

1. 22972-51-6
2. Cis-isolimonenol
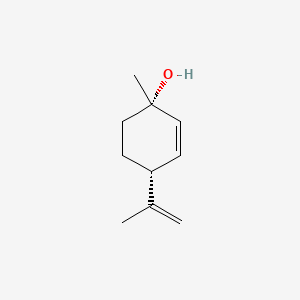
3. (1s,4r)-p-mentha-2,8-dien-1-ol
4. 2-cyclohexen-1-ol, 1-methyl-4-(1-methylethenyl)-, (1s,4r)-
5. 2,8-p-menthadien-1-ol, Cis-(+)-
6. (1s,4r)-1-methyl-4-prop-1-en-2-ylcyclohex-2-en-1-ol
7. (+)-p-mentha-2,8-dien-1-ol
8. (+)-cis-p-mentha-2,8-dien-1-ol
9. (+)-(1s,4r)-p-mentha-2,8-dien-1-ol
10. 1eok52zyqh
11. Fema No. 4411, Cis-(+)-
12. (1s,4r)-p-menth-2,8-dien-1-ol
13. (1s,4r)-4-isopropenyl-1-methylcyclohex-2-en-1-ol
14. (1s,4r)-1-methyl-4-(prop-1-en-2-yl)cyclohex-2-en-1-ol
15. Rel-(1s,4r)-1-methyl-4-(prop-1-en-2-yl)cyclohex-2-enol
16. 2-cyclohexen-1-ol, 1-methyl-4-(1-methylethenyl)-, (1s-cis)-
17. Unii-1eok52zyqh
18. Mfcd08460036
19. P-mentha-2,8-dien-1-beta-ol
20. Schembl1114907
21. Cis-p-mentha-2,8-diene 1-ol
22. Dtxsid50177519
23. Chebi:171955
24. Bcp12523
25. Cs-m3276
26. Zinc5158345
27. Cis-(+)-2,8-p-menthadien-1-ol
28. Fd7167
29. Akos006288260
30. Ds-4115
31. Ac-26319
32. Hy-41094
33. P-mentha-2,8-dien-1-.beta.-ol
34. P-mentha-2,8-dien-1-ol, (1s,4r)-
35. 972m516
36. A846922
37. (1s,4r)-4-isoprenyl-1-methylcyclohex-2-en-1-ol
38. Q27252332
39. (1s,4r)-1-methyl-4-(1-methylvinyl)-cyclohex-2-ene-1-ol
1. 4-methyl-beta-methylenecyclohex-3-ene-1-ethanol
2. 3269-90-7
3. 38142-45-9
4. Menthadienol
| Molecular Weight | 152.23 g/mol |
|---|---|
| Molecular Formula | C10H16O |
| XLogP3 | 2.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 152.120115130 g/mol |
| Monoisotopic Mass | 152.120115130 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 193 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
BUILDING BLOCK

MARKET PLACE


