

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF
0
Europe

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Finished Drug Prices
NA
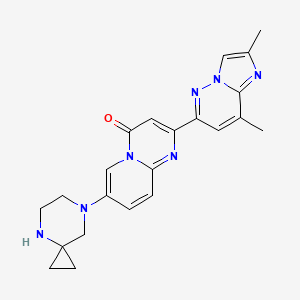

1. 7-(4,7-diazaspiro(2.5)oct-7-yl)-2-(2,8-dimethylimidazo(1,2-b)pyridazin-6-yl)-4h-pyrido(1,2-a)pyrimidin-4-one
1. 1825352-65-5
2. Rg7916
3. Evrysdi
4. Ro7034067
5. Risdiplam [inn]
6. Risdiplam [usan]
7. Rg-7916
8. 76rs4s2et1
9. Ro-7034067
10. 7-(4,7-diazaspiro[2.5]octan-7-yl)-2-(2,8-dimethylimidazo[1,2-b]pyridazin-6-yl)pyrido[1,2-a]pyrimidin-4-one
11. 2-(2,8-dimethylimidazo[1,2-b]pyridazin-6-yl)-7-(4,7-diazaspiro[2.5]octan-7-yl)-4h-pyrido[1,2-a]pyrimidin-4-one
12. 4h-pyrido(1,2-a)pyrimidin-4-one, 7-(4,7-diazaspiro(2.5)oct-7-yl)-2-(2,8-dimethylimidazo(1,2-b)pyridazin-6-yl)-
13. Evrysdi (tn)
14. Risdiplam; Rg7916
15. Risdiplam [jan]
16. Risdiplam [mi]
17. Risdiplam [usan:inn]
18. Risdiplam [who-dd]
19. Unii-76rs4s2et1
20. Risdiplam (jan/usan/inn)
21. Risdiplam [orange Book]
22. Chembl4297528
23. Schembl17260852
24. Gtpl11170
25. Dtxsid701109185
26. Amy23728
27. Ex-a2074
28. Mfcd31657372
29. Who 10614
30. Compound 1 [pmid: 30044619]
31. Db15305
32. Ac-36304
33. Br166842
34. Rg7916;ro7034067
35. Hy-109101
36. Cs-0039501
37. D11406
38. F53623
39. Q48969152
40. (7-(4,7-diazaspiro(2.5)octan-7-yl)-2-(2,8-dimethylimidazo(1,2-b)pyridazin-6-yl)pyrido(1,2-a)pyrimidin-4-one
41. 7-(4,7-diazaspiro(2.5)oct-7-yl)-2-(2,8-dimethylimidazo(1,2-b)pyridazin-6-yl)-4h-pyrido(1,2-a)pyrimidin-4-one
| Molecular Weight | 401.5 g/mol |
|---|---|
| Molecular Formula | C22H23N7O |
| XLogP3 | 0.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Exact Mass | 401.19640838 g/mol |
| Monoisotopic Mass | 401.19640838 g/mol |
| Topological Polar Surface Area | 78.1 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 886 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Risdiplam is indicated for the treatment of spinal muscular atrophy (SMA) in patients 2 months of age and older.
Evrysdi is indicated for the treatment of 5q spinal muscular atrophy (SMA) in patients 2 months of age and older, with a clinical diagnosis of SMA Type 1, Type 2 or Type 3 or with one to four SMN2 copies.
Risdiplam helps to alleviate symptoms of spinal muscular atrophy by stimulating the production of a critical protein in which these patients are deficient. Early trials with risdiplam demonstrated up to a 2-fold increase in SMN protein concentration in SMA patients after 12 weeks of therapy.
Neuromuscular Agents
Drugs used for their actions on skeletal muscle. Included are agents that act directly on skeletal muscle, those that alter neuromuscular transmission (NEUROMUSCULAR BLOCKING AGENTS), and drugs that act centrally as skeletal muscle relaxants (MUSCLE RELAXANTS, CENTRAL). Drugs used in the treatment of movement disorders are ANTI-DYSKINESIA AGENTS. (See all compounds classified as Neuromuscular Agents.)
M09AX10
M - Musculo-skeletal system
M09 - Other drugs for disorders of the musculo-skeletal system
M09A - Other drugs for disorders of the musculo-skeletal system
M09AX - Other drugs for disorders of the musculo-skeletal system
M09AX10 - Risdiplam
Absorption
The Tmax following oral administration is approximately 1-4 hours. Following once-daily administration with a morning meal (or after breastfeeding), risdiplam reaches steady-state in approximately 7-14 days. The pharmacokinetics of risdiplam were found to be approximately linear between all studied dosages in patients with SMA.
Route of Elimination
Following the oral administration of 18mg risdiplam, approximately 53% of the dose was excreted in the feces and 28% was excreted in the urine. Unchanged parent drug comprised 14% of the dose excreted in feces and 8% of the dose excreted in urine.
Volume of Distribution
Following oral administration, risdiplam distributes well into the central nervous system and peripheral tissues. The apparent volume of distribution at steady-state is 6.3 L/kg.
Clearance
For a 14.9kg patient, the apparent clearance of risdiplam is 6.3 L/kg.
The metabolism of risdiplam is mediated primarily by flavin monooxygenases 1 and 3 (FMO1 and FMO3), with some involvement of CYP1A1, CYP2J2, CYP3A4, and CYP3A7. Parent drug comprises approximately 83% of circulating drug material. A pharmacologically-inactive metabolite, M1, has been identified as the major circulating metabolite - this M1 metabolite has been observed _in vitro_ to inhibit MATE1 and MATE2-K transporters, similar to the parent drug.
The terminal elimination half-life of risdiplam is approximately 50 hours in healthy adults.
Spinal muscular atrophy (SMA) is a severe and progressive congenital neuromuscular disease resulting from mutations in the survival of motor neuron 1 (_SMN1_) gene responsible for making SMN proteins. Clinical features of SMA include degeneration of motor neurons in the spinal cord which ultimately leads to muscular atrophy and, in some cases, loss of physical strength. SMN proteins are expressed ubiquitously throughout the body and are thought to hold diverse intracellular roles in DNA repair, cell signaling, endocytosis, and autophagy. A secondary _SMN_ gene (_SMN2_) can also produce SMN proteins, but a small nucleotide substitution in its sequence results in the exclusion of exon 7 during splicing in approximately 85% of the transcripts - this means that only ~15% of the SMN proteins produced by _SMN2_ are functional, which is insufficient to compensate for the deficits caused by _SMN1_ mutations. Emerging evidence suggests that many cells and tissues are selectively vulnerable to reduced SMN concentrations, making this protein a desirable target in the treatment of SMA. Risdiplam is an mRNA splicing modifier for _SMN2_ that increases the inclusion of exon 7 during splicing, which ultimately increases the amount of functional SMN protein produced by _SMN2_. It does so by binding to two sites in _SMN2_ pre-mRNA: the 5' splice site (5'ss) of intron 7 and the exonic splicing enhancer 2 (ESE2) of exon 7.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
