

Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
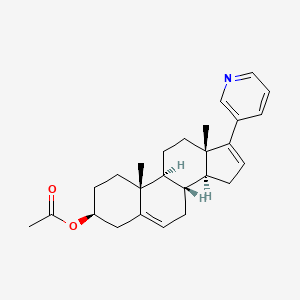

1. 17-(3-pyridyl)-5,16-androstadien-3beta-acetate
2. Cb 7630
3. Cb-7630
4. Cb7630
5. Zytiga
1. 154229-18-2
2. Zytiga
3. Cb7630
4. Cb-7630
5. Yonsa
6. Cb 7630
7. Abiraterone Acetate [usan]
8. 17-(3-pyridyl)-5,16-androstadien-3beta-acetate
9. Em5ocb9yj6
10. [(3s,8r,9s,10r,13s,14s)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,7,8,9,11,12,14,15-decahydro-1h-cyclopenta[a]phenanthren-3-yl] Acetate
11. Nsc-748121
12. Nsc-749227
13. Chebi:68639
14. (3s,8r,9s,10r,13s,14s)-10,13-dimethyl-17-(pyridin-3-yl)-2,3,4,7,8,9,10,11,12,13,14,15-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl Acetate
15. 17-(pyridin-3-yl)androsta-5,16-dien-3beta-yl Acetate
16. (3beta)-17-(pyridin-3-yl)androsta-5,16-dien-3-yl Acetate
17. Abiraterone Acotate
18. 17-(3-pyridyl)androsta-5, Acetate
19. Androsta-5, 17-(3-pyridinyl)-, 3-acetate,
20. Unii-em5ocb9yj6
21. Abiraterone-acetate
22. Ncgc00186462-01
23. 3s,9s,10r,13s,14s)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,7,8,9,11,12,14,15-decahydro-1h-cyclopenta[a]phenanthren-3-ol, 3-acetate
24. Abiraterone Acetate
25. Mfcd00934213
26. Zytiga (tn)
27. Abiraterone (acetate)
28. Jnj-212082
29. Abiraterone Acetate- Bio-x
30. Dsstox_cid_28969
31. Dsstox_rid_83234
32. Dsstox_gsid_49043
33. Schembl93715
34. Mls006010090
35. Chembl271227
36. Gtpl9288
37. Abiraterone Acetate (jan/usp)
38. 3beta-17-(3-pyridyl)androsta-5,16-dien-3-ol Acetate
39. Abiraterone (as Acetate)
40. Abiraterone Acetate [mi]
41. Dtxsid3049043
42. Abiraterone Acetate [jan]
43. Ex-a107
44. Abiraterone Acetate [vandf]
45. Abiraterone Acetate [mart.]
46. Bcp02949
47. Zinc3809191
48. Tox21_113589
49. Abiraterone Acetate [usp-rs]
50. Abiraterone Acetate [who-dd]
51. Bdbm50407398
52. Nsc748121
53. Nsc749227
54. S2246
55. Abiraterone Acetate, >=98% (hplc)
56. Akos015896502
57. Cs-0544
58. Ds-2007
59. Jnj 212082
60. Nsc 748121
61. Nsc 749227
62. Jnj-2012082
63. Abiraterone Acetate [orange Book]
64. Ncgc00379022-06
65. Ac-25760
66. Ba164133
67. Hy-75054
68. Smr004701235
69. Abiraterone Acetate [usp Monograph]
70. Cas-154229-18-2
71. D09701
72. 3b-acetoxy-17-(3-pyridyl)androsta-5,16-diene
73. A809510
74. 3beta-acetoxy-17-(3-pyridyl)androsta-5,16-diene
75. W-201385
76. Brd-k24048528-001-02-5
77. Q27888393
78. (3s)-3-acetoxy-17-(pyridin-3-yl)androsta-5,16-diene
79. (3?)-17-(3-pyridinyl)androsta-5,16-dien-3-yl Acetate
80. (3beta)-17-(3-pyridinyl)androsta-5,16-dien-3-ol Acetate
81. 17-(pyridin-3-yl)androsta-5,16-dien-3.beta.-yl Acetate
82. Abiraterone Acetate, United States Pharmacopeia (usp) Reference Standard
83. Androsta-5,16-dien-3-ol, 17-(3-pyridinyl)-, Acetate (ester), (3beta)-
84. Androsta-5,16-dien-3-ol, 17-(3-pyridinyl)-, Acetate (ester), (3.beta.)-
85. (1s,2r,5s,10r,11s,15s)-2,15-dimethyl-14-(pyridin-3-yl)tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-7,13-dien-5-yl Acetate
86. [(3s,8r,9s,10r,13s,14s)-10,13-dimethyl-17-pyridin-3-yl-2,3,4,7,8,9,11,12,14,15-decahydro-1h-cyclopenta[a]phenanthren-3-yl] Ethanoate
87. Acetic Acid [(3s,8r,9s,10r,13s,14s)-10,13-dimethyl-17-(3-pyridinyl)-2,3,4,7,8,9,11,12,14,15-decahydro-1h-cyclopenta[a]phenanthren-3-yl] Ester
| Molecular Weight | 391.5 g/mol |
|---|---|
| Molecular Formula | C26H33NO2 |
| XLogP3 | 5.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Exact Mass | 391.251129295 g/mol |
| Monoisotopic Mass | 391.251129295 g/mol |
| Topological Polar Surface Area | 39.2 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 739 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Zytiga is indicated with prednisone or prednisolone for:
- the treatment of metastatic castration resistant prostate cancer in adult men who are asymptomatic or mildly symptomatic after failure of androgen deprivation therapy in whom chemotherapy is not yet clinically indicated
- the treatment of metastatic castration resistant prostate cancer in adult men whose disease has progressed on or after a docetaxel based chemotherapy regimen.
Abiraterone Mylan is indicated with prednisone or prednisolone for:
- the treatment of newly diagnosed high risk metastatic hormone sensitive prostate cancer (mHSPC) in adult men in combination with androgen deprivation therapy (ADT).
- the treatment of metastatic castration resistant prostate cancer (mCRPC) in adult men who are asymptomatic or mildly symptomatic after failure of androgen deprivation therapy in whom chemotherapy is not yet clinically indicated.
- the treatment of mCRPC in adult men whose disease has progressed on or after a docetaxel based chemotherapy regimen.
Abiraterone Krka is indicated with prednisone or prednisolone for:
- the treatment of newly diagnosed high risk metastatic hormone sensitive prostate cancer (mHSPC) in adult men in combination with androgen deprivation therapy (ADT) (see section 5. 1)
- the treatment of metastatic castration resistant prostate cancer (mCRPC) in adult men who are asymptomatic or mildly symptomatic after failure of androgen deprivation therapy in whom chemotherapy is not yet clinically indicated (see section 5. 1)
- the treatment of mCRPC in adult men whose disease has progressed on or after a docetaxel-based chemotherapy regimen.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Cytochrome P-450 Enzyme Inhibitors
Drugs and compounds which inhibit or antagonize the biosynthesis or actions of CYTOCHROME P-450 ENZYMES. (See all compounds classified as Cytochrome P-450 Enzyme Inhibitors.)
Steroid Synthesis Inhibitors
Compounds that bind to and inhibit enzymes involved in the synthesis of STEROIDS. (See all compounds classified as Steroid Synthesis Inhibitors.)
L02BX03
L02BX03
L02BX03

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
We have 25 companies offering DSSTox_CID_28969
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
