rx only
1.6 mL cartridges
5 cartridges per package