NDC Code(s) : 0074-3224-14, 0074-3224-56, 0074-3271-14, 0074-3271-56, 0074-3197-16, 0074-3197-30, 0074-3282-14, 0074-3282-56, 0074-3239-14, 0074-3239-56
Packager : AbbVie Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| ModeribaRibavirin KIT | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ModeribaRibavirin KIT | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ModeribaRibavirin TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| ModeribaRibavirin TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| ModeribaRibavirin TABLET, FILM COATED | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
NDC 0074-3197-16
Moderiba™
(ribavirin, USP) Tablets
200 mg
168 Tablets
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
Rx only abbvie

NDC 0074-3224-14
Moderiba™ 600 Dose Pack
(ribavirin, USP) Tablets
200 mg / 400 mg
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
Contains 7 ribavirin tablets (200 mg each) and 7 ribavirin tablets (400 mg each)
AM Dose: 200 mg / PM Dose: 400 mg
Patient instructions for each daily dose:
AM – In the morning, push one ribavirin tablet, 200 mg through the cardboard backing.
PM – In the evening, push one ribavirin tablet, 400 mg through the cardboard backing.
Please read the enclosed Medication Guide
Rx only abbvie

NDC 0074-3224-56
Moderiba™ 600 Dose Pack
(ribavirin, USP) Tablets
200 mg / 400 mg
56 Tablets in 4 Unit Dose Packs
Each Unit Dose Pack contains
7 ribavirin tablets (200 mg each) and
7 ribavirin tablets (400 mg each)
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.

NDC 0074-3239-14
Moderiba™ 800 Dose Pack
(ribavirin, USP) Tablets
400 mg / 400 mg
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
Contains 14 ribavirin tablets (400 mg each)
AM Dose: 400 mg / PM Dose: 400 mg
Patient instructions for each daily dose:
AM – In the morning, push one ribavirin tablet, 400 mg through the cardboard backing.
PM – In the evening, push one ribavirin tablet, 400 mg through the cardboard backing.
Please read the enclosed Medication Guide
Rx only abbvie

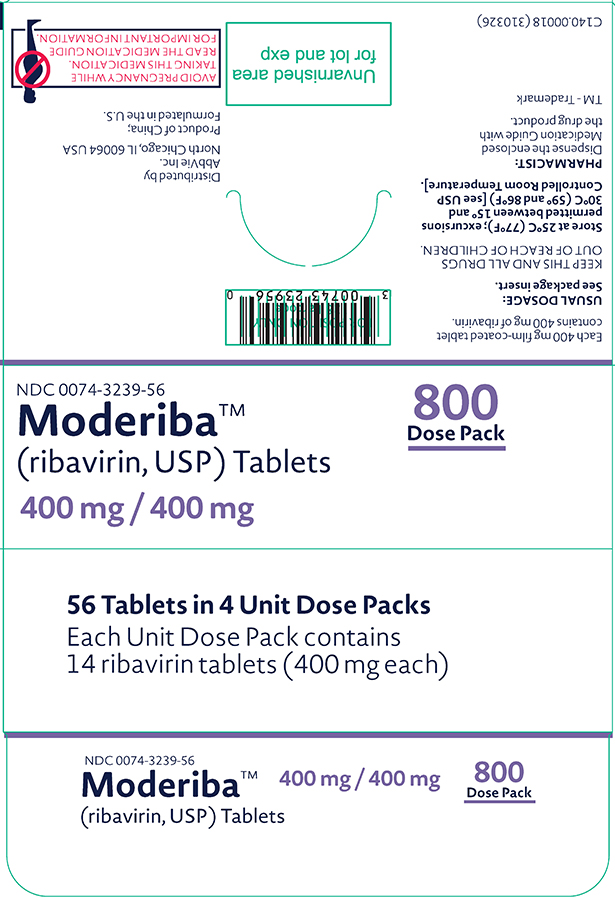
NDC 0074-3239-56
Moderiba™ 800 Dose Pack
(ribavirin, USP) Tablets
400 mg / 400 mg
56 Tablets in 4 Unit Dose Packs
Each Unit Dose Pack contains
14 ribavirin tablets (400 mg each)
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
NDC 0074-3271-14
Moderiba™ 1000 Dose Pack
(ribavirin, USP) Tablets
600 mg / 400 mg
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
Contains 7 ribavirin tablets (600 mg each) and 7 ribavirin tablets (400 mg each)
AM Dose: 600 mg / PM Dose: 400 mg
Patient instructions for each daily dose:
AM - In the morning, push one ribavirin tablet, 600 mg through the cardboard backing.
PM - In the evening, push one ribavirin tablet, 400 mg through the cardboard backing.
Please read the enclosed Medication Guide
Rx only abbvie

NDC 0074-3271-56
Moderiba™ 1000 Dose Pack
(ribavirin, USP) Tablets
600 mg / 400 mg
56 Tablets in 4 Unit Dose Packs
Each Unit Dose Pack contains
7 ribavirin tablets (600 mg each) and
7 ribavirin tablets (400 mg each)
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.

NDC 0074-3282-14
Moderiba™ 1200 Dose Pack
(ribavirin, USP) Tablets
600 mg / 600 mg
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.
Contains 14 ribavirin tablets (600 mg each)
AM Dose: 600 mg / PM Dose: 600 mg
Patient instructions for each daily dose:
AM - In the morning, push one ribavirin tablet, 600 mg through the cardboard backing.
PM - In the evening, push one ribavirin tablet, 600 mg through the cardboard backing.
Please read the enclosed Medication Guide
Rx only abbvie

NDC 0074-3282-56
Moderiba™ 1200 Dose Pack
(ribavirin, USP) Tablets
600 mg / 600 mg
56 Tablets in 4 Unit Dose Packs
Each Unit Dose Pack contains
14 ribavirin tablets (600 mg each)
AVOID PREGNANCY WHILE TAKING THIS MEDICATION. READ THE MEDICATION GUIDE FOR IMPORTANT INFORMATION.









