

Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
API
0
FDF
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
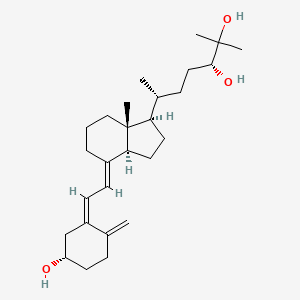

1. (24r)-24,25-dihydroxyvitamin D3
2. 24,25 Dihydroxycholecalciferol
3. 24,25 Dihydroxyvitamin D 3
4. 24,25 Dihydroxyvitamin D3
5. 24,25-dihydroxycholecalciferol
6. 24,25-dihydroxyvitamin D 3
7. 24,25-dihydroxyvitamin D 3, (3beta,5z,7e,24r)-isomer
8. 24,25-dihydroxyvitamin D3
9. 24r,25 Dihydroxycholecalciferol
10. 24r,25-dihydroxycholecalciferol
11. Dihydroxyvitamin D3, 24,25
1. Osteo D
2. 24r,25-dihydroxyvitamin D3
3. 55721-11-4
4. K-dr
5. (24r)-hydroxycalcidiol
6. (24r)-24,25-dihydroxyvitamin D3
7. 24r,25-dihydroxycholecalciferol
8. 24(r),25-dihydroxyvitamin D3
9. 24(r),25-dihydroxycholecalciferol
10. 24,25-dihydroxyvitamin D3
11. Chebi:28818
12. 460029iuda
13. 24(r),25-(oh)2d3
14. (3r,6r)-6-[(1r,3as,4e,7ar)-4-[(2z)-2-[(5s)-5-hydroxy-2-methylidenecyclohexylidene]ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro-1h-inden-1-yl]-2-methylheptane-2,3-diol
15. Ncgc00161328-03
16. (3s,5z,7e,24r)-9,10-secocholesta-5,7,10-triene-3,24,25-triol
17. 24,25 Dihydroxyvitamin D3
18. (24r)-24,25-dihydroxycalciol
19. Secalciferolum
20. (24r)-24,25-dihydroxyvitamin D3 / (24r)-24,25-dihydroxycholecalciferol
21. (5z,7e)-(3s,24r)-9,10-seco-5,7,10(19)-cholestatriene-3,24,25-triol
22. 40013-87-4
23. Secalciferolum [inn-latin]
24. 24, 25-dihydroxy Vitamin D3
25. Unii-460029iuda
26. Ccris 8678
27. Secalciferol [usan:inn:ban]
28. Osteo D (tn)
29. C07712
30. Ro 21-5816
31. Dsstox_cid_1260
32. Secalciferol [inn]
33. Secalciferol [jan]
34. Secalciferol [usan]
35. Schembl3597
36. Dsstox_rid_76045
37. Dsstox_gsid_21260
38. Bspbio_001409
39. Bml2-e04
40. Secalciferol (jan/usan/inn)
41. Chembl1590076
42. Dtxsid4021260
43. (24r),25-dihydroxyvitamin D3
44. Chebi:108603
45. Hms1361g11
46. Hms1791g11
47. Hms1989g11
48. Hms3402g11
49. Tox21_111989
50. Lmst03020273
51. Mfcd00941417
52. (3-beta,5z,7e,24r)-9,10-secocholesta-5,7,10(19)-triene-3,24,25-triol
53. (5z,7e,24r)-9,10-secocholesta-5,7,10(19)-triene-3beta,24,25-triol
54. 24r,25(oh)2d3
55. Zinc100051969
56. Cs-0929
57. (24r)-24,25-dihydroxycholecalciferol
58. Idi1_033879
59. 24, 25-dihydroxycholecalciferol
60. Ncgc00161328-01
61. Hy-32343
62. Cas-55721-11-4
63. 24r,25-dihydroxyvitamin D3 (secalciferol)
64. D00628
65. Q27103914
66. (24r)-24,25-dihydroxyvitamin D3, >=98% (vitamin + Pre-vitamin, Hplc)
67. 9,10-secocholesta-5,7,10-triene-3,24,25-triol, (3s,5z,7e,24r)-
68. (5z,7e,24r)-9,10-secocholesta-5,7,10(19)-triene-3b,24,25-triol
69. 9,10-secocholesta-5,7,10(19)-triene-3,24,25-triol, (3beta,5z,7e,24r)-
70. (3r,6r)-6-[(1r,3as,4e,7ar)-4-[(2z)-2-[(5s)-5-hydroxy-2-methylene-cyclohexylidene]ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro-1h-inden-1-yl]-2-methyl-heptane-2,3-diol
71. 2,3-heptanediol, 2-methyl-6-((1r,3as,4e,7ar)-octahydro-4-((2z)-2-((5s)-5-hydroxy-2-methylenecyclohexylidene)ethylidene)-7a-methyl-1h-inden-1-yl)-, (3r,6r)-
| Molecular Weight | 416.6 g/mol |
|---|---|
| Molecular Formula | C27H44O3 |
| XLogP3 | 5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 6 |
| Exact Mass | 416.32904526 g/mol |
| Monoisotopic Mass | 416.32904526 g/mol |
| Topological Polar Surface Area | 60.7 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 688 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 2 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Bone Density Conservation Agents
Agents that inhibit BONE RESORPTION and/or favor BONE MINERALIZATION and BONE REGENERATION. They are used to heal BONE FRACTURES and to treat METABOLIC BONE DISEASES such as OSTEOPOROSIS. (See all compounds classified as Bone Density Conservation Agents.)
Calcium-Regulating Hormones and Agents
Hormones and molecules with calcium-regulating hormone-like actions that modulate OSTEOLYSIS and other extra-skeletal activities to maintain calcium homeostasis. (See all compounds classified as Calcium-Regulating Hormones and Agents.)
Vitamins
Organic substances that are required in small amounts for maintenance and growth, but which cannot be manufactured by the human body. (See all compounds classified as Vitamins.)
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
