

Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF
0
FDA Orange Book

0
Europe

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
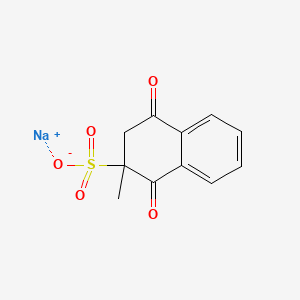

1. 2-methyl-1,4-naphthalenedione
2. 2-methyl-1,4-naphthoquinone
3. 2-methylnaphthoquinone
4. Bisulfite, Menadione
5. Bisulfite, Menadione Sodium
6. Menadione
7. Menadione Bisulfite
8. Menadione Sodium Bisulfite, Trihydrate
9. Sodium Bisulfite, Menadione
10. Vicasol
11. Vikasol
12. Vitamin K 3
13. Vitamin K3
14. Vitamin K3 Sodium Bisulfite
1. 130-37-0
2. Dohyfral
3. Hemoklot
4. Kavitamin
5. Sodium Menadione Bisulfite
6. Vitamin K3 Sodium Bisulfite
7. Msbc
8. Menadione Sodium Bisulfite Anhydrous
9. Natrium Menadionsulfonicum
10. Menadione Bisulfite Sodium
11. Menaphthone Sodium Bisulphite
12. Menadioni Natrii Hydrogensulfis
13. 2-methyl-1,4-naphthoquinone Sodium Bisulfite
14. Menachinonum Natrium Bisulfurosum
15. Menadion Sodium Bisulfite
16. Menadione Sodium Hydrogen Sulfite
17. Menadioni Natrii Bisulfis
18. Menaphthone Sodium Bisulfite
19. Bisulfito Sodico De Menadiona
20. Bisulfite Sodique De Menadione
21. 2-methylnaphthoquinone Sodium Hydrogen Sulfite
22. 2,3-dihydro-2-methyl-1,4-naphthoquinone-2-sulfonate Sodium
23. Vitamin K2(menadione Sodium Bisulfite
24. 2-methyl-1,4-naphthoquinone Sodium Hydrogen Sulfite
25. Sodium;2-methyl-1,4-dioxo-3h-naphthalene-2-sulfonate
26. Chebi:63928
27. Menadione Sodium Bisulfite [inn]
28. 97k34v88sa
29. 1,2,3,4-tetrahydro-2-methyl-1,4-dioxo-2-naphthalenesulfonic Acid Sodium Salt
30. 2-naphthalenesulfonic Acid, 1,2,3,4-tetrahydro-2-methyl-1,4-dioxo-, Sodium Salt
31. 57414-02-5
32. Menadione Sodium Sulfonate
33. Kavitan
34. Kawitan
35. Vikasol
36. Vikasolum
37. Kalzon
38. Hetrogen K
39. K-trombina
40. Klotogen F
41. Sodium 2-methyl-1,4-dioxo-1,2,3,4-tetrahydronaphthalene-2-sulfonate
42. Golagen K
43. Hetroge K Premix
44. Menadione (sodium Bisulfite)
45. Vitamin K Injection
46. Ido-k
47. Klotogen F 16
48. Klotogen F 227
49. Menadione Sodium Bisulfite (inn)
50. Sulfurous Acid, Monosodium Salt, Compd. With 2-methyl-1,4-naphthalenedione (1:1)
51. Ccris 7112
52. Menadione Sodio Bisolfito [dcit]
53. Menadione Sodio Bisolfito
54. Ncgc00160478-01
55. Einecs 204-987-0
56. Menadioni Natrii Bisulfis [inn-latin]
57. Unii-97k34v88sa
58. Bisulfite Sodique De Menadione [inn-french]
59. Bisulfito Sodico De Menadiona [inn-spanish]
60. Mfcd00051456
61. Vitamin K3 Sodium Bisulfite Trihydrate
62. Menadionesodiumbisulfite
63. 2-methyl-1,4-naphthoquinone Sodiumbisulfite
64. Dsstox_cid_20547
65. Dsstox_rid_79505
66. 1,4-naphthoquinone, 2-methyl-, Sodium Bisulfite
67. Dsstox_gsid_40547
68. Schembl435171
69. 2-methyl-1,4-naphthochinon-natrium-bisulfit Trihydrat
70. Chembl2358118
71. Dtxsid0040547
72. Hy-b1897a
73. Sodium Dihydromenadione-2-sulfonate
74. 2-methyl-1,4-naphthochinon-natrium-bisulfit Trihydrat [german]
75. Bcp19124
76. Tox21_113121
77. K-50 (tn)
78. Stk700858
79. 1,4-naphthalenedione, 2-methyl-, Compd. With Sodium Hydrogen Sulfite (1:1)
80. Akos005607578
81. Akos015895594
82. Cs-6059
83. Menadione Sodium Bisulfite [mi]
84. As-15191
85. Cas-130-37-0
86. Menadione Sodium Bisulfite [who-dd]
87. Ft-0628195
88. Ft-0741767
89. Menaphthone Sodium Bisulfite Anhydrous
90. C18378
91. D08177
92. D88058
93. A806075
94. A831437
95. Q27132914
96. Sodium 2-methyl-1,4-dioxo-3h-naphthalene-2-sulfonate
97. Sodium 2-methyl-1,4-bis(oxidanylidene)-3h-naphthalene-2-sulfonate
98. 2-naphthalenesulfonic Acid, 1,2,3,4-tetrahydro-2-methyl-1,4-dioxo-, Sodium Salt (1:1)
99. Sodium; Hydrogen Sulfite; 2-methylnaphthalene-1,4-dione; Trihydrate;menadione Sodium Bisulfite
| Molecular Weight | 276.24 g/mol |
|---|---|
| Molecular Formula | C11H9NaO5S |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Exact Mass | 276.00683884 g/mol |
| Monoisotopic Mass | 276.00683884 g/mol |
| Topological Polar Surface Area | 99.7 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 466 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Antifibrinolytic Agents
Agents that prevent fibrinolysis or lysis of a blood clot or thrombus. Several endogenous antiplasmins are known. The drugs are used to control massive hemorrhage and in other coagulation disorders. (See all compounds classified as Antifibrinolytic Agents.)
Vitamins
Organic substances that are required in small amounts for maintenance and growth, but which cannot be manufactured by the human body. (See all compounds classified as Vitamins.)
About the Company : Anthem Biosciences is a leading contract research and innovation service provider (CRISP) in Bengaluru, India, which manufactures novel drug actives. Established in 2007, it specia...

About the Company : MENADIONA is an independent privately-owned company, founded in 1970. Ever since, the company has been involved in manufacturing active ingredients (APIs) and key intermediates for...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
