

Synopsis
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
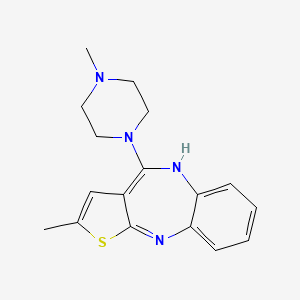

| Molecular Weight | 312.4 g/mol |
|---|---|
| Molecular Formula | C17H20N4S |
| XLogP3 | 2.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Exact Mass | 312.14086783 g/mol |
| Monoisotopic Mass | 312.14086783 g/mol |
| Topological Polar Surface Area | 56.2 A^2 |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 543 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 8 | |
|---|---|
| Drug Name | Olanzapine |
| PubMed Health | Olanzapine |
| Drug Classes | Antipsychotic |
| Drug Label | Olanzapine is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresponds to a... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet; Injectable; Tablet, orally disintegrating |
| Route | oral; Oral; Intramuscular |
| Strength | 2.5mg; 10mg/vial; 7.5mg; 15mg; 5mg; 10mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Mylan Pharms; Apotex; Aurobindo Pharma; Sun Pharm Inds; Torrent Pharms; Barr Labs; Sandoz; Par Pharm; Roxane; Teva Pharms; Macleods Pharms; Jubilant Generics; Luitpold; Dr Reddys Labs; Orchid Hlthcare; Mylan |
| 2 of 8 | |
|---|---|
| Drug Name | Symbyax |
| PubMed Health | Olanzapine/Fluoxetine (By mouth) |
| Drug Classes | Antidepressant, Antipsychotic |
| Active Ingredient | Fluoxetine hydrochloride; olanzapine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | eq 50mg base; eq 6mg base; eq 12mg base; eq 3mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Lilly |
| 3 of 8 | |
|---|---|
| Drug Name | Zyprexa |
| Drug Label | ZYPREXA (olanzapine) is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresp... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet; Injectable |
| Route | Oral; Intramuscular |
| Strength | 2.5mg; 10mg/vial; 7.5mg; 15mg; 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Lilly |
| 4 of 8 | |
|---|---|
| Drug Name | Zyprexa zydis |
| Drug Label | ZYPREXA (olanzapine) is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresp... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet, orally disintegrating |
| Route | Oral |
| Strength | 15mg; 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Lilly |
| 5 of 8 | |
|---|---|
| Drug Name | Olanzapine |
| PubMed Health | Olanzapine |
| Drug Classes | Antipsychotic |
| Drug Label | Olanzapine is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresponds to a... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet; Injectable; Tablet, orally disintegrating |
| Route | oral; Oral; Intramuscular |
| Strength | 2.5mg; 10mg/vial; 7.5mg; 15mg; 5mg; 10mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Mylan Pharms; Apotex; Aurobindo Pharma; Sun Pharm Inds; Torrent Pharms; Barr Labs; Sandoz; Par Pharm; Roxane; Teva Pharms; Macleods Pharms; Jubilant Generics; Luitpold; Dr Reddys Labs; Orchid Hlthcare; Mylan |
| 6 of 8 | |
|---|---|
| Drug Name | Symbyax |
| PubMed Health | Olanzapine/Fluoxetine (By mouth) |
| Drug Classes | Antidepressant, Antipsychotic |
| Active Ingredient | Fluoxetine hydrochloride; olanzapine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | eq 50mg base; eq 6mg base; eq 12mg base; eq 3mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Lilly |
| 7 of 8 | |
|---|---|
| Drug Name | Zyprexa |
| Drug Label | ZYPREXA (olanzapine) is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresp... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet; Injectable |
| Route | Oral; Intramuscular |
| Strength | 2.5mg; 10mg/vial; 7.5mg; 15mg; 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Lilly |
| 8 of 8 | |
|---|---|
| Drug Name | Zyprexa zydis |
| Drug Label | ZYPREXA (olanzapine) is an atypical antipsychotic that belongs to the thienobenzodiazepine class. The chemical designation is 2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b] [1,5]benzodiazepine. The molecular formula is C17H20N4S, which corresp... |
| Active Ingredient | Olanzapine |
| Dosage Form | Tablet, orally disintegrating |
| Route | Oral |
| Strength | 15mg; 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Lilly |

GDUFA
DMF Review : Reviewed
Rev. Date : 2014-03-03
Pay. Date : 2013-01-23
DMF Number : 26694
Submission : 2013-01-24
Status : Active
Type : II

NDC Package Code : 64567-0017
Start Marketing Date : 2013-01-24
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Samo Pharmaceutical Co., Ltd.
Registration Date : 2020-11-13
Registration Number : 20201113-209-J-568
Manufacturer Name : INKE S.A.
Manufacturer Address : C/Argent, 1. Area Industrial del Llobregat, 08755 Castellbisbal (Barcelona), Spain

| Available Reg Filing : ASMF |
 Synnat Pharma is one of the leading active pharmaceutical ingredients and intermediates manufacturers.
Synnat Pharma is one of the leading active pharmaceutical ingredients and intermediates manufacturers.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19282
Submission : 2006-03-24
Status : Active
Type : II

Certificate Number : R1-CEP 2012-190 - Rev 01
Issue Date : 2021-09-07
Type : Chemical
Substance Number : 2258
Status : Valid

Date of Issue : 2022-07-28
Valid Till : 2025-06-16
Written Confirmation Number : WC-0036
Address of the Firm : Sy. No. 347,473,474, 490/2 Bonthapally village, Veerabhadraswamy Temple Road, Jinnaram Mandal, Medak Dist. Telangana State India

NDC Package Code : 58032-0122
Start Marketing Date : 2017-12-14
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Samil Pharmaceutical Co., Ltd.
Registration Date : 2022-08-22
Registration Number : 20220822-209-J-1347
Manufacturer Name : Neuland Laboratories Ltd
Manufacturer Address : Unit-I, Survey No: 347, 473, 474, 490/2 Veerabhadraswamy temple road,Bonthapally Village, Gummadidala Mandal,Sangareddy District - 502313, Telangana, India
 Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.

GDUFA
DMF Review : Reviewed
Rev. Date : 2021-07-13
Pay. Date : 2021-07-09
DMF Number : 18260
Submission : 2005-04-08
Status : Active
Type : II

Certificate Number : R1-CEP 2012-050 - Rev 02
Issue Date : 2023-02-21
Type : Chemical
Substance Number : 2258
Status : Valid

Date of Issue : 2022-09-30
Valid Till : 2025-08-04
Written Confirmation Number : WC-0127
Address of the Firm : A1/B, SIPCOT Industrial Complex, Kudikadu, Cuddalore-607 005

NDC Package Code : 72643-023
Start Marketing Date : 2020-05-25
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Inseong Trading Co., Ltd.
Registration Date : 2021-07-21
Registration Number : 20210721-209-J-1041
Manufacturer Name : Solara Active Pharma Sciences Limited
Manufacturer Address : A1/B, SIPCOT Industrial Complex, Kudikadu, Cuddalore- 607 005, India


GDUFA
DMF Review : Reviewed
Rev. Date : 2014-02-03
Pay. Date : 2013-09-23
DMF Number : 26763
Submission : 2012-12-28
Status : Active
Type : II

Certificate Number : R1-CEP 2012-406 - Rev 01
Issue Date : 2023-07-20
Type : Chemical
Substance Number : 2258
Status : Valid

NDC Package Code : 49587-103
Start Marketing Date : 2014-12-22
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : Complete
Rev. Date : 2014-03-03
Pay. Date : 2013-01-23
DMF Number : 26694
Submission : 2013-01-24
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19282
Submission : 2006-03-24
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15052
Submission : 2000-09-18
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-01-24
Pay. Date : 2013-12-10
DMF Number : 20591
Submission : 2007-06-11
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2012-11-23
Pay. Date : 2012-12-17
DMF Number : 20990
Submission : 2007-10-22
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20354
Submission : 2007-03-16
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-07-13
Pay. Date : 2021-07-09
DMF Number : 18260
Submission : 2005-04-08
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-02-18
Pay. Date : 2014-02-10
DMF Number : 20218
Submission : 2007-01-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18108
Submission : 2005-02-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18867
Submission : 2005-10-14
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Olanzapin Zentiva
Dosage Form : Tablet
Dosage Strength : 10mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Olanzapin Zentiva
Dosage Form : Tablet
Dosage Strength : 15mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Olanzapine Zentiva
Dosage Form : Tabl
Dosage Strength : 2.5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Olanzapine Zentiva
Dosage Form : Tabl
Dosage Strength : 5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : OLAWIN FILM COATED 2,5 MG
Dosage Form : FCT
Dosage Strength : 20mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Prexolan ODT 5
Dosage Form : TAB
Dosage Strength : 5mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Prexolan ODT 10
Dosage Form : TAB
Dosage Strength : 10mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Prexolan 2,5
Dosage Form : TAB
Dosage Strength : 2.5mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Prexolan 5
Dosage Form : FCT
Dosage Strength : 5mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Prexolan 10
Dosage Form : FCT
Dosage Strength : 10mg
Packaging : 30X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET, ORALLY DISINTEGRATING;ORAL
Dosage Strength : 5MG
Approval Date : 2011-10-24
Application Number : 78109
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET, ORALLY DISINTEGRATING;ORAL
Dosage Strength : 10MG
Approval Date : 2011-10-24
Application Number : 78109
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET, ORALLY DISINTEGRATING;ORAL
Dosage Strength : 15MG
Approval Date : 2011-10-24
Application Number : 78109
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET, ORALLY DISINTEGRATING;ORAL
Dosage Strength : 20MG
Approval Date : 2011-10-24
Application Number : 78109
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
OLANZAPINE; SAMIDORPHAN L-MALATE
Brand Name : LYBALVI
Dosage Form : TABLET;ORAL
Dosage Strength : 10MG;EQ 10MG BASE
Approval Date : 2021-05-28
Application Number : 213378
RX/OTC/DISCN : RX
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET;ORAL
Dosage Strength : 5MG
Approval Date : 2012-04-23
Application Number : 90798
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET;ORAL
Dosage Strength : 7.5MG
Approval Date : 2016-03-15
Application Number : 203333
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code : AB
Brand Name : ZYPREXA
Dosage Form : TABLET;ORAL
Dosage Strength : 2.5MG
Approval Date : 1996-09-30
Application Number : 20592
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET;ORAL
Dosage Strength : 2.5MG
Approval Date : 2012-04-23
Application Number : 202287
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : OLANZAPINE
Dosage Form : TABLET, ORALLY DISINTEGRATING;ORAL
Dosage Strength : 15MG
Approval Date : 2020-12-31
Application Number : 206892
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
