

Synopsis
Synopsis
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
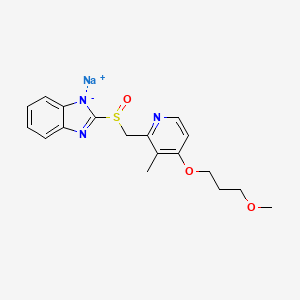

1. 1h-benzimidazole, 2-(((4-(3-methoxypropoxy)-3-methyl-2-pyridinyl)methyl)sulfinyl)-, Sodium Salt
2. 2-((4-(3-methoxypropoxy)-3-methylpyridin-2-yl)methylsulfinyl)-1h-benzimidazole
3. Aciphex
4. Dexrabeprazole
5. E 3810
6. E3810
7. Ly 307640
8. Ly-307640
9. Ly307640
10. Pariet
11. Rabeprazole
12. Sodium, Rabeprazole
1. 117976-90-6
2. Rebeprazole Sodium
3. Pariet
4. Aciphex
5. Rabeprazole Sodium Salt
6. Dexrabeprazole Sodium
7. Rabeprazole (sodium)
8. (s)-rabeprazole Sodium Salt
9. Aciphex Sprinkle
10. Ly307640 Sodium
11. Sodium Rabeprazole
12. Rabeprazole Sodium [usan]
13. Idiazole
14. Rabeprazole Na
15. Chebi:8769
16. 171440-19-0
17. Ly-307640
18. E3810
19. E-3810 Sodium
20. Sodium;2-[[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methylsulfinyl]benzimidazol-1-ide
21. Ly-307640 Sodium
22. 3l36p16u4r
23. 1h-benzimidazole, 2-(((4-(3-methoxypropoxy)-3-methyl-2-pyridinyl)methyl)sulfinyl)-, Sodium Salt
24. 2-(((4-(3-methoxypropoxy)-3-methyl-2-pyridyl)methyl)sulfinyl)benzimidazole Sodium Salt
25. Nsc-759270
26. Dsstox_cid_24205
27. Dsstox_rid_80118
28. Dsstox_gsid_44205
29. 1h-benzimidazole, 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]methyl]sulfinyl]-, Sodium Salt (1:1)
30. Rabicip
31. Pepcia
32. Aciphex Sodium
33. Ly 307640 Sodium
34. Habeprazole Sodium
35. Sodium 2-((4-(3-methoxypropoxy)-3-methylpyridin-2-yl)methylsulfinyl)benzo[d]imidazol-1-ide
36. Sodium 2-({[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methyl}sulfinyl)benzimidazol-1-ide
37. Smr000550493
38. Cas-117976-90-6
39. Ncgc00159518-02
40. E 3810
41. Pariete
42. Unii-3l36p16u4r
43. Pariprazole Sodium
44. Dexrabeprazolesodium
45. Rebeprazole Sodium;
46. Aciphex (tn)
47. Pariet (tn)
48. Rebeprazole Sodium Salt
49. Rabeprazole Sodium- Bio-x
50. Mls001165734
51. Mls006010621
52. Schembl140494
53. Rabeprazole Na [vandf]
54. Chembl1200930
55. Dtxsid3044205
56. Rabeprazole Sodium [jan]
57. Hy-b0656a
58. Rabeprazole Sodium (jp17/usp)
59. Ex-a176
60. Hms2093h10
61. Hms2231e04
62. Hms3373b05
63. Rabeprazole Sodium [mart.]
64. Bcp06639
65. Rabeprazole Sodium [usp-rs]
66. Rabeprazole Sodium [who-dd]
67. Tox21_111736
68. Tox21_302317
69. Ac-715
70. Bdbm50247881
71. Mfcd02092688
72. Rabeprazole Sodium Salt [mi]
73. S4665
74. Rabeprazole Sodium, >=98% (hplc)
75. Akos015895734
76. Akos015962116
77. Akos025310160
78. Tox21_111736_1
79. Am81231
80. Ccg-213574
81. Ccg-268423
82. Nsc 759270
83. Rabeprazole Sodium [orange Book]
84. Ncgc00159518-05
85. Ncgc00255838-01
86. Rabeprazole Sodium [ep Monograph]
87. As-13338
88. Br167318
89. Rabeprazole Sodium [usp Monograph]
90. Ft-0631090
91. Ft-0689461
92. R0115
93. C07865
94. D00724
95. F17418
96. 976r906
97. Sr-01000799133
98. J-010726
99. Q-201655
100. Sr-01000799133-2
101. Q27108142
102. [2-[[4-(3-methoxypropoxy)-3-methyl-2-pyridyl]methylsulfinyl]benzimidazol-1-yl]sodium
103. Sodium;2-[[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methylsulfinyl]-1h-benzimidazole
104. 1h-benzimidazole,2-[[[4-(3-methoxypropoxy)- 3-methyl-2-pyridinyl]methyl]sulfinyl]-,sodium Salt
105. 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]methyl]sulfinyl]-1h-benzimidazole, Sodium Salt
106. Sodium 2-(((4-(3-methoxypropoxy)-3-methylpyridin-2-yl)methyl)sulfinyl)benzo[d]imidazol-1-ide
| Molecular Weight | 381.4 g/mol |
|---|---|
| Molecular Formula | C18H20N3NaO3S |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 8 |
| Exact Mass | 381.11230696 g/mol |
| Monoisotopic Mass | 381.11230696 g/mol |
| Topological Polar Surface Area | 81.5 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 446 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Aciphex |
| PubMed Health | Rabeprazole (By mouth) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ACIPHEX Delayed-Release Tablets is rabeprazole sodium, a substituted benzimidazole that inhibits gastric acid secretion. Rabeprazole sodium is known chemically as 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]-methyl]sulfiny... |
| Active Ingredient | Rabeprazole sodium |
| Dosage Form | Tablet, delayed release |
| Route | oral; Oral |
| Strength | 20mg |
| Market Status | Prescription |
| Company | Eisai; Eisai Medcl Res |
| 2 of 4 | |
|---|---|
| Drug Name | Rabeprazole sodium |
| Drug Label | The active ingredient in ACIPHEX Delayed-Release Tablets is rabeprazole sodium, a substituted benzimidazole that inhibits gastric acid secretion. Rabeprazole sodium is known chemically as 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]-methyl]sulfiny... |
| Active Ingredient | Rabeprazole sodium |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 20mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Kremers Urban Dev; Teva Pharms Usa; Torrent Pharms; Lupin; Dr Reddys Labs |
| 3 of 4 | |
|---|---|
| Drug Name | Aciphex |
| PubMed Health | Rabeprazole (By mouth) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ACIPHEX Delayed-Release Tablets is rabeprazole sodium, a substituted benzimidazole that inhibits gastric acid secretion. Rabeprazole sodium is known chemically as 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]-methyl]sulfiny... |
| Active Ingredient | Rabeprazole sodium |
| Dosage Form | Tablet, delayed release |
| Route | oral; Oral |
| Strength | 20mg |
| Market Status | Prescription |
| Company | Eisai; Eisai Medcl Res |
| 4 of 4 | |
|---|---|
| Drug Name | Rabeprazole sodium |
| Drug Label | The active ingredient in ACIPHEX Delayed-Release Tablets is rabeprazole sodium, a substituted benzimidazole that inhibits gastric acid secretion. Rabeprazole sodium is known chemically as 2-[[[4-(3-methoxypropoxy)-3-methyl-2-pyridinyl]-methyl]sulfiny... |
| Active Ingredient | Rabeprazole sodium |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 20mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Kremers Urban Dev; Teva Pharms Usa; Torrent Pharms; Lupin; Dr Reddys Labs |
Treatment of duodenal ulcer, Treatment of gastric ulcer, Treatment of gastro-oesophageal reflux disease, Treatment of Helicobacter pylori in patients with peptic ulcer disease, Treatment of Zollinger-Ellison syndrome
Anti-Ulcer Agents
Various agents with different action mechanisms used to treat or ameliorate PEPTIC ULCER or irritation of the gastrointestinal tract. This has included ANTIBIOTICS to treat HELICOBACTER INFECTIONS; HISTAMINE H2 ANTAGONISTS to reduce GASTRIC ACID secretion; and ANTACIDS for symptomatic relief. (See all compounds classified as Anti-Ulcer Agents.)
Proton Pump Inhibitors
Compounds that inhibit H(+)-K(+)-EXCHANGING ATPASE. They are used as ANTI-ULCER AGENTS and sometimes in place of HISTAMINE H2 ANTAGONISTS for GASTROESOPHAGEAL REFLUX. (See all compounds classified as Proton Pump Inhibitors.)
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Registrant Name : Leaders Bio Co., Ltd.
Registration Date : 2023-01-19
Registration Number : 20190910-209-J-241(2)
Manufacturer Name : Metrochem API Private Limite...
Manufacturer Address : Plot No. 62/C/6, Pipeline Road, Phase - I, IDA., Jeedimetla, Quthbullapur Mandal Medc...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Registrant Name : Kyungbo Pharmaceutical Co., Ltd.
Registration Date : 2019-09-10
Registration Number : 20190910-209-J-241
Manufacturer Name : Metrochem API Private Limite...
Manufacturer Address : Plot No. 62/C/6, Pipeline Road, Phase - I, IDA., Jeedimetla, Quthbullapur Mandal Medc...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Registrant Name : SG Biochem Co., Ltd.
Registration Date : 2022-01-26
Registration Number : 20190910-209-J-241(1)
Manufacturer Name : Metrochem API Private Limite...
Manufacturer Address : Plot No. 62/C/6, Pipeline Road, Phase - I, IDA., Jeedimetla, Quthbullapur Mandal Medc...
Registrant Name : Genuine Science Co., Ltd.
Registration Date : 2021-01-18
Registration Number : 20210118-209-J-822
Manufacturer Name : Kashima Plant, Eisai Co., Lt...
Manufacturer Address : 22 Sunayama, Kamisu-shi, Ibaraki-ken, 314-0255, Japan

Registrant Name : Hana Pharmaceutical Co., Ltd.
Registration Date : 2021-03-23
Registration Number : 20210323-209-J-905
Manufacturer Name : Hana Pharmaceutical Co., Ltd...
Manufacturer Address : 133, Jaekongdan 3-gil, Hyangnam-eup, Hwaseong-si, Gyeonggi-do

Registrant Name : Hanseochem Co., Ltd.
Registration Date : 2019-10-01
Registration Number : 20191001-209-J-439
Manufacturer Name : Hanseochem Co., Ltd. @Enal D...
Manufacturer Address : 41@D-33, 34 & 35, Poseunggongdan-ro, Poseung-eup, Pyeongtaek-si, Gyeonggi-do, Phase �...

Registrant Name : Ildong Pharmaceutical Co., Ltd.
Registration Date : 2018-12-19
Registration Number : 20181219-209-J-154
Manufacturer Name : Ildong Pharmaceutical Co., L...
Manufacturer Address : 53, Gongdan-ro 98beon-gil, Heungdeok-gu, Cheongju-si, Chungcheongbuk-do@[Starting mat...

Registrant Name : JW Pharmaceutical Co., Ltd.
Registration Date : 2022-06-29
Registration Number : 20220629-209-J-1321
Manufacturer Name : JW Pharmaceutical Co., Ltd.@...
Manufacturer Address : 56 Gyeongje-ro, Siheung-si, Gyeonggi-do, Sihwa Industrial Complex 1ma 301 (Jeongwang-...

Registrant Name : Korea Biochem Pharmaceutical Co., Ltd.
Registration Date : 2023-12-27
Registration Number : 20231227-209-J-1590
Manufacturer Name : Korea Biochem Pharmaceutical...
Manufacturer Address : 204 Sandan-gil, Jeonui-myeon, Sejong Special Self-Governing City

Registrant Name : Daesin Pharmaceutical Co., Ltd.
Registration Date : 2020-10-20
Registration Number : 20201020-209-J-514
Manufacturer Name : Nifty Labs Pvt. Ltd.
Manufacturer Address : Plot No. 50-A,B,G&H, 64-A,B,C&D, 65-A,B,C&D, 66-A&B, 67-A&B, IDA, Kondapally, Ibrahim...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Regulatory Info : Registered in EU
Registration Country : Germany
Brand Name :
Dosage Form : Gastro Retentive Tablet
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Registered in EU
Registration Country : Germany
Regulatory Info : Registered in EU
Registration Country : Germany
Brand Name :
Dosage Form : Gastro Retentive Tablet
Dosage Strength : 20MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Registered in EU
Registration Country : Germany
Regulatory Info :
Registration Country : Italy
Brand Name : RABEPRAZOLE THINKS PHARMA
Dosage Form : Gastro-Resistant Tablets
Dosage Strength : 10 mg
Packaging : 14 UNITS 10 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
Regulatory Info :
Registration Country : Italy
Brand Name : RABEPRAZOLE THINKS PHARMA
Dosage Form : Gastro-Resistant Tablets
Dosage Strength : 20 mg
Packaging : 14 UNITS 20 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : RABEPRAZOLE DOC GENERIC
Dosage Form : Gastro-Resistant Tablets
Dosage Strength : 20 mg
Packaging : 28 UNITS 20 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Rabeprazole
Dosage Form : Rabeprazole 10Mg 14 Joined' Oral Use
Dosage Strength : 14 cpr gastrores 10 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Rabeprazole Sandoz
Dosage Form : Film-Coated Tablets
Dosage Strength : 20mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : STARAB
Dosage Form : Gastro-Resistant Tablets
Dosage Strength : 20 mg
Packaging : 28 UNITS 20 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Regulatory Info :
Registration Country : Spain
Brand Name : Combix Rabeprazole 10Mg 28 Tablets Gastro-Resistant Efg
Dosage Form : Gastro-Resistant Tablet
Dosage Strength : 10 Mg/Gastro-resistant Tablet
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Spain
Brand Name : Rabeprazole 20 Mg 14 Tablets Aurobindo Gastro-Resistant Efg
Dosage Form : Gastro-Resistant Tablet
Dosage Strength : 20 Mg/Gastro-resistant Tablet
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
Patent Expiration Date : 2012-09-12
Date Granted : 1995-09-12
Brand Name : PARIET
Patent Number : 1336958
Filing Date : 1989-05-02
Strength per Unit : 10mg
Dosage Form : Enteric Coated Tablet
Human Or VET : Human
Route of Administration : Oral
Patent Expiration Date : 2012-09-12
Date Granted : 1995-09-12

Patent Expiration Date : 2013-08-20
Date Granted : 1999-01-05
Brand Name : PARIET
Patent Number : 2104531
Filing Date : 1993-08-20
Strength per Unit : 10mg
Dosage Form : Enteric Coated Tablet
Human Or VET : Human
Route of Administration : Oral
Patent Expiration Date : 2013-08-20
Date Granted : 1999-01-05

Patent Expiration Date : 2012-09-12
Date Granted : 1995-09-12
Brand Name : PARIET
Patent Number : 1336958
Filing Date : 1989-05-02
Strength per Unit : 20mg
Dosage Form : Enteric Coated Tablet
Human Or VET : Human
Route of Administration : Oral
Patent Expiration Date : 2012-09-12
Date Granted : 1995-09-12

Patent Expiration Date : 2013-08-20
Date Granted : 1999-01-05
Brand Name : PARIET
Patent Number : 2104531
Filing Date : 1993-08-20
Strength per Unit : 20mg
Dosage Form : Enteric Coated Tablet
Human Or VET : Human
Route of Administration : Oral
Patent Expiration Date : 2013-08-20
Date Granted : 1999-01-05

Patent Expiration Date : 2007-11-12
Date Granted :
Brand Name :
Patent Number : 1265138
Filing Date :
Strength per Unit :
Dosage Form :
Human Or VET :
Route of Administration :
Patent Expiration Date : 2007-11-12
Date Granted :

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Patent Expiration Date : 2007-11-12
Date Granted :
Brand Name :
Patent Number : 1265138
Filing Date :
Strength per Unit :
Dosage Form :
Human Or VET :
Route of Administration :
Patent Expiration Date : 2007-11-12
Date Granted :

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
